Extraction of rare earth and heavy metals, using ionic solvents as extraction medium (A Review)
Didier Villemin1, Mohamed Amine Didi*2
1Laboratoire de Chimie Moléculaire et Thioorganique, UMR CNRS 6507, INC3M, FR 3038, Labex EMC3, ENSICAEN, 14050 Caen, France 2Laboratory of Separation and Purification Technology, Tlemcen University, Faculty of Sciences, Department of Chemistry,Box 119, Algeria
DOI : http://dx.doi.org/10.13005/ojc/290402
Article Received on :
Article Accepted on :
Article Published : 01 Dec 2013
A review. In this paper, separation of metals with solvent extraction, ionic liquid extraction phase and chelate extraction of metal ion were reviewed.
KEYWORDS:ionic liquid;solvent extraction;metals;chelate
Download this article as:| Copy the following to cite this article: Villemin D, Didi M. A. Extraction of rare earth and heavy metals, using ionic solvents as extraction medium (A Review). Orient J Chem 2013;29(4) |
| Copy the following to cite this URL: Villemin D, Didi M. A. Extraction of rare earth and heavy metals, using ionic solvents as extraction medium (A Review). Orient J Chem 2013;29(4). Available from: http://www.orientjchem.org/?p=1013 |
Introduction
Ionic liquids show unique properties such as nonvolatility (negligible vapor pressure), thermal stability, nonflammable nature, lower reactivity, strong ability to dissolve a large variety of organic and inorganic compounds… That’s why we should study the potential of it to the extraction of rare earth (lanthanides plus Sc and Y) and heavy metals. Indeed, heavy metals are a big problem for industrial companies who want to recycle the waste like mercury or cadmium to be more economically profitable as well as for the environment. There is a much bigger application. In fact, the extraction of rare earth metals is becoming more and more important due to the fact that it’s everywhere in technological devices like cellphones, television, computers, tablet, permanent magnets… So, we need to find a way to recycle it (to extract it) because China is almost the only supplier with more than 95% of the worldwide production of rare earth metals and they have decided to limit the exportation forcing people to increase the price of raw materials.[[i]]
In this report, we will study the different extractions techniques of rare earth metals and after the ones for heavy metals.
Extraction of rare earth metals
Different techniques are used to extract rare earth metals (REM) and the number of publications regarding this topic is increasing. In this part, we will focus on the extraction using extractants and another part using coordinated anions in the ionic liquid.
RTILs
Room-temperature ionic liquids RTILs are the first ionic liquids used to extract metals from aqueous solutions. They’ve become the base for most of the new ILs made after. Those ILs are made with imidazolium salts combined with different anions like PF6– or NTf2–. They are noted [CnMIM] with n equal to the number of carbon for the alkyl chain on the imidazole (Figure 1).
![Figure 1: [CnMIM][PF6] and [CnMIM][NTf2]](http://www.orientjchem.org/wp-content/uploads/2013/12/Vol29_No3_didi_extrctn_fig1-150x150.jpg) |
Figure 1: [CnMIM][PF6] and [CnMIM][NTf2]Click here to View figure |
This was the first potential ILs to be used for extraction but their problem is that the solubility of the lanthanides in it is really poor. A lot of examples are present in literature; one of them is the study made by Zuo et al in which they studied the potential of those RTIL to separate Cerium from aqueous solution containing thorium and lanthanides.[2]
The results showed quite good results for Cerium but when other lanthanides were tried, the results weren’t good enough. This study proves the need of the IL to have an extractant to facilitate the extraction.
Extraction with extractants
First method
Charged metal ions are usually insoluble in hydrophobic ionic liquids (IL), so extractants are necessary in the IL’s phase just as in organic solvent. First workswere about the use of CMPO: Octyl(phenyl)-N,N-diisobutyl carbamoylmethyl phosphine oxide(Figure 2).[3]
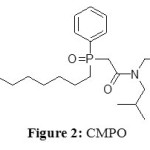 |
Figure 2: CMPOClick here to View figure |
As CMPO is a neutral extractants, an anion (such as NO3–) is required in acidic conditions to extract the cationic metal ions. The IL used was [BMIM][PF6] (Figure 3).
![Figure 3: [BMIM][PF6]](http://www.orientjchem.org/wp-content/uploads/2013/12/Vol29_No3_didi_extrctn_fig3-150x150.jpg) |
Figure 3: [BMIM][PF6] Click here to View figure |
Three rare earth metals are studied Ce3+, Eu3+ and Y3+with an increasing concentration of CMPO (black symbols) in comparison to the same extraction but in dodecane. They prove that the extractability of CMPO is highly better in IL than in a classic organic solvent (the volume of needed extractant is divided by 17).
The main problem with this kind of process was that after the metals comes to the IL phase, the extraction of it was hard due to the mechanism in the IL system. Indeed, the extraction involves cation exchange between the IL and aqueous phase, and the IL anions in the extracting phase which act as the counter anions of the extracted phase.
The AHA, DTPA, EDTA and citric acid have been chosen because it’s known that they form water-soluble complex with many metal ions. The optimum solution is the number 5. This was the first accomplished stripping of lanthanides in literature.
Improvements
Recently, others extractants were studied to extract specific lanthanides to improve this difficulty.[4] Some reviews already explained the role and the consequences of the addition of the extractants.[5]The different extractants are listed in the Table 1:
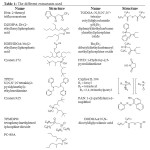 |
Table 1: The different extractants used Click here to View table |
The use of Htta was studied by Jensen et al in 2003.[6] With that goal, they studied trivalent lanthanides Ln3+ with Htta in RTILs [C4MIM][NTf2]. The big advantage of this extractant is the facility to strip the metal ion from the IL after the extraction. This was the first time the recovering of the metal ions was efficient in the literature.
