Synthesis and Benchmarking of CH₃NH₃PbBr₃ Perovskite Guided by the BR3 Framework for Green LED Applications
1Department of Physics, , Sudan University of Science and Technology, Khartoum, Sudan,
2Department of Engineering and Industrial Laser Application, Institute of Laser, Sudan 3University of Science and Technology, Khartoum, Sudan,
3University of Science and Technology, Khartoum, Sudan4University of Technology and Applied Science- Rustaq, Oman.
54Sudanese Chemical Society, Sudan.
Corresponding Author E-mail: sahlyasin@hotmail.com
Download this article as:
ABSTRACT:Methylammonium lead bromide (CH₃NH₃PbBr₃) perovskite was synthesized via a solution-based route and systematically characterized to establish structure–morphology–property relationships. X-ray diffraction (XRD) confirmed a highly crystalline cubic phase, Fourier-transform infrared spectroscopy (FTIR) verified characteristic organic and inorganic vibrational modes, and scanning electron microscopy (SEM) revealed uniform, well-faceted grains, indicative of controlled crystal growth. By implementing the BR3 framework (Benchmarking, Refinement, Replication, Reporting), this study provides a reproducible, fully documented synthesis pathway that addresses the typical lack of rigorous structural and morphological benchmarking in MAPbBr₃ research. The resulting high-quality crystals demonstrate suitability for green-emitting optoelectronic devices, particularly LEDs, and establish a validated foundation for future device-focused optimization.
KEYWORDS:Crystallite size; Methylammonium lead bromide (MAPbBr₃); Optoelectronic applications; Perovskite materials; Solution synthesis; Structural characterization
Introduction
Hybrid organic–inorganic halide perovskites, with the general formula CH₃NH₃PbX₃ (X = I, Br, Cl), have emerged as a transformative class of semiconductors in solid-state physics and materials science. Their prominence arises from a highly adaptable crystal structure in which the organic cation (CH₃NH₃⁺) is incorporated into an inorganic PbX₆ octahedral framework, combining the chemical versatility of organic molecules with the excellent charge-transport properties of inorganic semiconductors.1
A defining feature of these materials is their tunable optoelectronic behavior. The bandgap of CH₃NH₃PbX₃ perovskites can be precisely tailored by varying the halide composition, typically spanning ~1.6 eV for X = I to ~2.3 eV for X = Br.2 This ability to modulate absorption and emission spectra makes them highly attractive for a wide range of applications, including high-efficiency solar cells, bright light-emitting diodes (LEDs), and low-threshold lasers.3,4 In addition, these perovskites exhibit strong optical absorption, long charge-carrier lifetimes, high mobility, and high photoluminescence quantum yields, properties that are critical for high-performance optoelectronic devices.3,5
Despite their remarkable potential, translating laboratory-scale advances into reproducible, high-quality devices remains challenging. Variations in synthesis protocols can lead to inconsistencies in crystallinity, morphology, and optoelectronic performance, impeding reliable device fabrication [6]. Most studies focus on optical or device properties, while synthesis and structural–morphological benchmarking are treated as routine rather than systematically optimized. In particular, there is limited work that explicitly targets MAPbBr₃ as a green-light emitter, documents each synthesis step, correlates XRD, SEM, and FTIR results, and frames the material as a reproducible reference for future device studies. To address this gap, the present study applies the BR3 framework, Benchmarking, Refinement, Replication, and Reporting, to synthesize and comprehensively characterize CH₃NH₃PbBr₃. This structured methodology enables reproducible production of high-crystallinity, phase-pure, morphology-controlled MAPbBr₃, explicitly positioned as a validated starting material for green perovskite LEDs and other optoelectronic applications.
Experimental
Materials and Synthesis
The following chemicals were used for synthesis without further purification: lead(II) acetate (Pb(C₂H₃O₂)₂, 99%), lead(II) nitrate (Pb(NO₃)₂, 99%), potassium bromide (KBr, 99.5%), potassium iodide (KI, 99%), methylamine solution (CH₃NH₂, 40% w/w in water), and hydrobromic acid (HBr, 48% w/w aqueous solution). The solvents N, N-dimethylformamide (DMF, analytical grade) and diethyl ether (analytical grade) were also used. Lead (II) bromide (PbBr₂, 99%) was used directly when commercially available; otherwise, it was synthesized in situ from Pb(C₂H₃O₂)₂ and KBr. The key physical and chemical properties of these reagents are summarized in Table 1. Other instruments and apparatus included 500-mL beakers, graduated cylinders, funnels, 250-mL conical flasks, an analytical balance, a water bath, a magnetic stirrer, a heating mantle, and filter paper.
