Eggshell as a Green Catalyst in Water for the Easy and Very Effective Synthesis of Thioamide Derivatives
1Department of Chemistry, Arts and Science College, Bhalod (M. S.) India.
2 Department of Chemistry, Dhanaji Nana Mahavidyalaya, Faizpur (M. S.) India.
Corresponding Author E-mail: drgrc76@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/420129
ABSTRACT:The Preparation of thioamide derivatives in aqueous media at 100 oC has been accomplished in a straightforward, easy, and very effective manner. Using recyclable catalyst Eggshell Powder in a single vessel, a reaction that involves multiple components, including secondary amines, substituted aldehydes, and Sulphur powder, produced a total of nine compounds, demonstrating the method's wide substrate range. Water provides good to exceptional product yields, is easy to use, eggshell powder is a non-toxic catalyst, and water is a green solvent. ease of operation, environmental friendliness, and simple product isolation are the key characteristics of the current methodology. A crucial component of green synthesis, catalytic activity, was not significantly reduced throughout the recovery and several reuses of the utilized catalyst.
KEYWORDS:Eggshell Powder; Eco-friendliness; Green synthesis; Multicomponent reaction; Secondary amines
Introduction
The demand for ecologically friendly settings, green paths, cheap cost, and rapid reaction times in organic chemical synthesis is increasing. As a result, it is better to use natural or bio-derived biodegradable catalysts1. Because of the higher toxicity and cost-effectiveness of organic compounds, it is critical to reuse and recycle all chemicals and catalysts as much as possible for a brighter and more sustainable future. Given the challenges of the future and the hazard level of harmful chemicals, researchers are designing catalysts or chemical intermediates with more sustainable and eco-friendly alternatives2. The catalyst’s important job is to increase the reaction rate without changing the thermodynamic equilibrium of a specific reaction3-4. As a result, the current requirement to create an alternative catalyst system for an eco-friendly environment that is safer for humans and has a lower toxicitylevel5-6. The two most common types of catalyst systems are homogeneous and heterogeneous. At the same time, We shall be concentrating on heterogeneous catalysis and its applications in this review. environmentally benign and organically sourced eggshell powder7-8 or calcined eggshell9. Homogeneous catalysis is well known in synthetic organic chemistry, although it has a few drawbacks, including difficulty separating soluble complexes from the reaction mixture, non-recyclability, the likelihood of complex breakdown, decreased catalytic activity, and so on10-11.
Thioamides are a category in which the presence of a sulphur atom is attached to a carbonyl group for the purpose of distinguishing organic molecules from other ones. They are analogues of amides, with the oxygen atom replaced by sulfur12. Thioamides are important intermediates in the synthesis of a variety of pharmaceuticals and agrochemicals, and they are also essential in material science13.
The use of thioamides in natural and manufactured compounds has several benefits because of the subtle but dramatic changes in amide interactions that result from changing only one atom. Thioamides are often used as chemical synthesis intermediates because of their higher reactivity to both nucleophiles and electrophiles than their amide cousins, which is ascribed to a weaker carbonyl link. Their usefulness in a variety of chemical situations is further increased by the fact that thioamides exhibit a higher affinity for particular metals than amides14.
Thioamides have an important role in both chemical and biological systems. They are essential components of many therapeutic substances and act as building blocks for the synthesis of pharmacologically active molecules15. These compounds, which include nitrogen and sulphur heterocycles, are critical for generating effective anticancer therapies and enzyme inhibitors16.
Thioamides are chemical cousins of a common protein component that has received extensive scientific research. They are significant because they contribute to the production of various FDA-approved medications, such as those for thyroid disorders and tuberculosis. Scientists also employ them to create other types of compounds and change peptides.17
The traditional procedures for synthesising thioamides frequently involve extreme conditions, such as high temperatures and the use of hazardous chemicals, which can be harmful to the environment.18 In recent years, the emphasis has switched to building more sustainable and efficient synthetic pathways that reduce environmental damage. One such advancement is the production of thioamide derivatives with a heterocyclic secondary amine, such as morpholine/piperidine/pyrrolidine. This approach takes use of the nucleophilic substitution reaction, in which morpholine/piperidine/pyrrolidine functions as a nucleophile, enabling the conversion of carbonyl compounds into thioamides.
