Sustainable Production of ZnO Nanoparticles from Trianthema Portulacastrum and Their Efficacy against Pathogenic Microbes
1Department of Chemistry, Nehru Memorial College (Autonomous) /Affiliated to Bharathidasan University, Puthanampatti, Tiruchirappalli, Tamilnadu, India.
2Department of Chemistry, School of Engineering and Technology, Dhanalakshmi Srinivasan University, Samayapuram, Tiruchirappalli, Tamilnadu, India.
3Department of Chemistry, Thanthai Periyar Government Arts and Science College (Autonomous) / Affiliated to Bharathidasan University, Tiruchirappalli, Tamilnadu, India.
Corresponding Author E-mail:ilavenilkk.set@dsuniversity.ac.in
Download this article as:
ABSTRACT:This research investigates the eco-friendly manufacture of zinc oxide nanoparticles utilising the leaf extract of Trianthema portulacastrum, a medicinal plant recognized for its bioactive phytochemicals. The synthesis process avoids harmful ingredients, conforming to sustainable and environmentally responsible procedures. Thorough structural and chemical evaluations utilising FTIR, UV-Vis spectroscopy, SEM, DLS, and XRD confirmed the successful production of stable ZnO nanoparticles, which have a distinct hexagonal wurtzite crystal structure and surfaces functionalized by plant-derived proteins. The UV-Vis spectrum exhibited a red-shifted absorption, signifying distinctive optical characteristics. The DLS data indicated moderate aggregation with a hydrodynamic diameter of approximately 97.8 nm. The antibacterial efficiency was assessed against various pathogenic strains, demonstrating substantial inhibitory zones, particularly surpassing traditional medications in effectiveness against Mycobacterium tuberculosis.
KEYWORDS:Antimicrobial activity; Green synthesis; Phytochemicals; Trianthema portulacastrum; UV-Visible spectroscopy; Zinc oxide nanoparticles
Introduction
Nanomaterials (NMs) derived from natural sources have emergent as a result of the growing need for environmentally friendly materials. NMs are utilised in a variety of sectors, including medicine, biosensors, and catalysts, due to their nanoscale size and significantly larger surface area. The conventional methods of synthesis frequently involve the use of hazardous chemicals and a significant amount of energy, which can be hazardous to both the environment and human health. Green synthesis, on the other hand, makes use of natural reagents such as plant extracts and microorganisms, and thus provides an option that is both friendly to the environment and economical. This method is said to be a promising strategy for the production of non-toxic metal oxide nanoparticles, and it is in accordance with the principles of green chemistry. Because of its one-of-a-kind characteristics and applicability in a wide range of sectors, including biomedicine, electronics, and catalysis, zinc oxide nanoparticles are the subject of much research1. Their significant antibacterial and anti-inflammatory activities suggest potential as viable substitutes for traditional antibiotic therapies. Replacing traditional chemical methods with green synthesis approaches for metal oxide nanoparticles promotes sustainability and reduces environmental impact. The conventional method of chemical synthesis frequently includes the use of dangerous chemicals and toxic solvents2, 3, and it necessitates the application of high temperatures and pressures, which eventually results in environmental contamination and health dangers. Physical and chemical methods can synthesize nanoparticles with precise particle size and shape for specific purposes. Infrared irradiation, pulsed laser deposition, sol-gel processing, thermal evaporation4, microwave-assisted synthesis5, chemical vapor deposition6, electrochemical reactions, and sputtering7 are some of the techniques that are commonly used. The green synthesis method, on the other hand, makes use of biological resources such plant extracts, bacteria, fungi, and algae, all of which serve as natural reducing and stabilizing agents. This strategy functions in ambient conditions, so reducing the amount of energy that is consumed and doing away with the requirement for potentially hazardous compounds8. Therefore, green synthesis results in the production of nanoparticles that are non-toxic and biocompatible. This is in accordance with the principles of green chemistry and promotes safer uses in a variety of disciplines, including medicine and environmental cleanup.