Safety Note: All experiments involving lead compounds, hydrobromic acid, and methylamine were performed in a well-ventilated fume hood using standard personal protective equipment (lab coat, gloves, and safety goggles). Lead-containing waste and acidic residues were collected and disposed of in accordance with institutional hazardous waste management protocols to minimize health and environmental risks.
Table 1: Properties of the chemicals used for the synthesis of CH₃NH₃PbBr₃ perovskite.
| Compound | Formula | Molar mass (g·mol⁻¹) | Density (g·cm⁻³) | Melting point (°C) | Purity / Concentration |
| Methylamine (aq.) | CH₃NH₂ | 31.05 | 0.656 | −93 | 40% (w/w) aqueous solution |
| Hydrobromic acid | HBr | 80.91 | 1.49 | −11 | 48% (w/w) aqueous solution |
| Potassium bromide | KBr | 119.01 | 2.75 | 734 | 99.5% (purity) |
| Potassium iodide | KI | 166.00 | 3.12 | 681 | 99% (purity) |
| Lead(II) acetate | Pb(C₂H₃O₂)₂ | 325.29 | 3.25 | 280 | 99% (purity) |
| Lead(II) nitrate | Pb(NO₃)₂ | 331.20 | 4.53 | 470 | 99% (purity) |
| Lead(II) bromide *¹ | PbBr₂ | 367.01 | 6.66 | 373 | 99% (purity) |
| N,N-Dimethylformamide | C₃H₇NO | 73.09 | 0.944 | −61 | Analytical grade |
| Diethyl ether | C₄H₁₀O | 74.12 | 0.713 | −116 | Analytical grade |
Method
All synthesis steps described below were conducted under the safety precautions to ensure safe handling of hazardous reagents. The procedures included the synthesis of methylammonium bromide (CH₃NH₃Br), lead (II) bromide, and the final organometallic halide perovskite CH₃NH₃PbBr₃. The characterization methods employed for product analysis are described in following:
Synthesis of Lead (II) Bromide (PbBr₂)
Lead(II) bromide was obtained via a precipitation reaction between lead(II) acetate [Pb(C₂H₃O₂)₂] and potassium bromide (KBr). In a typical procedure, an aqueous solution of Pb(C₂H₃O₂)₂ (0.1 mol, 32.5 g dissolved in 100 mL of water) was added dropwise to an aqueous solution containing a two-fold molar excess of KBr (0.2 mol) relative to Pb²⁺ to ensure complete precipitation of PbBr₂. A fine white precipitate of PbBr₂ formed immediately according to the double place reaction:
![]()
The precipitate was collected by vacuum filtration, washed thoroughly with deionized water to remove residual potassium acetate, and dried at 60 °C for 24 h. Alternatively, high-purity commercial PbBr₂ (≥99%) was used directly.
Synthesis of Methylammonium Bromide (CH₃NH₃Br)
Methylammonium bromide was prepared by reacting methylamine solution (CH₃NH₂, 40% w/w, 0.3 mol, 38 mL) with an equimolar amount of hydrobromic acid (HBr, 48% w/w, 0.3 mol, ~34 mL) under ice-bath conditions to control the exothermic reaction. The mixture was stirred for two hours to yield a clear solution of CH₃NH₃Br. The solvent was evaporated to dryness at 60 °C, and the resulting crystalline solid was recrystallized from cold diethyl ether, filtered, and dried at room temperature for 120 h. The reaction is represented as:
![]()
Synthesis of Organometallic Halide Perovskite Sensitizer (CH₃NH₃PbBr₃)
The organometallic halide perovskite CH₃NH₃PbBr₃ was synthesized by dissolving equimolar amounts of CH₃NH₃Br and PbBr₂ (1:1 molar ratio, total ~2.84 g) in 20 mL of anhydrous DMF under continuous stirring at room temperature for two hours until a clear orange solution was obtained. Slow solvent evaporation yielded bright orange crystals of CH₃NH₃PbBr₃. The reaction can be expressed as:
![]()
Characterization
The synthesized CH₃NH₃PbBr₃ perovskite samples were characterized using complementary structural, morphological, and spectroscopic techniques. The crystal structure was examined using X-ray diffraction (XRD) to confirm phase formation, crystallinity, and lattice features. Surface morphology and grain texture were analyzed by scanning electron microscopy (SEM) at an accelerating voltage of 10.0 kV, with a view field of 136 µm, a working distance of 18.18 mm, and a magnification of approximately 1,020× (1.02 k×), using a secondary electron (SE) detector. Fourier-transform infrared (FTIR) spectroscopy was employed to identify characteristic vibrational modes and verify the incorporation of the methylammonium cation within the Pb–Br perovskite framework.