The process is noted by its simplicity, great efficiency, and lack of complicated catalysts and solvents. It marks a substantial advance in green chemistry, offering a viable and environmentally acceptable method of thioamide production. This approach not only adheres to the principles of atom economy, but it also provides a varied pathway to a wide range of thioamide derivatives, which are useful in a variety of chemical industries. Consequently, the present study has determined that it is worthwhile to synthesise some Thioamide derivatives using morpholine/piperidine/pyrrolidine.
Materials and Methods
They are all prevalent solvents and reagents received from vendors and were not subjected to additional purification. The preparation of eggshell powder followed earlier reports in the literature. The process of thin-layer chromatography (TLC) employing plates made of silica gel were utilized in order totrack a reaction’s growth, and the “Digital Analab Scientific Instrument” was used to record the melting points. The “Alpha II Bruker spectrophotometer” was used to measure the FT-IR spectra. A “Bruker Advance Neo 500 MHz spectrometer” was used to record 1HNMR and 13CNMR spectra in CDCl3. Using a “Waters Q-ToF Micro LC-MS spectrometer,” mass spectra were acquired.
Experimental
Catalysts preparation
The waste eggshell powder catalyst was prepared in accordance with the procedure that was previously reported19. Household waste eggshells are collected, and the inner membrane is manually removed. The shells are then rinsed with scalding water. After boiling, dry the material and pulverise it in a mortar and pestle. After the powder has formed, dry it at 150°C for 4 hours. The basic powder material that was obtained was depicted as eggshell powder (ESP).
Synthesis Methods for Thioamide Derivatives
In a 25 ml round bottom flask Substituted aldehyde (2.0 mmol), morpholine (4.0 mmol), sulfur (6.0 mmol) were taken, then 10 ml water was mixed in it and stirred for a few seconds and then 20 mol℅ Eggshell Powder was added as a catalyst. The reacting components were refluxed under stirring at 100°C, and Thin-layer chromatography was employed should keep monitoring on the reaction. In consideration of the results of the reaction, the solid was obsessed in a flask, after that, the bulk of the reaction mass was put in a mixture of water with ice.. Under continuous stirring, A filtration process was used to gather the material. and drying. Later on, the product underwent a process of purification that involved recrystallization with hot ethanol.
Spectral data
morpholino(phenyl)methanethione
IR υmax Cm-1: 1475.11, 1284.03, 1104.92, 1020.41, 752.68, 655.89. 1HNMR (500 MHz, CDCl3): δ7.32-7.38(3H,m),7.26-7.28(2H,t),4.43-4.45(2H,t),3.87-3.89(2H,t),3.63-3.65(2H,t),3.58-3.60(2H,t). 13CNMR (500 MHz, CDCl3): 201.08, 142.50, 128.87, 125.89, 77.29, 77.03, 76.78, 66.75, 66.53, 52.51, 49.55. Mass (m/z): 208.27.
(1H-indol-3-yl) (morpholino)methanethione
IR υmax Cm-1: 3098.72, 1624.46, 1510.47, 1436.19, 1235.00,1121.38, 751. 1HNMR (500 MHz, CDCl3):δ 12.12(1H,s),9.94(4H,s),8.28(1H,s),7.51(1H,s),7.51-7.52(2H,t),7.20-7.28(2H,t),3.33(1H,s),2.50-2.51(2H,t). 13CNMR (500 MHz, CDCl3): 184.82, 138.30, 136.93, 124.00, 123.33, 121.99, 120.69, 118.05, 112.29, 39.92, 39.67, 39.42, 38.91. Mass (m/z): 247.32.