The green synthesis of zinc oxide nanoparticles (ZnO-NPs) has been produced using plant extracts rich in phytochemicals, including flavonoids and phenolics, which serve as natural reducing and stabilising agents9. A cost-effective and environmentally responsible alternative to conventional procedures, this eco-friendly approach is in line with the principles of green chemistry. ZnO-NPs provide antibacterial effects by forming reactive oxygen species (ROS), releasing Zn2+ ions, and directly contacting microbial cell membranes10. Several different pathogens are effectively inhibited as a result of these actions, which affect the structures and functions of the cells. Due to this, green-synthesized ZnO-NPs have the potential to be utilized in a variety of applications, including the treatment of cancer11-12, antimicrobial therapy13, wound healing14, and environmental clean-up. Temperature, pH, and precursor concentration greatly affect zinc oxide (ZnO) nanostructure shape. These conditions can change ZnO’s physical and chemical properties. It has a high aspect ratio, big surface area, and considerable optical and electrical properties15.
ZnO NPs show significant antibacterial action against several Gram-positive and Gram-negative microorganisms. Staphlococcus aureus, Escherichia coli, Pseudomonas aeruginosa, Klebsiella pneumoniae, and Bacillus subtilis are vulnerable to ZnO NPs. The broad-spectrum antibacterial properties of these nanoparticles make them viable bacterial infection treatments16. Notably, their efficacy persists even in the absence of light, attributed to mechanisms such as reactive oxygen species generation, membrane disruption, and zinc ion release17. ZnO nanoparticles are a promising alternative to conventional antibiotics, particularly when it comes to combating drug-resistant bacteria, due to the qualities that they possess. Their physicochemical properties, on the other hand, might change depending on the conditions of the surrounding environment. Furthermore, in larger concentrations, they may present a hazard to living species and ecosystems. Nanoparticles of zinc oxide (ZnO) have been successfully synthesised using Trianthema portulacastrum leaf extract, which is environmentally friendly. The natural phytochemicals of the plant, such as flavonoids, phenolic compounds, terpenoids, and alkaloids, are utilised in this environmentally friendly approach. These phytochemicals fulfil the roles of reducing and stabilising agents during the synthesis process. In the realms of biological and environmental cleanup, the ZnO nanoparticles that emerge as a result show intriguing uses.
Sharunai plant
Aizoaceae member Trianthema portulacastrum is known as huge pigweed, black pigweed, desert horse purslane, or Olowonjeja. Although its origin is unknown, it is found in tropical and subtropical locations including Africa, India, Southeast Asia, West Asia18, southern China, and tropical America. Traditional Ayurvedic medicine uses T. portulacastrum for treatment. It is used in herbal cough, uteralgia, and inflammatory therapies. It is also valued as a green vegetable in the diets of economically disadvantaged populations in the Indian subcontinent19. The plant is known by various vernacular names in India, including Bishkhapra or Safed Bishkhapra in Hindi, Gaddicheraku or Kotthimeeraaku in Telugu, Sarunai Keerai or Thotta Keerai in Tamil, Attum Parippu in Malayalam, Elachikkirai or Garikai in Kannada, and Shukhatara in Marathi. Traditionally, the plant has been used for treating edema in the liver and spleen, uteralgia, and cough. It is considered lithotriptic for the kidney and bladder and is widely recognized in Indian traditional medicine as a diuretic. Studies have demonstrated its hepatoprotective activity, particularly against paracetamol and thioacetamide-induced intoxication in rats20. Terpenoids, phenolic chemicals, alkaloids, saponins, and flavonoids have been found in Trianthema portulacastrum phytochemical studies. Antioxidant, anti-inflammatory, analgesic, and anti-infective properties characterise these components. An increasing number of in vitro and in vivo research show that the plant can prevent or treat hepatotoxicity, nephrotoxicity, hyperglycemia, hyperlipidaemia, infectious illnesses, and cancer. Notable chemical components of the plant 21 include 15-hydroxymethyl-2,6,10,18,22,26,30-heptamethyl-14-methylene-17-hentriacontene (commonly known as trianthenol), ketoconazole, glibenclamide, 3-methoxy-4-hydroxybenzoic acid, N-nitrosodiethylamine, aflatoxin B1, miconazole, phenobarbital, bilirubin, and 3,7-dihydro-1,3,7-trimethyl-1H-purine-2,6-dione22. Trianthema portulacastrum is used to treat eye-related issues 23.