Implementation of the BR3 Framework
The synthesis process was structured and optimized according to the BR3 framework to ensure reproducibility and high material quality.
Benchmarking: Existing literature on precursor ratios, solvent selection, and crystallization conditions for MAPbBr₃ was comprehensively surveyed to establish a baseline synthesis protocol.
Refinement: Multiple repeated trial syntheses were performed, systematically adjusting parameters including solvent evaporation rate, precursor concentration, and stirring duration. This process revealed that a controlled, slower solvent-evaporation rate was the most critical factor for achieving enhanced crystallinity and a larger, more uniform grain size.
Replication: The final optimized synthesis protocol, defined through the refinement stage, was reproduced in triplicate. The resulting samples showed consistent structural and morphological properties, with less than 3% variation in XRD peak intensity and SEM-observed grain morphology, confirming the robustness of the method.
Reporting: All synthesis steps, characterization procedures, and optimized parameters have been thoroughly documented in this work to promote reproducibility and facilitate further research.
This structured BR3 approach was instrumental in consistently and reliably producing phase-pure, well-crystallized MAPbBr₃ material, as characterized in this study.
Results and discussion
X-Ray Diffraction XRD Structural Analysis:
X-ray diffraction (XRD) was used to verify the crystal structure and phase purity of the synthesized CH₃NH₃PbBr₃ perovskite. The diffraction pattern in (Fig. 1) shows several sharp peaks within the 2θ range of 10°–18°, confirming the formation of a highly crystalline phase. The main reflections were recorded at 11.9°, 14.9°, and 17.0° match the expected fingerprint of cubic CH₃NH₃PbBr₃. The intense peak at 14.9° suggests a preferred orientation and high structural order.
The major peaks were indexed to the cubic perovskite structure and assigned to the (100), (110), and (200) planes, consistent with standard MAPbBr₃ reference patterns. These results confirm the successful synthesis of a phase-pure and well-crystallized CH₃NH₃PbBr₃ perovskite.
The XRD pattern of the synthesized CH₃NH₃PbBr₃ perovskite (Fig. 1) exhibits a dominant, sharp diffraction peak at 14.9°, corresponding to the (100) crystallographic plane of the cubic perovskite phase with Pm3m space group symmetry. This reflection is recognized in the literature as the primary fingerprint of cubic-phase MAPbBr₃ perovskite structure [13,14]. The sharpness and high intensity of the peak confirm excellent crystallinity and indicate high phase purity, as impurity or byproduct phases would introduce additional peaks of comparable intensity. Additional weaker reflections were observed near 11.9° and 12.6°, which may originate from low-index planes, residual precursor materials, or minor secondary phases; however, their negligible intensity relative to the 14.9° peak demonstrates that they do not compromise the dominant cubic phase. These observations are consistent with previously reported XRD patterns for CH₃NH₃PbBr₃ perovskite, in which the cubic (100) reflection near 14.9° is consistently identified as the defining structural signature. While minor variations in peak intensity and low-angle features are known to arise from synthesis methods, sample thickness, or trace impurities, the persistence of the strong 14.9° peak in this work confirms the successful synthesis of highly crystalline and phase-pure cubic CH₃NH₃PbBr₃.
 |
Figure 1: XRD diffraction pattern of CH₃NH₃PbBr₃ perovskite showing characteristic peaks indexed to the cubic phase. Major reflections are assigned to the (100), (110), and (200) planes. The sharp and well-defined peaks indicate high crystallinity and phase purity of the synthesized material. |
Crystallite Size Calculation
The crystallite size of the synthesized perovskite was calculated using the Scherrer equation:
![]()
Where D demonstrated the crystallite size, K is the shape factor (assumed to be 0.9), λ is the X-ray wavelength (typically Cu Kα = 0.15406 nm), β is the full width at half maximum (FWHM, in radians), and θ is the Bragg angle. The calculated values are summarized in Table 2.