D2EHPA
The first reproducible study was made by Yoon et al in 2010 as an extractant in RTILs [C4MIM][PF6].[7] Five different lanthanides elements were studied in their chloride form: CeCl3, NdCl3, SmCl3, DyCl3 and YbCl3.It was proved that the extractability of lanthanides increases as the ionic radius of the lanthanides decreases.
Moreover, a quite little quantity of D2EHPA is needed to have a good extraction making this process not expensive.
D2EHPA– HDEHDGA
A comparison of those two extractants has been made by Rout et al in 2011 particularly on Americium and Europium.[8-9] This study showed extraordinary power of extraction for these metals in RTIL like [OMIM[1]][NTf2].
This study revealed that there is an optimum pH but also that HDEHDGA is superior in terms of extractability of metals than D2EHPA. D2EHPA was before studied with other organophosphorus extractants but with those results, the tendency would be to use derivatives of HDEHDGA.
D2EHPA –C4mim
A novel room temperature ionic liquid (2-butyl-imidazolium di-(2-ethylhexyl) phosphate) ([C4mim][ D2EHPA], figure 4) was synthesized and tested as extractant in the mercury (II) liquid-liquid extraction.
The effects of parameters such as aqueous to organic phase’s volume ratio, metal concentration IL concentration, pH levels, ionic strength, and temperature were reported.[10]
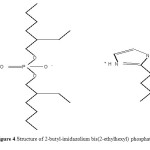 |
Figure 4.Structure of 2-butyl-imidazolium bis(2-ethylhexyl) phosphate Click here to View figure |
PC-88A
Continuously with the previous extractants, PC-88A (commercially available) was study as another organophosphorus extractant in RTILs [CnMIMI][Tf2N] (n=2, 4, 8, 12).[11] It appeared that higher extraction efficiency was given by [C8MIM].
However, the stripping was easier with [C12MIM] thanks to HNO3.
The extractability of the metal is controlled by pH of the aqueous phase. Indeed, in function of the metal, the pH is not the same. This property was evaluated by the pH0.5 corresponding to the pH needed to extract 50% of the metal.
The problem with that extractant is the poor solubility of it in the IL.
TPEN
TPEN4 is a neutral hexadentate ligand effectively coordinating with lanthanides. It’s soluble in ILs due to the high affinity of the pyridyl groups for the ILs and showed high extraction efficiency for lanthanides.
Cyanex 925
This extractant was used by Sun et al with the goal to separate Scandium from Yttrium and other lanthanides.[12] This extractant was first mixed with A336[1] as cation and NO3 as the anion. The only result of this study is the potential of Cyanex 925 to separate Scandium from Yttrium whereas for the other lanthanides, this extractant is not sufficient enough.
Cyphos IL 104
This extractant was used in RTIL [C8MIM][PF6].[13] The final IL was synthesized by a sol/gel method. The name of this new IL is B104SG. This extractant showed incredible results compared to the previous one Cyanex 925 (noted IL923SG).
With those results, this extractant needs to be more studied in the future.
HYD
This was first studied by Mallah et al and more recently by Jamali and al.[14-15] The difference between the two publications is the presence of PAN in the mixture to ameliorate the extractability of lanthanides with HYD. But this extractant didn’t prove real property to extract lanthanides but just to create IL which can be used after in a DLLME: Dispersive liquid-liquid micro-extraction.
DODGAA and TODGA
Those extractants are members of the same family and have similar properties. TODGAA was the first to be studied4it has high affinity for lanthanides in RTIL systems. Its extraction ability was strongly demonstrated although the selectivity in the lanthanide series was different.
However, the stripping and the recovery of the TODGA were not sufficient enough. That’s why its analog, DODGAA, was studied.[16] It has a molecular structure in which one of the two carbamoyl groups in TODGA is replaced by a carboxyl group. It is soluble in RTIL and has high affinity for rare earth metals. They try different RTIL to evaluate the potential of DODGAA ([CnMIM], n=4, 8, 12).
This study shows two important things:
– The high extractability for lanthanides
– The poor one for Zn.
This phenomenon can be explained by the tridentate diglycolamic acid trapping the metal with big radius. Moreover, high extraction performance was observed for the metal ions when ILs having shorter alkyl chains in the imidazolium cation.
Finally strippingwas easily accomplished with an acid solution in poor proportions such as nitric acid and the IL is after then reusable.
Another study has the same results as this one.[17] Indeed, they evaluate the extractability of the whole series of lanthanides and showed the pH0.5 too for different sizes of alkyl chain.
All the rare earth elements could be extracted at low pH, but like the previous analysis, the optimum conditions needs to be defined before the use of such a system.
Finally, the stripping of those elements was successfully demonstrated by Baba et alshowing once again that for transition metals, this extractant isn’t good whereas for lanthanides, the extraction and the recovery is quite good.[18]
This extractant is a promising one to the recovery of rare earth metals in waste solutions.
Recently, this extractant has been used to form membranes to ameliorate the extraction processes.
The others
Some of others extractants mentioned earlier have been studied such as Cyanex 272, TPMDPO added with Ph2Et2 or Bu2Et2. But they’re still not sufficient enough to be interesting in the recycling of rare earth metals from waste.[19]
Extractions using ionic liquids with coordinated anions.