(4-chlorophenyl) (morpholino)methanethione
IR υmax Cm-1: 1478.51, 1287.19, 1103.71,811.48, 719.05. 1HNMR (500 MHz, CDCl3): δ 7.32-7.35(2H,t),7.22-7.24(2H,t),4.40-4.42(2H,t),3.87-3.98(2H,t),3.63-3.65(2H,t),3.58-3.60(2H,t). 13CNMR (500 MHz, CDCl3): 199.62, 140.77, 134.96, 128.79, 127.42, 77.30, 76.79, 66.68, 66.49, 52.60, 49.61. Mass (m/z): 242.14.
phenyl(piperidin-1-yl) methanethione
IR υmax Cm-1: 1477.87, 1431,12, 1285.17, 1002.26, 852.25, 686.43. 1HNMR ( 500 MHz, CDCl3): δ 7.28-7.35(3H,m),7.24-7.27(2H,t),4.34-4.36(2H,t),3.49-3.52(2H,t),1.79-1.84(2H,t),1.72-1.73(2H,t),1.53-1.58(2H,t). 13CNMR ( 500 MHz, CDCl3): 199.59, 143.41, 128.38, 125.42, 77.30, 76.80, 53.15, 50.59, 26.87, 25.48, 24.15. Mass (m/z): 194.32.
(1H-indol-3-yl) (piperidin-1-yl) methanethione
IR υmax Cm-1: 3098.72, 1537.44, 1263.65, 1069.14, 802.20, 710.00. 1HNMR (500 MHz, CDCl3): δ 10.07(1H,s),8.32-8.89(4H,m),7.85(1H,s),3.87-3.89(4H,t),3.63-3.64(4H,t),3.58-3.60(2H,t). 13CNMR (500 MHz, CDCl3): 185.19, 136.68, 135.31, 124.46, 123.08, 122.00, 121.12, 119.77, 114.21, 113.111.53, 77.27, 77.02, 76.77. Mass (m/z): 245.35.
(4-bromophenyl) (piperidin-1-yl) methanethione
IR υmax Cm-1: 1526.47, 1418.96, 1344.45, 816.04. 1HNMR (500 MHz, CDCl3): δ 7.46-7.49(2H,t),7.13-7.26(2H,t),4.31-4.34(2H,t),3.49-3.52(2H,t),1.80-1.83(2H,t),1.72-1.77(2H,t),1.54-1.59(2H,t). 13CNMR (500 MHz, CDCl3): 198.18, 142.17, 131.59, 127.20, 122.48, 77.28, 76.78, 53.24, 50.66, 26.89, 25.45, 24.11. Mass (m/z): 285.18.
phenyl(pyrrolidin-1-yl) methanethione
IR υmax Cm-1: 1629.71, 1432, 1237.34, 1125.10, 872.79, 751.28, 635.24. 1HNMR (500 MHz, CDCl3): δ 7.26-7.37(5H,m),3.96-3.99(2H,t),3.45-3.48(2H,t),2.05-2.11(2H,t),1.55-1.99(2H,t). 13CNMR (500 MHz, CDCl3): 197.36, 144.03, 128.70, 128.30, 125.64, 77.29, 77.03, 76.78, 53.76, 53.39, 26.49. Mass (m/z): 192.22.
(1H-indol-3-yl) (pyrrolidin-1-yl) methanethione
IR υmax Cm-1: 3098.72, 1537.44, 1441.75, 1380.49, 869.61, 732.23. 1HNMR (500 MHz, CDCl3): δ 10.01(1H,s),9.12(1H,s),7.38-7.42(2H,t),7.29-7.32(2H,t),4.05-4.08(2H,t),2.06-2.12(2H,t),1.92-1.97(4H,t). 13CNMR (500 MHz, CDCl3): 190.60, 135.60, 122.81, 120.97, 111.64, 77.27, 77.02, 76.77, 53.78, 53.69, 26.47, 24.75. Mass (m/z): 231.45.
(3,4-dihydroxyphenyl) (pyrrolidin-1-yl) methanethione
IR υmax Cm-1: 1440.64, 1258.53, 760.51, 699.11. 1HNMR (500 MHz, CDCl3): δ 9.66(1H,s),8.29(1H,s),7.16-7.35(2H,t),6.96-6.98(2H,t),6.83-6.86(2H,t),6.68-6.75(2H,t),6.28(1H,s),5.45(1H,s),5.25(1H,s). 13CNMR (500 MHz, CDCl3): 202.08, 141.50, 128.87, 125.89, 77.29, 77.03, 76.78, 66.75, 66.53, 52.51, 49.55. Mass (m/z): 223.29.