![]()
The average crystallite size was found 314.07 nm, consistent with nanoscale crystalline domains characteristic of perovskite materials.
Table 2: XRD peak parameters and crystallite
| 2θ (°) | FWHM (°) | Crystallite Size (nm) |
| 11.944 | 0.34382 | 232.32 |
| 12.615 | 0.14548 | 549.40 |
| 14.915 | 0.26277 | 304.91 |
| 16.462 | 0.04676 | 171.66 |
The calculated crystallite sizes are presented in Table 2. The obtained average crystallite size (~314 nm) aligns well with the nanocrystalline regime characteristic of perovskite materials.
The obtained average crystallite size (~314 nm) aligns well with previous studies on solution processed MAPbBr₃ perovskites, where crystallite sizes typically range from 200 to 400 nm, depending on the synthesis route and processing conditions 6,15,16. Such values indicate well-developed nanocrystalline domains with excellent long-range order. Larger crystallite sizes are particularly beneficial for optoelectronic applications, as they reduce grain boundary density, minimize non-radiative recombination pathways, and enhance charge carrier transport. This is consistent with earlier findings that high-crystallinity MAPbBr₃ materials demonstrate superior photophysical properties and device performance in solar cells and light-emitting diodes.17-19
In short, the Scherrer analysis confirms that the synthesized CH₃NH₃PbBr₃ perovskite possesses nanoscale crystallite domains of high quality, validating its structural suitability for advanced perovskite-based optoelectronic applications.
SEM Morphology and Microstructure
Scanning electron microscopy (SEM) images (Fig. 2) confirmed the cubic grains characteristic of CH₃NH₃PbBr₃ crystals. The grains display clean facets and a relatively uniform size distribution, consistent with controlled nucleation and growth during synthesis. Clear embedded scale bars (50 µm and 20 µm) are included directly in the images to ensure accurate interpretation of grain size and morphology.
 |
Figure 2: SEM micrographs of CH₃NH₃PbBr₃ crystals at different magnifications. Clear embedded scale bars ((a) 20 µm, and (b) 50 µm respectively) are included within each image. The micrographs reveal well-defined cubic grains with uniform morphology, indicative of high-quality crystallization. |
The SEM micrograph of the synthesized CH₃NH₃PbBr₃ perovskite reveals well-defined, blocky microcrystals with sharp, faceted edges, a feature typical of high-quality methylammonium lead bromide prepared via solution-based methods. Numerous cuboid and plate-like crystals with distinct, flat faces are visible, indicating preferential growth along specific crystallographic directions, most likely the (001) plane, which is widely reported as a dominant growth facet for MAPbBr₃ in literature15,20. The relative uniformity in crystal shape suggests a controlled layered crystallization mechanism, in which cubes and plates are progressively formed by sequential layer deposition, consistent with previously observed growth pathways for hybrid perovskites.21-23
The crystals appear interconnected, forming larger agglomerates while still maintaining their individual faceted structures, a hallmark of well-crystallized perovskite domains. The sharply defined geometries and the absence of rounded or amorphous particles confirm the high crystallinity and phase purity of the product, in agreement with the XRD results, which showed strong, single-phase reflections. Furthermore, the microcrystal surfaces appear relatively smooth, with minimal observable surface defects. Such high-quality surface morphology is desirable for optoelectronic applications, as it reduces the density of surface recombination centers that can otherwise limit charge-carrier lifetimes.
From the scale bar (20 μm), it is evident that some block crystals reach several micrometers in size. This observation is consistent with the XRD-derived average crystallite size of hundreds of nanometers, suggesting that the larger grains observed in SEM may result from the coalescence of smaller crystallites. Large grain domains are beneficial for device performance, as they reduce grain boundary density, enhance charge transport, and suppress trap-mediated recombination. The plate- and cube-like morphologies observed are also linked to uniform optical properties and reduced ion migration, which contribute to improved long-term stability of perovskite-based devices.24,25
Overall, the SEM analysis confirms that the synthesized CH₃NH₃PbBr₃ perovskite demonstrates excellent crystalline quality, with well-faceted cubes and plates characteristic of optimal solution-processed growth. These morphological features are highly advantageous for photovoltaic and light-emitting applications, where grain size, crystallinity, and surface smoothness play critical roles in determining device efficiency and durability.