The main problem of ionic liquids with weakly coordinated anions is that the metal salts are poorly soluble in them. A solution to this problem is to attach functional groups to the cationic core. That’s what Nockemann et al studied in a recent publication. [20] They showed that hydrophobic ionic liquids can be obtained by using the hexafluoroacetylacetonate anion [hfac]. In this study, cations are imidazolium salts and several member of this family have been studied: [Cxmin] (x=2,4,6,8,10,16,18) represented in the Figure 5.
![Figure 5: [Cnmim][hfac]](http://www.orientjchem.org/wp-content/uploads/2013/12/Vol29_No3_didi_extrctn_fig5-150x150.jpg) |
Figure 5: [Cnmim][hfac] Click here to View figure |
Those ionic liquids were prepared y a methathesis between an organic chloride salt and ammonium hexafluoroacetylacetonate in acetonitrile. The precipitate was filtered and the acetonitrille solvent was removed under reduced pressure. The residue was then redissolved in dichloromethane and washed with water and re-evaporated to obtain the pure ionic liquid. It is noticeable that the ionic liquid is better when the alkyl chain is a butyl or even longer to reinforce the hydrophobicity.
This study, based on neodymium, cobalt and copper, showed excellent results to extract metals. An aqueous solution of neodymium salt was poured on top of a sample of the ionic liquid [C4min][hfac] (with a butyl chain because it was the most hydrophobic compound). This extraction process occurred at the interface between the two liquid phases without stirring.
Instantaneously after the addition of neodymium, the ionic liquid changes color explaining by the fact that there is a transfer of Nd3+ between the two phases. The final step of this extraction is the formation of a complex [Nd(hfac)4] in the ionic liquid phase. A crystal structure of this complexe was made showing that the neodymium is surrounded by four bidentately coordinating diketone anions in a quadratic antiprismatic geometry.
The acidic hydrogen atom forms hydrogen bond with the oxygen making this complex even more stable. That’s why this process is so efficient.
Another study was made by Binnemans et al showing an improvement into solubilizing lanthanides into ionic liquids which is the main problem with lanthanides and ionic liquids.[23] With that intention, the synthesis of [Ln(NCS)4(H2O)y]3-x (x=6-8, y=0-2) was made by a metathesis procedure starting from stoichiometric amounts of lanthanides(III) perchlorate, ammonium thiocyanate, and thiocyanate ionic liquid with the corresponding imidazolium salt. The thiocyanate was introduced to ameliorate the solubilization of the lanthanide into the ionic liquid.
![Structure of [BMIM]x-3[Ln(NCS)x(H2O)y]3-x(x=6-8; y=0-2, BMIM=1-butyl-3-methylimidazolium.](http://www.orientjchem.org/wp-content/uploads/2013/12/Vol29_No3_didi_extrctn_fig6-150x150.jpg) |
Figure 6 : Structure of [BMIM]x-3[Ln(NCS)x(H2O)y]3-x(x=6-8; y=0-2, BMIM=1-butyl-3-methylimidazolium. Click here to View figure |
This study showed great results and almost all lanthanides was extracted by this process by the creation of a new ionic liquid. The main problem of this process is that the ionic liquid is not regenerate at the end of the process.
The second view of extracting rare earth metals with extractants are the phosphine oxide functionalized imidazolium. That’s what Vicente et al studied in 2012.[21]
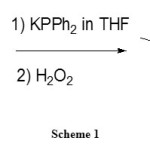 |
Scheme 1 Click here to View figure |
According to Scheme 1, they synthesized those ILs in two steps, using alkyl bromide-functionalized 1,2 dimethyl-imidazolium ionic liquid with [Tf2]– and potassium diphosphine. The resulting phosphine is oxidized by hydrogen peroxide to give the desired [DMImCnP(O)Ph2][NTf2].
When this new IL is in contact with a solution of Eu(hfac)3.6H2O, there is the formation of crystal which is the complex of Eu(hfac)4 (DMImCnP(O)Ph2). The structure of this complex is shown on Figure 7. It surprisingly indicates that those new ILs have the ability to be tuneable for lanthanides.
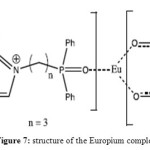 |
Figure 7: structure of the Europium complex Click here to View figure |
This property shows the potential of these new ILs in the extraction process of lanthanides, actinides or transition metals.
Finally, Nockemann et al are studying the extraction of lanthanides using trihexyltetradecylphosphonium [P666 (14)]+ (Figure 11) as cation to trap lanthanides by making new ionic liquids containing lanthanides, allowing it to extract the metal from water.[22]
![Figure 8: [P666 (14)]+](http://www.orientjchem.org/wp-content/uploads/2013/12/Vol29_No3_didi_extrctn_fig8-150x150.jpg) |
Figure 8: [P666 (14)]+ Click here to View table |
They studied a lot of lanthanides. But the major application of this is not the extraction of lanthanides but the realization of OLED materials for futures screens.
The structure of the final IL created is in the Figure 9. As we can see, this is the evolution of the previous Europium complex.
![Figure 9: Structure of the new IL composed with [P666(14)]+](http://www.orientjchem.org/wp-content/uploads/2013/12/Vol29_No3_didi_extrctn_fig9-150x150.jpg) |
Figure 9: Structure of the new IL composed with [P666(14)]+ Click here to View figure |
Task specifics ionic liquids
Task –Specific Ionic Liquids (TSIL) are ionic liquids with functional group covalently tethered to the cationic or anionic part. When the functional group has the ability to coordinate to the metal ion (preferably as a bidentate or a polydentate liguand), it is easier to dissolve metal oxides or metal salts into the ionic liquids. The major drawback of these TSIL is that they are often only accessible after multistep synthetic procedure. In a search for cheap and easily accessible cationic building blocks for ionic liquid alternatives, Binnemans et al tried to make TSIL with betaine.[23] Betaine is the trivial name for 1-carboxy-N,N,N-trimethylmethanaminium. (Figure 10).