Methodology
Preparation of Trianthema portulacastrum leaf extract
Fresh leaves of Trianthema portulacastrum were collected and meticulously rinsed with distilled water to eliminate surface contaminants prior to preparing the dry leaf powder for aqueous extraction. The cleansed leaves were subsequently shade-dried in a well-ventilated environment to retain heat-sensitive phytochemicals. After thorough drying, the leaves were pulverised into a fine powder with a mechanical grinder and sifted to achieve consistent particle size. The resultant powder was preserved in sealed containers to avert moisture absorption and deterioration. During the extraction process, 30 grams of the produced leaf powder were subjected to boiling in 100 mL of distilled water for 1 hour to promote the release of bioactive components. Subsequent to boiling, the mixture was allowed to cool to ambient temperature and then filtered with Whatman No. 44 filter paper to isolate the solid residues from the liquid extract. The filtrate underwent additional purification via centrifugation, and the resultant clear supernatant was meticulously collected and preserved at 4 °C for subsequent analysis or experimental applications.
Nanoparticle Zinc Oxide Synthesis
The nanoparticles were synthesised using Trianthema portulacastrum leaf powder and 0.03 M zinc sulphate heptahydrate (ZnSO₄•7H₂O) as the zinc precursor in a green reflux method. The aqueous plant extract was made by boiling 10 g of air-dried leaf powder in 250 mL of distilled water at 80 °C for 1 hour with steady agitation. Zinc sulphate hepta hydrate was dissolved in 100 mL of distilled water with continuous stirring to create a 0.03 M zinc sulphate solution. Subsequently, 25 mL of the produced plant extract was incorporated into the zinc sulphate solution while maintaining continuous agitation. While stirring, 0.1 M sodium hydroxide (NaOH) solution was added dropwise to adjust the liquid’s pH to 10. After refluxing at 80-90 °C for 3 hours, the solution reduced zinc ions and synthesised ZnO nanoparticles, with phytochemicals from the plant extract acting as reducing and stabilising agents. After reflux, the mixture cooled to room temperature and was centrifuged at 10,000 rpm for 15 minutes to separate nanoparticles. To remove impurities, the pellet was cleaned with distilled water and ethanol, then dried in a 60 °C hot air oven for 12 hours.
 |
Figure 1: Biosynthesis of Trianthema portulacastrum – ZnONPs Click here to View Figure |
Chemical Profiling of Medicinal Plant
The phytochemical analysis of primary and secondary metabolites in medicinal plants utilises various traditional methodologies. The Fehling’s carbohydrate test was conducted by combining equal volumes of leaf extract with Fehling’s A and B reagents, followed by heating for 3-5 minutes until a crimson precipitate formed, signifying the presence of reducing sugars 24 . The iodine test for starch involved mixing 2 mL of Trianthema portulacastrum leaf extract with iodine and potassium iodide (Lugol’s solution), producing a blue hue that signifies the presence of starch. The Biuret test for proteins was conducted by combining 1 mL of leaf extract with 3% zinc sulphate and 10% sodium hydroxide, yielding a violet or red colouration, indicative of the presence of proteins or peptides. Salkowski’s test for steroids was employed to identify secondary metabolites, wherein Trianthema portulacastrum leaf extract was mixed with chloroform, followed by the addition of sulphuric acid, yielding a crimson hue indicative of steroids25. The alkaloids test by Wagner involved mixing the leaf extract with potassium iodide and iodine solution, resulting in a precipitate that signified the presence of alkaloids.26
 |
Figure 2: Phytochemicals in Trianthema portulacastrum Click here to View Figure |
Results and Discussion
Functional group analysis
The FTIR spectra of the synthesised ZnO nanoparticles exhibits peaks at 3432.53 cm⁻¹ (O–H stretching), 3209.64 cm⁻¹ (N–H stretching), and 2927.26 cm⁻¹ (C–H stretching), signifying the presence of hydroxyl, amine, and aliphatic groups from plant-derived biomolecules. The signal at 1646.02 cm⁻¹ indicates C=O stretching, implying the presence of amide or carboxylic groups. Supplementary peaks at 1484.58 cm⁻¹ and 1401.77 cm⁻¹ are ascribed to aromatic C–C stretching and carboxylate groups, respectively. The bands at 1307.07 cm⁻¹ and 1083.61 cm⁻¹ signify C–N and C–O stretching vibrations, indicating the presence of amines and alcohols. Peaks at 880.26 cm⁻¹ and 778.56 cm⁻¹ correspond to out-of-plane bending of =C–H in alkenes, while peaks at 693.2, 617.95, 511.24, and 464.45 cm⁻¹ are indicative of Zn–O stretching vibrations, thereby validating the creation of ZnO nanoparticles. The results indicate that phytochemicals from the plant extract function as reducing and capping agents, hence stabilising the ZnO nanoparticles during production.