Fourier-Transform Infrared Spectroscopy:
The FTIR spectrum of the synthesized CH₃NH₃PbBr₃ perovskite (Fig. 3) shows the characteristic vibrational modes of both the methylammonium cation and the Pb–Br inorganic lattice. A distinct band at 662.85 cm⁻¹ is assigned to Pb–Br bending vibrations coupled with rocking motions of the MA⁺ cation,7, confirming the formation of the coordinated Pb–Br framework. The mid-IR region displays several key features, including the MA⁺ rocking mode at 929.08 cm⁻¹, the C–N stretching vibration at 1018.62 cm⁻¹, and the strong symmetric NH₃ bending mode at 1412.47 cm⁻¹ with contributions from CH₃ deformation.8,9 A sharper feature just below 1600 cm⁻¹ corresponds to N–H bending, indicating the protonated state of the ammonium group and its hydrogen-bonding interaction with Br⁻ ions.9,10 In the high-frequency region, the broad absorption centered near 3147.88 cm⁻¹ is attributed to N–H stretching of the –NH₃⁺ group, with the broadening reflecting the strong hydrogen-bonding network within the perovskite lattice.11,12 A surrounding Br⁻ ions, an interaction essential for stabilizing the hybrid perovskite lattice. The C–H stretching modes of the CH₃ group appear as well-defined peaks between 2800 and 3000 cm⁻¹, confirming the incorporation of the organic cation. Vibrations observed between 1400 and 1600 cm⁻¹ correspond to N–H bending modes, while a band near ~1000 cm⁻¹ is assigned to C–N stretching, further verifying the structural integrity of the CH₃NH₃⁺ ion. The absence of additional unassigned peaks suggests that the synthesized CH₃NH₃PbBr₃ is phase-pure and free from residual precursors or degradation products.
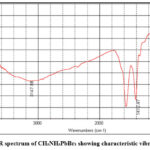 |
Figure 3: FTIR spectrum of CH₃NH₃PbBr₃ showing characteristic vibrational bands Click here to View table |
As expected for lead–halide perovskites, Pb–Br lattice vibrations occur below ~600 cm⁻¹ and appear as weak low-wavenumber features due to the limited sensitivity of standard mid-IR detectors in this region. Importantly, the absence of strong O–H stretching near 3200–3600 cm⁻¹ indicates minimal moisture incorporation, a key factor in preventing degradation of hybrid halide perovskites. No additional bands attributable to precursor salts or solvent residues were detected, supporting the high purity of the synthesized material.
Furthermore, the FTIR spectrum provides clear evidence of intact organic–inorganic bonding, strong hydrogen-bonding interactions within the perovskite cage, and the absence of impurity signatures, all of which enhance the phase purity and structural stability inferred from XRD and SEM analyses.
The synthesized CH₃NH₃PbBr₃ perovskite exhibited high structural integrity and phase purity, as confirmed by XRD analysis, which revealed the expected cubic lattice without secondary phases. FTIR spectra confirmed the presence of characteristic organic–inorganic vibrational modes, while SEM imaging demonstrated well-defined grain morphology, indicative of uniform crystallization. Such structural coherence is crucial since morphological defects and secondary phases are known to compromise carrier transport and optical efficiency in perovskite-based devices.
The results establish a clear structure–property–application relationship: the synthesis method produces high-quality CH₃NH₃PbBr₃ with structural features known to support favorable optical behavior, highlighting its potential for next-generation optoelectronic devices. Further optimization of film thickness, interface engineering, and stability will be key to fully realizing this material’s performance in solar cells, light-emitting diodes, and perovskite-based lasers.
Applications Supported by the Structural and Morphological Results
Perovskite-Based Green Light-Emitting Diodes (LEDs)
Among the potential applications of CH₃NH₃PbBr₃, the experimental results presented in this study most strongly support its use in green perovskite light-emitting diodes (LEDs). Several key findings align directly with the material requirements for high-performance emissive devices.
Alignment with Results
XRD and Crystallite Size
The material exhibits high crystallinity with large nanoscale crystallites (average ≈ 314 nm). Such large grains reduce grain boundary density, suppress non-radiative recombination, and improve radiative efficiency, all critical factors for bright, stable LED emission.