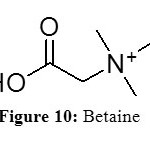 |
Figure 10: Betaine Click here to View figure |
With that intention, they introduced protonated betaine with bis(trifluoromethysulfonyl)imide, [Hbet][Tf2N], as a potential new TSIL for selective solubilization of metal oxides and metal salts (Figure 11).
![Figure 11: [Hbet][Tf2N]](http://www.orientjchem.org/wp-content/uploads/2013/12/Vol29_No3_didi_extrct_fig11-150x150.jpg) |
Figure 11: [Hbet][Tf2N] Click here to View figure |
The solubility of different metal oxides and metal salts has been studied. It appeared that this kind of TSIL is sensible of temperature and pH. Indeed, [Hbet][Tf2N] forms a two phases system with water at room temperature and slightly above but when it’s hotter, it’s a one-phase mixture.
This is the same problem with the pH. Indeed, when the pH is around 8, the TSIL and the water form a one-phase mixture whereas it’s a two-phase mixtures when the pH is equal to 1.5. This property shows the obligation to work in an acidic solution to extract metal oxides.
When those properties are established, it’s interesting to study the ability of [Hbet][Tf2N] to dissolve metal oxide. So, Binnemans et al, demonstrated that this system is able to dissolve the following oxides: Sc2O3, Y2O3, La2O3, Pr6O11, Nd2O3, Sm2O3, Eu2O3, Gd2O3, Tb4O7, Dy2O8, Ho2O3, Er2O3, Tm2O3, Yb2O3, Lu2O3, UO3, PbO, ZnO, CdO, HgO, CuO, Ag2O, NiO, PdO and MnO.[22] The metal oxide is mixed with [Hbet][Tf2N] and water and it’s stirred at room temperature for several hours. It appeared that not all metal oxides were soubilized in [Hbet][Tf2N]. Insoluble or very poorly soluble are iron, manganese and cobalt oxides as well as aluminum and silicon oxides. This property can be explain by the fact that the metal oxide reacts with the carboxylic acid group of the ionic liquid to form a carboxylate complex into water.
Then, the metal can be stripped from betaine by extracting the ionic liquid by an acidic aqueous solution of diluted hydrochloride or diluted nitric acid. The metal complex of protonated betaine is decomposed and the betaine ionic liquid is regenerated. The ratio of the metal content in the aqueous phase to the ionic liquid phase is 1:82 after the first extraction. After a second one, almost all the metal is extracted.
[Hbet][Tf2N] can be formed towards several synthetic routes. The main one is by reaction of the zwitterionic betaine with the acid hydrogen and Tf2NH. This reaction involves a simple proton transfer from the bis(trifluoromethylsulfonyl)imide to the more basic carboxylate group, so that the betaine will be protonated. This is a general method, applicable for the preparation of other betaine salts.
Extraction of heavy metals
Heavy metals are big challenges in industry because of the wastes produced by the fabrication of various products. Those wastes are a problem for environment: we need to extract them to reuse it. In this part, we will study different ways to accomplish that.
RTILs
The room-temperature ionic liquids, RTILs, are formed by imidazolium salts with alkyl chain. This is the same process that for the rare earth metals. The major products are made with BMIM or CMIM. The question was the influence of the alkyl chain on the extraction process. With that intention, Germani et al studied the effects of the alkyl chain on ILs composed of CnMIM (Figure 12) for the extraction of mercury.[24]
![Figure 12: [CnMIM] , n=4,6,8](http://www.orientjchem.org/wp-content/uploads/2013/12/Vol29_No3_didi_extrct_fig12-150x150.jpg) |
Figure 12: [CnMIM] , n=4,6,8 Click here to View figure |
Mercury was chosen because of its facility to be extracted by that kind of IL. This study showed that longer the chain is, better the extraction is (Table 2).
 |
Table 2: Ion percentage of Hg2+ at 60°C as a function of time Click here to View table |
The second parameter studied was the temperature. Indeed, they showed that lower the temperature is, higher the time of extraction is (Figure 13).
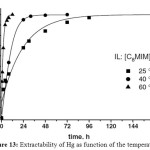 |
Figure 13: Extractability of Hg as function of the temperature Click here to View figure |
So, to have better conditions for that kind of extraction, we need to have long alkyl chain like octyl and temperature around 60°C.
Coordinated anions:
This is the same process as earlier, with rare earth metals.[24] After the reaction at the interface between [C4min][hfac] and a solution of Cobalt, two phases are observed and a change of color for the ionic liquid instantaneously after the addition of the cobalt complex.
A crystal structure was also made, showing that the cobalt(II) ion is octahedrally surrounded by three hexafluoroacetylacetonate anions.
Addition of extractant
2-aminothiophenol
An improvement of the previous works has been made by Lerlapwasin et al added the 2-aminothiophenol as an extractant into a RTIL 1-butyl-3-methylimidazolium hexafluorophosphate [BMIM][PF6]( Figure 14:2-aminothiophenol;Figure 15:[BMIM] [PF6].[25]
 |
Figure 14: 2-aminothiophenolFigure 15: [BMIM][PF6] Click here to View figure |
This addition of extractant improves the extraction percentage of Ni, Cu and Pb (Table 3: Values of Extraction).
 |
Table 3: Values of Extraction Click here to View table |
The percentage of Pb is still low but this is the best actually. After the extraction I the ionic liquid, we need to strip the metal from the IL. It was proved that with the addition of nitric acid, the stripping level is high.