Results and Discussion
We describe a simple, practical, and extremely well-structured Synthesis of multiple components in a single pot method for thioamide derivatives in this article. This article describes how several aldehydes, secondary amines like ‘morpholine/piperidine/pyrrolidine and sulfur powder’ react at 100 degrees Celsius in H2O when the catalyst is recyclable and disposable. Eggshell Powder is present. Our continuous attempts to create environmentally friendly synthetic processes, which are aided by Eggshell Powder, include this technique. The first scheme shows the main plan to synthesize thioamide derivatives.
Reaction scheme-1
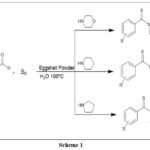 |
Scheme 1 Click here to View Scheme |
  Scheme – 1 With the use of sulfur powder, morpholine, and benzaldehyde as a model reaction as an initial step, we investigated the influence of temperature on the synthesis of “morpholino(phenyl)methanethione” in an effort to elevate the reaction conditions and evaluate the effectiveness of eggshell powder as a catalyst. Temperature was a key factor in this transformation; at low temperatures, a restricted quantity of product was produced, and a longer reaction time was needed. It was discovered that while reaction time decreased, the level of yields improved as the temperature rose from ambient temperature to 100 degrees Celsius. All of the reactions were conducted at 100 oC since we found that this temperature produced the best results (Table 1, item 5).
Model Reaction Scheme-2
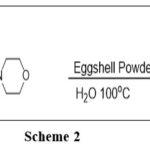 |
Scheme 2 Click here to View table |
  Table 1: Temperature’s impact on morpholino (phenyl) methanethione synthesis reaction time and yields
| Entry | Temp. (oC) | Time (h) | Yieldb (%) |
| 1 | R. T. | 24 | No reaction |
| 2 | 40 | 12 | 30 |
| 3 | 60 | 8 | 45 |
| 4 | 80 | 5 | 78 |
| 5 | 100 | 2.5 | 90 |
| aReaction Conditions:- Benzaldehyde(2mmol), morpholine(4mmol), sulfur powder(6mmol), Eggshell powder catalyst(20 mol%) and H2O (10 mL) at 100oC. bIsolated yield. | |||
At 100 degrees Celsius in 10 milliliters of H2O, the amount of catalyst was assessed in the model reaction (Scheme 2). The optimal outcome was achieved with 20 mol% eggshell powder (Table 2, entry 5), providing 90% of the yield in 150 minutes. The speed of reaction and product yield were not influenced by the addition of more catalyst. (Table 2, entry 6).
Table 2: Utilizing varying amounts of catalyst to optimize the reaction to synthesize morpholino(phenyl) methanethione at 100oC in H2O.
| Entry | Catalyst (mol %) | Time (hrs) | Yieldb (%) |
| 1 | — | 24 | 20 |
| 2 | 5 mol % | 12 | 42 |
| 3 | 10 mol % | 06 | 62 |
| 4 | 15 mol % | 04 | 82 |
| 5 | 20 mol% | 2.5 | 90 |
| 6 | 25 mol % | 2.5 | 90 |
| aReaction conditions: benzaldehyde (2 mmol), morpholine (4 mmol), sulfur powder (6 mmol), Eggshell powder catalyst (20 mol%) and H2O (10 mL) at 100 oC. b Isolated yield. | |||
These findings suggest that the catalyst is a critical component of this reaction. Therefore, we implemented an effective plan on a diverse array of secondary amines and aldehydes. All of the designed The products were obtained in exceptional to excellent yields. by performing all the reactions at 100oC with 20 mol % of Eggshell Powder in H2O for 120-180 minutes. Additionally, the amount of sulfur was optimized. At first, using 1 mmol of sulfur results in a small product yield (35%) and a longer reaction time (330 min). It was shown that the product yield improved with decreasing time as the quantity of sulfur increased from 1 to 6 mmol. With a 90% yield in 155 minutes, 6 mmol of sulfur powder produced the greatest results (Table 3, entry 6).