SEM Morphology
The SEM micrographs display well-defined, faceted microcrystals with a relatively uniform size distribution. This morphology facilitates smooth charge transport and minimizes leakage currents, supporting uniform and efficient electroluminescence.
FTIR Spectroscopy
FTIR analysis confirms the structural integrity of both the methylammonium cation and the Pb–Br framework. The absence of significant O–H bands indicates minimal moisture exposure, which is essential for operational stability in LED structures.
A Reliable Platform for Optoelectronic Device Prototyping and Fundamental Studies
Beyond specific device applications, the reproducibility and phase purity achieved in this synthesis make the material a valuable platform for exploring broader optoelectronic phenomena.
Alignment with Results
Integrated Structure–Property Relationship
The study demonstrates a clear link between synthesis parameters, crystallinity, and microstructure, confirming that the BR3-guided synthesis approach yields structurally reliable CH₃NH₃PbBr₃.
Reproducibility Across Techniques
Consistent results from XRD, SEM, and FTIR confirm phase purity and stable organic–inorganic bonding. Such reproducibility is advantageous for:
evaluating new device architectures,
studying charge transport and recombination mechanisms, and
benchmarking emerging perovskite compositions against a well-characterized reference material.
Limited Direct Support for Solar Cell Applications
Although CH₃NH₃PbBr₃ has recognized relevance in photovoltaic research, the present work does not include key photovoltaic characterization data, such as optical absorption spectra, optical bandgap determination, or electrical transport measurements, required to support solar-cell-oriented conclusions fully. Therefore, the applicability of solar cells cannot be strongly justified based solely on the reported XRD, SEM, and FTIR results.
Recommended Next Step: Development of a Green CH₃NH₃PbBr₃ LED Prototype
The structural and morphological quality demonstrated in this study provides a strong foundation for fabricating a green perovskite LED prototype. A simple device architecture may be adopted, such as:
ITO / Hole Transport Layer / CH₃NH₃PbBr₃ Emissive Layer / Electron Transport Layer / Metal Cathode
Future work should focus on
Measuring the electroluminescence spectrum to verify green emission,
Determining luminance and external quantum efficiency (EQE), and
Evaluating operational stability under electrical bias.
Completing these steps would directly validate the practical viability of the synthesized CH₃NH₃PbBr₃ for LED applications.
The study demonstrates that the BR3 framework represents a methodological novelty by shifting MAPbBr₃ research from empirical synthesis toward a structured, reproducible, and benchmark-driven approach. Scientifically, the work goes beyond routine characterization by critically correlating controlled synthesis refinement with crystallinity, grain morphology, and organic–inorganic lattice integrity. The consistency across XRD, SEM, and FTIR results validates the framework’s ability to reduce variability and establish MAPbBr₃ as a reliable reference material rather than a one-off synthesis outcome. This depth of analysis justifies the manuscript’s contribution by positioning BR3 as a transferable strategy for improving rigor and reproducibility in perovskite materials research.
Conclusion
CH₃NH₃PbBr₃ perovskite was successfully synthesized via a solution-based route and systematically characterized by XRD, SEM, and FTIR. XRD confirmed a highly crystalline cubic phase, SEM revealed uniform, well-faceted grains, and FTIR verified characteristic organic and inorganic vibrational modes, demonstrating structural integrity and phase purity. These results establish a reproducible, high-quality MAPbBr₃ material suitable for green-emitting optoelectronic devices, particularly LEDs. Future work should focus on device fabrication, performance optimization, and stability studies to fully exploit the material’s potential in practical applications.
Acknowledgment
The authors gratefully acknowledge the support and guidance received during the work. We extend our sincere appreciation to The Sudanese Forensic Laboratory for providing laboratory facilities and technical assistance.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Author Contributions
Zeinab Abdallah conceptualization and writing original draft preparation; Nodar Osman; formal analysis. Ali A. S; draft preparation Sahl Yasin, and Mohamed Alzubair, review, and editing.