Dithiozone
Dithiozone (Figure 16) are sulphur compounds used by Chen et al to improve extraction of metals in RTIL composed with imidazolium salts.[26-27]
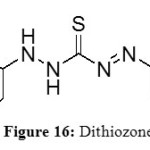 |
Figure 16: Dithiozone Click here to View figure |
This study is surprising in the sense that all others publications showed that the acidic pH is important but in this one, a basic pH is better to extract.
This process is proved to be reproducible and efficient towards some metals. Besides, the IL can be used again by stripping the metal from the IL by nitric acid.
Aza-Crown ether
This extractants have been studied by Huimin et al.[28] The structures are based on crown ether but with a nitrogen atom in the ring. The best results were obtained with N-Dodecyl Aza-18-crown-6 (Figure 17).
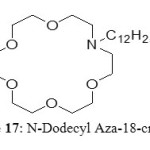 |
Figure 17: N-Dodecyl Aza-18-crown-6 Click here to View figure |
In way to study the extractability of some metals (Sr2+, K+, Cs+, Na+), the aza crown-ether was added in four different RTIL based on imidazolium salts [CnMIM][NTf2] (n=2,4,6,8). This study revealed the high power of aza crown-ether to extract particularly strontium.
Those results prove that the best extraction process is the addition of the aza crown-ether with an imidazolium salt composed with an alkyl chain of two or four atoms.
The problem with that process is the price of the extractant. That’s why they also studied the recycling of it. With a simple stripping of the metal with the addition of an aqueous phase containing nitric acid, the recovery of the metal is excellent which allows this process to be reproducible and profitable.
These observations demonstrate that the extraction system based on N-alkyl aza-18-crown-6 (especially dodecyl) in ILs offer not only easier recycling strategy but also better selectivity toward Sr2+.
Task specific ionic liquid
First try
TSIL for the elective liquid/liquid extraction of heavy metals from aqueous systems were first published by Rogers et al in 2001.[29] Functionalized imidazolium cations with thioether-, urea- or thiourea-derivatized side chains act as metal ligating moieties, whereas PF6– anions provides the desired water immiscibility. (Figure 18)
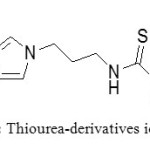 |
Figure 18: Thiourea-derivatives ionic liquid Click here to View figure |
Mercury(II) and Cadmium(II) were targeted with the goal to find alternative separations strategies for removing these toxic metal ions from the environment.[30] Those IL can be used directly as the bulk or may be doped as an extractant into less expensive IL such as [C4mim][PF6]. The best ILs used are listed in the following figure:
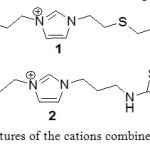 |
Figure 19: structures of the cations combined after with PF6- Click here to View figure |
In each case, they try to extract Hg2+ and Cd2+ directly using the IL and secondly by using [C4mim][PF6] with a 1:1 ratio and a pH at 1 or 7.
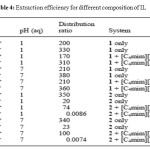 |
Table 4: Extraction efficiency for different composition of IL Click here to View table |
This table shows that 1 is a good extractants for both mercury and cadmium and with or without [C4mim][PF6]. However, with the derivative 2, the ratios are lower than with 1 and it’s much less effective at extraction of Cd2+. Besides, 2 doesn’t extract Cd2+ when it’s diluted with [C4mim][PF6]. So, 2 is more selective than 1 what’s could be a big advantage in an extraction process. The results about the superiority of 1 and 2 can be explained by the interaction between the S atom in the alkyl chain, able to react with the metal.
With those results, others derivatives have been made: Figure 20.
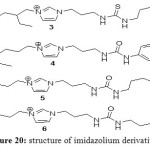 |
Figure 20: structure of imidazolium derivatives Click here to View figure |
All those IL have the same behavior as extractants for mercury and cadmium when they’re mixed in equal ratio with [C4mim][PF6]. In general, the ratios of Cd are lower than observed with Hg.
It is noticeable that both alkyl chains appear to affect the extraction[i] and that the adjustment of pH in acidic conditions doesn’t facilitate stripping of cadmium but practically doesn’t affect mercury.
It appears that increasing the alkyl chain length from butyl to octyl increases the hydrophobicity of the IL which ameliorates the ratio. Besides, the hydrophobic environment may prevent the hydrolysis.
To complete this study, tests have been made with different concentration of IL in [C4mim][PF6].
That kind of IL shows excellent results for extraction, but the problem is the instability of the hexafluorophosphate anion by hydrolysis producing toxic and corrosive HF or fluorides. Moreover, those ionic liquids are expensive due to the price of starting materials. Besides, we still need to find a way to strip the metal from the IL phase.
TOAH
TOAH, trioctylammonium are a new kind of ILs. TOAH (Figure 21) has they are composed of four alkyl chains on the ammonium with three octyl groups and one hydrogen.
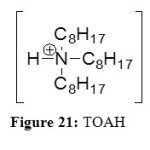 |
Figure 21: TOAH Click here to View figure |
A study from Katsuka et al shows excellent results for Platinum and Palladium. But the results were even better when there was two different ionic liquids of TOAH were mixed. The difference between the two ILs, was the anion.[31]
Another factor needs to be considered: the pH. Indeed, as the TOAH is an ammonium, the acidic proton on the ammonium can be removed if the pH isn’t acid enough. With that optic, Katsuka and co-workers have studied the power of pH on the extractability for several transition metals. The following table is giving results for some of them.