Table 3: Reaction optimization with varying quantity of Sulfur powder to produce morpholino (phenyl) methanethione in H2O at 100°C
| Entry | Sulfur(mmol) | Time(min) | Yieldb(%) |
| 1 | 1mmol | 330 | 35 |
| 2 | 2mmol | 260 | 45 |
| 3 | 3mmol | 210 | 68 |
| 4 | 4mmol | 190 | 72 |
| 5 | 5mmol | 175 | 82 |
| 6 | 6mmol | 155 | 90 |
| 7 | 7mmol | 155 | 90 |
| aReaction conditions: Benzaldehyde (2mmol), Morpholine (4mmol), Eggshell Powder (20 mol %) and water (10mL) at 100ºC. bIsolated yield. | |||
There was no discernible change in yield or reaction time when the quantity of sulfur powder was increased from 6 to 7 mmol (Table 3, entry 7). These findings show that 6 mmol of sulfur powder is sufficient to finish the process. After that, we applied the ideal conditions to a range of secondary amines and aldehydes, and we used 6 mmol of sulfur powder for every reaction. A diverse array of structurally divergent aldehydes (Scheme-2) were chosen to investigate the generality and scope of the Eggshell Powder-promoted thioamide synthesis. These aldehydes are characterized by a wide range of substituents and secondary amines. In Table 4 (entries 4A- 4I), the results are summarized. All aldehydes and secondary amines were subjected to the reaction at 100°C, with H2O serving as the solvent. All conversions were completed within 120-180 minutes, with yields that ranged from good to outstanding. Additional increases in reaction time did not have a substantial impact on the yields.
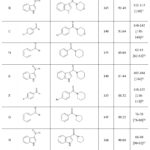 |
Table 4: To synthesize of a variety of thioamide derivatives in the existence of Eggshell Powder at one hundred degree Celsius. Click here to View Table |
  Conclusion
In conclusion, a straightforward, workable, and well-structured procedure has been devised to facilitate the synthesis of a number of thioamide derivatives with biological interest. With this technique, readily accessible aromatic aldehydes and secondary amines are reacted in a single pot with eggshell powder acting as a catalyst. Using morpholine, piperidine, and pyrrolidine, many thioamide compounds were produced in good to exceptional yields in 120–180 minutes. A non-toxic catalyst, a shorter reaction time, and the simplicity of recovery and catalyst reuse are only a few of the method’s noteworthy advantages. The biodegradable eggshell powder’s reusability is the main advantage of the current process.
Acknowledgement
We are grateful to the Principal, Arts and Science College, Bhalod for providing laboratory facilities and encouragement, and SAIF, Punjab University, Chandigarh for the Characterization facility.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Hosseinikhah, S. S.; Bi; Salehi, N.; RSC Advances 2020, 10 (66), 40508–40513. https://doi.org/10.1039/d0ra07199d.
CrossRef - Möllersten, K.; Yan, J.; Westermark, M. Energy 2003, 28 (7), 691–710. https://doi.org/10.1016/s0360-5442(03)00002-1.
CrossRef - Roy, I.; Gupta, M. N. Enzyme and Microbial Technology 2005, 36 (7), 896–899. https://doi.org/10.1016/j.enzmictec.2005.01.022.
CrossRef - Sugihara, T.; Yamada, M.; Ban, H.; Yamaguchi, M.; Kaneko, C. Angewandte Chemie International Edition 1997, 36 (24), 2801–2804. https://doi.org/10.1002/anie.199728011.
CrossRef - Evans, M. G.; Polanyi, M. Transactions of the Faraday Society 1936, 32 (0), 1333–1360. https://doi.org/10.1039/TF9363201333. CrossRef
- Hepburn, C. Polyurethane Elastomers; 1992. https://doi.org/10.1007/978-94-011-2924-4.
CrossRef - Mizuno, N.; Misono, M. Chemical Reviews 1998, 98 (1), 199–218. https://doi.org/10.1021/cr960401q.
CrossRef - Hincke, M., T. Frontiers in Bioscience 2012, 17 (1), 1266. https://doi.org/10.2741/3985.
CrossRef - Patil, S.; Jadhav, S. D.; Shinde, S. K. Organic Chemistry International 2012, 1–5. https://doi.org/10.1155/2012/153159.
CrossRef - Sabbe, M. K.; Reyniers, M.F.; Reuter, K. Catalysis Science & Technology 2012, 2 (10), 2010. https://doi.org/10.1039/c2cy20261a. CrossRef
- Sheldon, R. A.; Dakka, J. Catalysis Today 1994, 19 (2), 215–245. https://doi.org/10.1016/0920-5861(94)80186-x.