References
- Zhao Y, Zhu K. Organic–inorganic hybrid lead halide perovskites for optoelectronic and electronic applications. Chemical Society Reviews. 2016;45(3):655-89. DOI https://doi.org/10.1039/C4CS00458B
CrossRef - Sutter-Fella CM, Li Y, Cefarin N, Buckley A, Ngo QP, Javey A, Sharp ID, Toma FM. Low Pressure Vapor-assisted Solution Process for Tunable Band Gap Pinhole-free Methylammonium Lead Halide Perovskite Films. Journal of Visualized Experiments: Jove. 2017 Sep 8(127):55404.htpp://doi:2010.3791/55404
CrossRef - Tyagi D, Laxmi V, Basu N, Reddy L, Tian Y, Ouyang Z, Nayak PK. Recent advances in two-dimensional perovskite materials for light-emitting diodes. Discover Nano. 2024 Jul 2;19(1):109. https://doi.org/10.1186/s11671-024-04044-2
CrossRef - Xing G, Mathews N, Lim SS, Yantara N, Liu X, Sabba D, Grätzel M, Mhaisalkar S, Sum TC. Low-temperature solution-processed wavelength-tunable perovskites for lasing. Nature materials. 2014 May;13(5):476-80. https://doi.org/10.1038/nmat3911
CrossRef - Aftab S, Kabir F, Mukhtar M, Hussain I, Nazir G, Aslam M, Hegazy HH, Yewale MA. Perovskite quantum wires: A review of their exceptional optoelectronic properties and diverse applications in revolutionary technologies. Nano Energy. 2024 Oct 1;129:109995.https://doi.org/10.1016/j.nanoen.2024.109995
CrossRef - Seyisi T, Fouda-Mbanga BG, Mnyango JI, Nthwane YB, Nyoni B, Mhlanga S, Hlangothi SP, Tywabi-Ngeva Z. Major challenges for commercialization of perovskite solar cells: A critical review. Energy Reports. 2025 Jun 1;13:1400-15.https://doi.org/10.1016/j.egyr.2025.01.019
CrossRef - Zhang F, Karimata I, Wang HW, Tachikawa T, Tominaga K, Hayashi M, Sasaki T. Terahertz spectroscopic measurements and solid-state density functional calculations on CH3NH3PbBr3 perovskites: Short-range order of methylammonium. The Journal of Physical Chemistry C. 2021 Dec 29;126(1):339-48. https://doi.org/10.1021/acs.jpcc.1c06988
CrossRef - Panneerselvam V, Salammal ST, Chinnakutti KK, Manidurai P. Vibrational modes, chemical states and thermal stability of mechanochemically synthesized methylammonium lead iodide (CH3NH3PbI3) perovskites. Materials Letters. 2019 Apr 15;241:140-3.https://doi.org/10.1016/j.matlet.2019.01.069
CrossRef - Pérez-Osorio MA, Lin Q, Phillips RT, Milot RL, Herz LM, Johnston MB, Giustino F. Raman Spectrum of the Organic–Inorganic Halide Perovskite CH₃NH₃PbI₃ from First Principles and High-Resolution Low-Temperature Raman Measurements.https://doi.org/10.1021/acs.jpcc.8b04669
CrossRef - Feliciano GT. Vibrational properties of the organic-inorganic halide perovskite CH3NH3PbI3 from theory and experiment: factor group analysis, first-principles calculations, and low-temperature infrared spectra [dissertation]. São Paulo: Universidade de São Paulo; 2015. doi: 10.11606/D.43.2015.tde-20102015-145926.