![Table 5: Extraction percentage of metals with a mixture of [TOAH][NO3] in [TOAH][NTf2] at 25°C](http://www.orientjchem.org/wp-content/uploads/2013/12/Vol29_No3_didi_extrctn_t5-150x150.jpg) |
Table 5: Extraction percentage of metals with a mixture of [TOAH][NO3] in [TOAH][NTf2] at 25°C Click here to View table |
This table shows that for each metal, the dependency of pH is not the same. In fact, for Pd, the concentration of HCL needs to be not too high whereas for the Cd it’s the contrary. A compromise for each metal needs to be made to find the perfect process.
Concerning the back extraction of the metal from the IL, a simple stripping with nitric acid is enough for most of the metals. But some of them, like the palladium, the recycling is not so efficient or we need to provide really high concentration of n nitric acid.
So, TOAH has high extractability in acidic condition than general hydrophobic ionic liquids and the major advantage is the control of the extractability by the composition of the mixture. Besides, Most of the metals extracted in the IL can be stripped through back-extraction with aqueous nitric acid solution to regenerate IL.
TOMATS
Trioctylmethylammonium thiosalicylate known as TOMATS are the better results today for the extraction of heavy metals adapted from the TOAH derivatives. The difference is the longer alkyl chain on the fourth substituent.
This is the novel high-performance, TSIL for the extraction of heavy metals from aqueous solution. To overcome the previous problems, TOMATS (Figure 22) contain no fluorine and is absolutely stable towards hydrolysis.
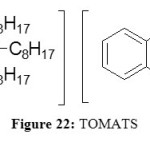 |
Figure 22: TOMATS Click here to View figure |
The efficiency of TOMATS can be explained by the chelating effect of the ortho-substituted carboxylate group relative to the thiol functionality.The bigger advantage is the facility to produce it at industrial scale. The first application is the extraction of copper from an aqueous Cu2+-tetramine phase. After the addition of TOMATS, two phases are seen: one with copper-free region and a dark copper-containing upper region. After shaking and separation of the phases, all the copper is extracted into the upper phase (third test tube).[32]
It is noticeable that sometimes separation takes quite a long time due to the high viscosity of TOMATS. This can be avoiding by the addition of immiscible organic solvent or by gentle heating of the mixture. Moreover, it can be improved by using centrifuge or adding small amount of sodium sulfate to the aqueous phase before shaking.
Most recently studies made by Hann and co-workers, used TOMATS as extraction ionic liquids from industrial water. The evaluation of the potential of anion functionalized IL as extracting agents for Cd, Ni, Hg, Pb… was studied.[31] With that intention, different TOMATS have been made, listed in the Table 6.
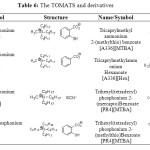 |
Table 6: The TOMATS and derivatives Click here to View table |
After 120 minutes of extraction, the ILs revealed high extraction efficiency for Ag, Hg and Pt, whereas only a limited fraction of As, Cr and Ni could be eliminated from the solutions.
The high extractability of mercury is due to the fact that it’s soluble either in functionalized or unfunctionalized ILs. The similar tendency is observed for Ag. Interestingly, Cd, Pb and Zn could be partially eliminated from the model solution by [PR4][TS] but there was no effect on the corresponding ammonium cation with the same anion [A336][TS]. So, it appeared that both cation ring and anion are strongly influencing the metal extraction from water. Generally, the extraction efficiency was higher for IL with [TS], [MTBA] or [SCN] than those with [BA], [hex] or [sal] indicating that the sulphur containing functional groups strongly affect the extraction efficiency. This property was already shown with others ILs.[33]
It is noticeable that there is a correlation between the contact time of IL and metals and the extraction efficiency.
Another factor influences the extraction efficiency is the pH. As it was demonstrated earlier, an acidic pH doesn’t facilitate the extraction.
According to those results, TSIL TOMATS-derivatives represent a good procedure for selective cleaning of contaminated waste water. In particular, thiol, thioether- functionalized are efficient for instance for communal waste cater. But, for industrial water, full of Zn, it’s recommended to use [A336][SCN]. Although those ILs are efficient, the problem still is the price of the extraction due to the price of the ILs.
That study is the following of a previous one made by Kalb et al in 2006, where they could extract heavy metals (principally mercury) from waste water using the commercially available TOMATS.[34] They also proved that TOMATS can extract heavy metals by forming metal-thiolate and additionally a complex-bond with the carboxylate group of the thiosalicylate anion, favoring the extraction process.
The high extractability of Ag Hg has been already demonstrated by Papaiconomou et alin 2008 with pyridines and piperidines derivatives ILs.[35] With that intention, they synthesized several new types of Ils summarized in the Figure 23.
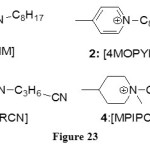 |
Figure 23 Click here to View figure |
After the study of RTILs 1, they studied the same property for the ILs 2 to 4.The results are that except for mercury, ILs containing pyridinium cations have poor extraction activity. However, TSILs containing a nitrile functionappear to be good and selective extracting agents for silver and palladium ions. Finally, piperidines cations don’t show any amelioration toward the extraction compared to the same with pyridinium cations.
Egorov et al. studied the extraction property of TOMATS toward Iron(III) and Copper(II).[36] The findings were that Iron and Copper are the most stable salicylate complexes so the results of extractions were high. However, for Nickel and Manganese, the ratios were low due to their poor stability as complexes.