CrossRef - Novak, I.; Klasinc, L.; McGlynn, S. P. Journal of Electron Spectroscopy and Related Phenomena 2016, 209, 62–65. https://doi.org/10.1016/j.elspec.2016.03.005.
CrossRef - Liao, Y.; Wang, M.; Jiang, X. Current Opinion in Chemical Biology 2023, 75, 102336–102336. https://doi.org/10.1016/j.cbpa.2023.102336.
CrossRef - Mahanta, N.; Szantai-Kis, D. M.; Petersson, E. J.; Mitchell, D. A. ACS Chemical Biology.2019, 14 (2), 142–163. https://doi.org/10.1021/acschembio.8b01022.
CrossRef - Huang, G.; Tomasz Cierpicki; Jolanta Grembecka. European Journal of Medicinal Chemistry 2024, 277, 116732–116732. https://doi.org/10.1016/j.ejmech.2024.116732.
CrossRef - Dilworth, J. R.; Hueting, R. Inorganica Chimica Acta 2012, 389, 3–15. https://doi.org/10.1016/j.ica.2012.02.019.
CrossRef - Wang, L.; Wang, N.; Zhang, W.; Cheng, X.; Yan, Z.; Shao, G.; Wang, X.; Wang, R.; Fu, C. Signal Transduction and Targeted Therapy 2022, 7 (1), 48. https://doi.org/10.1038/s41392-022-00904-4.
CrossRef - Aparna, E.; Rai, K. M. L.; Sureshbabu, M.; Jagadish, R. L.; Gaonkar, S. L.; Byrappa, K. Journal of Materials Science 2006, 41 (5), 1391–1393. https://doi.org/10.1007/s10853-006-7412-z.
CrossRef - Kerru N.; Gummidi, L.; Bhaskaruni, S.V.H.S.; Maddila S. N.; Jonnalagadda, S. B. Res Chem Intermed 2020, 46 (6), 3067–3083. https://doi.org/10.1007/s11164-020-04135-6.
CrossRef - Nguyen, T. B.; Ludmila Ermolenko; Al-Mourabit, Organic Letters 2012, 14 (16), 4274–4277. https://doi.org/10.1021/ol3020368. CrossRef
- Bakavoli, M.; Beyzaie, H.; Rahimizadeh, M.; Eshghi, H.; Takjoo, R. Molecules 2009, 14 (12), 4849–4857. https://doi.org/10.3390/molecules14114849.
CrossRef - Sharma, S.; Singh, D.; Kumar, S.; None Vaishali; Rahul Jamra; Naveen Banyal; None Deepika; Malakar, C. C.; Singh, V. Beilstein Journal of Organic Chemistry 2023, 19, 231–244. https://doi.org/10.3762/bjoc.19.22.
CrossRef - Kale, A. D.; Dalal, D. S. Catalyst ChemistrySelect 2022, 7 (48). https://doi.org/10.1002/slct.202203497.
CrossRef - Tayade, Y. A.; Jangale, A. D.; Dalal, D. S. ChemistrySelect 2018, 3 (31), 8895–8900. https://doi.org/10.1002/slct.201801553.
CrossRef - Fazylov, S. D.; Nurkenov, O. A.; Akhmetkarimova, Zh. S.; Zhienbaeva, D. R. Russian Journal of General Chemistry 2012, 82 (4), 781–782. https://doi.org/10.1134/s1070363212040317.
CrossRef - Jalil Noei; Khosropour, A. R. Tetrahedron Letters 2008, 49 (49), 6969–6971. https://doi.org/10.1016/j.tetlet.2008.09.084.
CrossRef - Pathak, U.; Pandey, L.; Mathur, S.; M. Suryanarayana. Synthesis 2011, 44 (03), 377–379. https://doi.org/10.1055/s-0031-1289994. CrossRef
- Mitra, B.; Gyan Chandra Pariyar; Ghosh, P. RSC Advances 2021, 11 (3), 1271–1281. https://doi.org/10.1039/d0ra09562a.
CrossRef
Accepted on: 02 Sep 2025
Second Review by: Dr. J S Yadav
Final Approval by: Dr. Ayssar Nahle

