- Hansen PE, Vakili M, Kamounah FS, Spanget-Larsen J. NH stretching frequencies of intramolecularly hydrogen-bonded systems: an experimental and theoretical study. Molecules. 2021 Dec 17;26(24):7651.Anikeeva, V.E., Boldyrev, K.N., Semenova, O.I., Sukhikh, T.S. and Popova, M.N., 2023. Broad-range high-resolution optical spectroscopy of CH3NH3PbBr3 hybrid perovskite single crystals: Optical phonons, absorption edge, phase transitions. Optical Materials: X, 20, p.100259. https://doi.org/10.3390/molecules26247651
CrossRef - Hussain S, Khan MI, Subhani WS, Mustafa GM, Saleem M, Abubshait SA, Abubshait HA, Saleh DI, Mahmoud SF. Decorating wide band gap CH3NH3PbBr3 perovskite with 4AMP for highly efficient and enhanced open circuit voltage perovskite solar cells. Solar Energy. 2021 Dec 1;230:501-8.Gidey, A.T., Haruta, Y., Herman, A.P., Grodzicki, M., Melnychenko, A.M., Majchrzak, D., Mahato, S., Rogowicz, E., Syperek, M., Kudrawiec, R. and Saidaminov, M.I., 2023. Surface Engineering of Methylammonium Lead Bromide Perovskite Crystals for Enhanced X-ray Detection. The Journal of Physical Chemistry Letters, 14(40), pp.9136-9144.https://doi.org/10.1016/j.solener.2021.10.01
CrossRef - Heindl MW, Lichtenegger MF, Kodalle T, Liu S, Solhtalab N, Zerhoch J, Shcherbakov A, Kivala M, Sutter‐Fella CM, Urban AS, Deschler F. Ligand‐Induced Crystallization Control in MAPbBr3 Hybrid Perovskites for High Quality Nanostructured Films. Advanced Optical Materials. 2025 Feb;13(6):2402441. https://doi.org/10.1002/adom.202402441
CrossRef - Zhao T, Guo Y, Yang X, Xiong Q, Gao P, Meng L, Xiong Z. Pure α-phase (FAPbI3) 0.83 (MAPbBr3) 0.17 powder for efficient perovskite solar cells with enhancing reproducibility. Materials Today Communications. 2024 Dec 1;41:110398. https://doi.org/10.1016/j.mtcomm.2024.110398
CrossRef - slam MJ, Islam MH. Tuning emission and bandgap dynamics of MAPbBr₃ single crystals through halide exchange with methyl iodide. Chemical Physics Impact. 2025 Jun 1;10:100807.https://doi.org/10.1016/j.chphi.2024.100807
CrossRef - Mahato S, Makowski M, Bose S, Kowal D, Kuddus Sheikh MA, Braueninger-Wemer P, Witkowski ME, Ray SK, Drozdowski W, Birowosuto MD. Improvement of light output of MAPbBr3 single crystal for ultrafast and bright cryogenic scintillator. The Journal of Physical Chemistry Letters. 2024 Mar 28;15(14):3713-20.https://doi.org/10.1021/acs.jpclett.4c00379
CrossRef - Vila M, Bartolomé J, El Astal-Quirós A, Martínez-Pastor JP, Suarez I, de Andrés A, Coya C. Morphology engineering of MAPbBr3 thin films for enhanced lighting applications. APL Materials. 2025 Jul 1;13(7). https://doi.org/10.1063/5.0263289
CrossRef - Cho Y, Jung HR, Jo W. Halide perovskite single crystals: growth, characterization, and stability for optoelectronic applications. Nanoscale. 2022;14(26):9248-77. https://DOI: 10.1039/D2NR00513A
CrossRef - Bonadio A, Souza JA. Hybrid MAPbI 3 Perovskite Growth Mechanism from Irregular Particles to Cuboid and Hopper-Type Morphologies. Journal of the Brazilian Chemical Society. 2022 Oct 24;33:1273-80. https://doi.org/10.21577/0103-5053.20220057
CrossRef - Gao Q, Fang H, Xiang D, Chen Y, Tanaka H, Tan P. Impact of impurities on crystal growth. Nature Physics. 2025 Apr 15:1-9. https://doi.org/10.1038/s41567-025-02870-4
CrossRef - O’Brien MN, Jones MR, Mirkin CA. The nature and implications of uniformity in the hierarchical organization of nanomaterials. Proceedings of the National Academy of Sciences. 2016 Oct 18;113(42):11717-25. https://doi.org/10.1073/pnas.1605289113
CrossRef - Wang S, Frisch S, Zhang H, Yildiz O, Mandal M, Ugur N, Jeong B, Ramanan C, Andrienko D, Wang HI, Bonn M. Grain engineering for improved charge carrier transport in two-dimensional lead-free perovskite field-effect transistors. Materials Horizons. 2022;9(10):2633-43. https://DOI: 10.1039/D2MH00632D
CrossRef - Yang Z, Qin A, Qin H, Li Z, Xiang Y, Liu H, Qiu J, Wang X. Ion migration and dark current suppression in quasi-2D perovskite-based X-ray detectors. RSC advances. 2024;14(27):19124-33.https://DOI: 10.1039/D4RA02606C
CrossRef
Second Review by: Dr. Shreya Shanyal
Final Approval by: Dr. Ioana Stanciu

