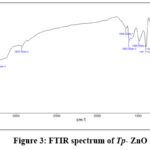 |
Figure 3: FTIR spectrum of Tp– ZnO Click here to View Figure |
UV-Vis absorbance spectrum
An absorption peak at 613.6 nm in the UV-Vis spectra of zinc oxide (ZnO) nanoparticles is unusual, as pure ZnO generally displays a prominent absorption peak about 375 nm owing to its large band gap of approximately 3.3 eV. The detected red-shifted absorbance at 613.6 nm indicates alterations in the structure or composition of the ZnO nanoparticles.
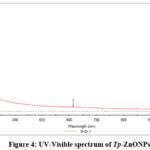 |
Figure 4: UV-Visible spectrum of Tp-ZnONPs Click here to View Figure |
Hydrodynamic Size Determination via DLS
Dynamic Light Scattering examination of zinc oxide nanoparticles synthesised from Trianthema portulacastrum leaf extract showed a hydrodynamic diameter of 97.8 nm, a PDI of 0.285, and a diffusion constant of 5.029 × 10⁻⁸ cm²/s. According to the PDI, the particles are mostly evenly spread out, indicating a fairly monodisperse distribution. It’s likely that the stated average size of 134.2 nm is an intensity-weighted mean, which means that it can be changed by bigger particles or groups of particles, since DLS measurements are sensitive to these kinds of changes.
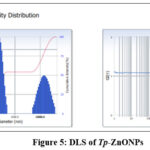 |
Figure 5: DLS of Tp-ZnONPs Click here to View Figure |
Morphological Features of ZnO Nanoparticles
To further characterise the ZnO NPs, their appearance and structure were examined using SEM. Figure 6 depicts ZnO nanoparticles with a particle-like morphological appearance and a spherical shape with a uniform size distribution, as described in the literature 27. Nanoparticles agglomerate and cluster as a direct result of their high surface energy.
 |
Figure 6: SEM images of Tp– ZnO NPs at 1µm and 2 µm. Click here to View Figure |
Crystallographic Characterization
X-ray diffraction (XRD) study of zinc oxide (ZnO) nanoparticles synthesised with Trianthema portulacastrum leaf extract exhibited unique peaks at 2θ values of 31.72°, 34.37°, and 36.21°, correlating with the (100), (002), and (101) crystallographic planes. The peaks are indicative of the hexagonal wurtzite structure of ZnO, as validated by JCPDS28 card no. 36-1451. The predominance of the (101) peak indicates a favoured growth orientation along this plane, signifying anisotropic crystal formation. This growth behaviour frequently results in the creation of rod-like or hexagonal prism-shaped nanoparticles29. The lack of extraneous impurity peaks in the XRD pattern (Figure 7) validates the high purity and crystallinity of the synthesised ZnO nanoparticles.
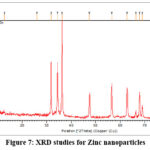 |
Figure 7: XRD studies for Zinc nanoparticles Click here to View Figure |
Antimicrobial assay
The zinc oxide nanoparticles (ZnO-NPs) synthesised with Trianthema portulacastrum (Sharunai leaf extract) exhibited diverse antibacterial efficacy against various microbes owing to their distinctive physicochemical characteristics. Nanoparticles effectively hinder microbial growth by generating reactive oxygen species (ROS), which induce oxidative stress that damages bacterial cell membranes, proteins, and DNA, ultimately leading to cell death. The discrepancies in inhibitory zones relative to gentamicin are probably attributable to variations in the susceptibility of bacteria to ZnO-NPs. The greater inhibition noted against Mycobacterium tuberculosis (25 mm/ml) in comparison to gentamicin (22 mm/ml) indicates that ZnO-NPs efficiently target its cell wall structure. The diminished inhibition of Candida albicans (16 mm/ml) and Fusarium (18 mm/ml) in comparison to gentamicin suggests a comparatively reduced antifungal efficacy, potentially attributable to the thick fungal cell wall serving as a barrier (Figure 8, 9). The differences in activity between gram-positive (Pneumococci) and gram-negative (Escherichia coli) bacteria show how cell wall composition affects susceptibility. The phytochemicals obtained from the Sharunai leaf extract may augment the antibacterial activity of ZnO-NPs, resulting in a synergistic effect. This highlights the capability of plant-mediated ZnO nanoparticles as environmentally acceptable and efficient antibacterial agents.