Finally, binary mixtures of the ionic liquid betainium bis(trifluoromethylsulfonyl) imide and water show an upper critique solution temperature.[37] This solvent system has been used to extract metal ions by phase-transition extraction, using zwitterionic betaine as extractant. The system is efficient for the extraction of trivalent rare-earth, indium, and gallium ions. This new type of metal extraction system avoids problems associated with the use of viscous ionic liquids.
Conclusion
Nowadays, rare earth elements and heavy metals are big challenges for industries both environmentally and economically with the increasing demand of rare earth elements. This work listed the different techniques used to extract them from aqueous solution with the goal to reuse those metals after their extraction in wastes.
Some promising techniques have been demonstrated like the task-specific ionic liquids for both heavy metals and rare earth elements.
Moreover, processes about the recovery of the metal from the IL extractant phases have been realized with excellent results. The only problem which remains is the price of the TSILs.
Finally, new applications are coming up for the use of IL in the recovery of nuclear wastes giving encouraging results.[38]
References
- Ionic Liquids Today Issue 2-11, Monday, 19th September, 2011.
- Zuo, Y.; Liu, Y.; Chen, J.; Li, D. The separation of cerium(IV) from nitric acid solutions containing thorium(IV) and lanthanides(III) using pure [C8mim] PF6 as extracting phase. Ind. Eng. Chem. Res. 2008, 47, 2349-2355.
- Nakashima, K.; Kubota, F.; Maruyama, T.; Goto, M. Ionic Liquids as a Novel Solvent for Lanthanide Extraction. Anal. Sci., 2003, 19, 1097-1098.
- Yang, F; Kubota, F.; Kamiya, N; Goto; M. Extraction and Separation of Rare Earth Metal Ions with DODGAA in Ionic liquids. Solv. Extr. Res. Dev. Jpn. 2012, 19 (1), 17-28.
- Baba, Y.; Kubota, F.; Kamiya, N.; Recent Advances in Extraction and Separation of Rare-Earth Metals Using Ionic Liquids. Journal of Chemical Engineering of Japan, 2011, 44(10), 679-685.
- Jensen, M. P.; Neuefeind, J.; Beitz, J. V.; Skanthakumar, S.;Soderholm, L. Mechanisms of Metal Ion Transfer into Room-Temperature Ionic Liquids: The Role of Anion Exchange. J. Am. Chem. Soc., 2003, 125, 15466-15473.
- Yoon, S. J.; Lee, J. G.; Tajima, H.; Yamasaki, A.; Kiyono, F.; Nakazato, T.; Tao, H. Extraction of lanthanide ions from aqueous solution by bis(2-ethylhexyl)phosphoric acid with room-temperature ionic liquids. J. Ind. Eng. Chem. 2010, 16, 350– 354.
- Rout, A.; Karmakar, S.; Venkatesan, K. A.; Srinivasan, T. G.; Vasudeva Rao, P. R. Room temperature ionic liquid diluent for the mutual separation of europium(III) from americium(III). Sep. Purif. Technol., 2011, 81, 109–115.
- C. Michael and R. W. Sugumar., Orient. J. Chem., 29(3), 1149-1154, 2013
- Didi, M.A.; Medjahed, B.; Benaouda, W. Adsorption by Liquid–Liquid extraction of Hg (II) from aqueous solutions using the 2-butyl-imidazolium di-(2-ethylhexyl) phosphate as ionic liquid. American Journal of Analytical Chemistry, 2013, 4, ** (in press) Published Online July 2013 (http://www.scirp.org/journal/ajac).
- Kubota, F.; Koyanagi, Y.; Nakashima, K.; Shimojo, K.; Kamiya, N.; Goto, M. Extraction of lanthanide ions with an organophosphorous extractant into ionic liquids. Solv. Extr. Res. Dev. Jpn, 2008, 15, 81-87.
- Sun, X. Q.; Ji, Y.; Guo, L.; Chen, J.; Li, D. Q. A novel ammonium ionic liquid based extraction strategy for separating scandium from yttrium and lanthanides. Sep. Purif. Technol., 2011, 81, 25-30.
- Liu, Y. H.; Zhu, L. L.; Sun, X. Q.; Chen, J.; Luo, F. Silica Materials Doped with Bifunctional Ionic Liquid Extractant for Yttrium Extraction. Ind. Eng. Chem. Res., 2009, 48(15), 7308-7313.
- Mallah, M.H. ; Shemirani, F. ; Maragheh, M.G. Ionic Liquids for Simultaneous Preconcentration of Some Lanthanoids Using Dispersive Liquid−Liquid Microextraction Technique in Uranium Dioxide Powder. Environ. Sci. Technol., 2009, 1947-1951.
- Mallah, M. H. ; Shemirani, F. ; Maragheh, M. G. ;Jamali, M. R. Evaluation of synergism in dispersive liquid–liquid microextraction for simultaneous preconcentration of some lanthanoids. J. Mol. Liq., 2010, 151, 122–124.
- Kubota, F.; Shimobori, Y.; Baba, Y.; Koyanagi, Y.; Shimojo, K.;Kamiya, N.; Goto, M. Application of Ionic Liquids to Extraction Separation of Rare Earth Metals. J.Chem. Eng. Japan, 2011, 44, 307-312.
- Yang, F.; Baba, Y.; Kubota, F.; Kamiya, N.;Goto, M. Extraction and Separation of Rare Earth Metal Ions with DODGAA in Ionic liquids. Extr. Res. Dev. Jpn, 2012, 19, 69-76
- Baba, Y.; Kubota, F.; Kamiya, N.; Goto, M. Selective Recovery of Dysprosium and Neodymium Ions by a Supported Liquid Membrane Based on Ionic Liquids. Solv. Extr. Res. Dev. Jpn, 2011, 18, 193-198.
- Mehdi, H.; Binnemans, K.; Van Hecke, K.; Van Meervelt, L.; Nockemann, P. Hydrophobic ionic liquids with strongly coordinating anions. Chem. Commun., 2010, 46, 234-236.
- Nockemann, P.; Thijs, B.; Postelmans, N.; Van Hecke, K.; Van Meervelt, L.; Binnemans, K. Anionic rare-earth thiocyanate complexes as building blocks for low-melting metal-containing ionic liquids. J. Am. Chem. Soc., 2006, 128 (42), 13658-13659.
- Vicente, J.A.; Mlonka, A.; Nimal Gunaratne, H.Q.; Swadźba-Kwaśny, M.; Nockemann, P. Phosphine oxide functionalised imidazolium ionic liquids as tuneable ligands for lanthanide complexation. Chem. Commun., 2012, 48, 6115-6117.
- Peter Nockemann. Metals in ionic liquids:positive or negative? conference 2012
- Nockemann, P.; Thijs, B.; Pittois, S.; Thoen, J.; Glorieux, C.; Van Hecke, K.; Van Meervelt, L.; Kirchner, B.; Binnemans, K. Task-specific ionic liquid for solubilizing metal oxides . J. Phys. Chem. B, 2006, 110, 20978-20992.
- Germani, R.; Mancini, M. V.; Savelli, G.; Spreti, N. Mercury extraction by ionic liquids: temperature and alkyl chain length effect. Tetrahedron Lett., 2007, 48, 1767–1769.
- Lertlapwasin, R.; Bhawawet, N.; Imyim, A. Fuangswasdi, SIonic liquid extraction of heavy metal ions by 2-aminothiophenol in 1-butyl-3-methylimidazolium hexafluorophosphate and their association constants. Sep. Purif. Technol., 2010, 72(1), 70-76.
- Wei, Guor-Tzo; Yang, Zusing; Chen, Chao-Jung. Room temperature ionic liquid as a novel medium for liquid/liquid extraction of metal ions . Analytica Chimica Acta, 2003, 488(2), 183-192.
- A. Yazolani and Mona Akbari., Orient. J. Chem., 28(3), 1219-1227, 2012
- Huimin,L.; Sheng,D.; Bonnesen, P.V. Solvent Extraction of Sr2+ and Cs+ Based on Room-Temperature Ionic Liquids Containing Monoaza-Substituted Crown Ethers. Analytical Chemistry, 2004,76 (10), 2773-2779.
- Visser, A. E.; Swatloski, R. P.; Reichert, W. M.; Mayton, R.; Sheff, S.; Wierzbicki, A.; Davis, Jr. J. H.; Rogers, R. D. Task-specific ionic liquids for the extraction of metal ions from aqueous solutions. Chem. Commun. 2001, 135-136.
- Visser, A. E.; Swatloski, R. P.; Reichert, W. M.; Mayton, R.; Sheff, S.; Wierzbicki, A.; Davis, J. H., Jr.; Rogers, R. D. Task Specific Ionic Liquids Incorporating Novel Cations for the Coordination and Extraction of Hg 2+ and Cd 2+: Synthesis, Characterization and Extraction Studies. Environ. Sci. Technol. 2002, 36, 2523–2529.
- Katsuta, S.; Yoshimoto, Y.; Okai, M.; Takeda, Y.; Bessho, K. Selective Extraction of Palladium and Platinum from Hydrochloric Acid Solutions by Trioctylammonium-Based Mixed Ionic Liquids. Ind. Eng. Chem. Res., 2011, 50 (22), 12735-12740.
- Ionic Liquids Chem Files 2006, Vol. 6, number 9.
- Fischer, L.; Falta, T.; Koellensperger, G.; Stojanovic, A.; Kogelnig, D.; Galanski, M.; Krachler, R.;Keppler, B.K., Hann, S. Ionic liquids for extraction of metals and metal containing compounds from communal and industrial waste water. Water Research 2011, 45(15), 4601-4614.
- Kalb, R.S.; Krachler, R.; Keppler, B.K.; In: Ho¨ flinger, W. (Ed.), Chemical Industry and Environment V, 2006, vol. I, Vienna, Austria.
- Papaiconomou, N.; Lee, J.M.; Salminen, J.; Von Stosch, M.; Prausnitz, J.M. Selective Extraction of Copper, Mercury, Silver, and Palladium Ions from Water Using Hydrophobic Ionic Liquids. I. Ind. Eng. Chem. Res. 2008, 47, 5080-5086.
- Egorov, V.M.; Djigailo, D.I.; Momotenko, D.S.; Chernyshov, D.V.; Torocheshnikova, I.I.; Smirnova, S.V.; Pletnev, I.V. Task-specific ionic liquid trioctylmethylammonium salicylate as extraction solvent for transition metal ions. Talanta, 2010, 80, 1177-1182.
- Hoogerstraete, T. V.; Onghena, B.; Binnemans, K.; Homogeneous Liquid-Liquid Extraction of Metal Ions with a Functionalized Ionic liquid. Journal of Physical Chemistry Letters, 2013, 4(10), 1659-1663.
- Nockemann, P.; Servaes, K.; Van Deun, R.; Van Hecke, K.; Van Meervelt, L.; Binnemans, K.; Görller-Walrand, C. Speciation of Uranyl Complexes in Ionic Liquids by Optical Spectroscopy. Inorganic Chemistry, 2007, 46 (26), 11335-11344.

This work is licensed under a Creative Commons Attribution 4.0 International License.