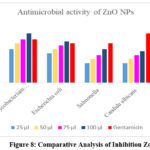 |
Figure 8: Comparative Analysis of Inhibition Zone Click here to View Figure |
Mechanism
Reactive Oxygen Species (ROS) generation
ROS such superoxide anions (O₂⁻), hydrogen peroxide (H₂O₂), and hydroxyl radicals (•OH) are produced by ZnO-NPs, contributing to their antibacterial capabilities. ROS damage microbial cell components, killing them. The figure 9 illustrates the same.
Superoxide anion formation: O2 + e– → O2 –
Hydrogen peroxide formation: 2O2 + 2H+ → H2O2 + O2
Hydroxyl radical formation: H2O2 + e– → .OH + OH–
Release of Zinc Ions (Zn²⁺)
ZnO-NPs can release Zn²⁺ ions in aqueous environments. These ions can interact with microbial enzymes and proteins, disrupting essential metabolic pathways and further inhibiting microbial growth.
Zinc ion release: ZnO + 2H+ → Zn2+ + H2O
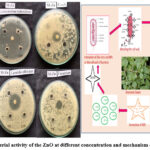 |
Figure 9: Antibacterial activity of the ZnO at different concentration and mechanism of Tp-ZnO NPS Click here to View Figure |
Conclusion
The effective green synthesis of ZnO nanoparticles utilising Trianthema portulacastrum extract illustrates a viable, non-toxic, and scalable alternative to traditional nanoparticle fabrication methods. This method utilises the plant’s own phytochemicals—flavonoids, phenolics, and alkaloids—as bioreductants and stabilisers, resulting in the creation of precisely defined nanostructures with elevated purity and bioactivity. Comprehensive spectroscopic and morphological evaluations confirmed the structural integrity, functionalisation, and homogeneity of the nanoparticles. The biosynthesised ZnO nanoparticles exhibited significant antibacterial efficacy, especially against Gram-positive bacteria and Mycobacterium tuberculosis, underscoring their medicinal potential. The plant-based synthesis process guarantees minimal environmental impact and financial efficiency, rendering it appropriate for large-scale production. This research advances the subject of green nanotechnology and establishes a scientific basis for the application of ethnomedicinal plants in sophisticated biomedical uses, including tailored antimicrobial delivery systems, wound healing, and biocompatible nanocomposites.
Acknowledgment
I thank the Management and the students for their support.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Author Contributions
All the authors contributed significantly to this manuscript, participated in reviewing/editing and approved the final draft for publication.
References
- Yadav, A.; Kumar, H.; Kumar, P.; Rani, G.; Maken, S. Mol. Struct. 2025, 1325, 141017. https://doi.org/10.1016/j.molstruc.2024.141017
CrossRef - Li, K.; Ren, Y.; Lin, D.; Tong, M.; Yang, B.; Xiao, F.; Hou, Y.; Lu, Y.; He, J. Chem. Commun. 2025, 151, 114577. https://doi.org/10.1016/j.inoche.2025.114577
CrossRef - Ilavenil, K. K.; Senthilkumar, V.; Kasthuri, A. Catal. 2025, 2, 3. https://doi.org/10.1007/s44344-025-00007-6
CrossRef - Mahmoud, A. E. D.; Stolle, A.; Stelter, M. ACS Sustainable Chem. Eng. 2018, 6, 25. https://doi.org/10.1021/acssuschemeng.8b00147
CrossRef - Ashwin, B. M., Yardily, A., & Dennison, M. S. Discover Applied Sciences, 2025, 7 (3), 177. https://doi.org/10.1007/s42452-025-06563-8
CrossRef - Mellinas, C.; Jiménez, A.; Garrigós, M. C. Molecules 2019, 24, 4048. https://doi.org/10.3390/molecules24224048
CrossRef - Naiel, B., Fawzy, M., Halmy, M. W. A., & Mahmoud, A. E. D. Scientific Reports, 2022, 12(1), 20370 https://doi.org/10.1038/s41598-022-24805-2
CrossRef - Aldeen, T. S.; Mohamed, H. E. A.; Maaza, M. Phys. Chem. Solids 2022, 160, 110313. https://doi.org/10.1016/j.jpcs.2021.110313
CrossRef - Mahmoud, A. E. D., El-Maghrabi, N., Hosny, M., & Fawzy, M. Sci. Pollut. Res., 2022, 29(59), 89772–89787.
https://doi.org/10.1007/s11356-022-21871-x
CrossRef - Golzarnezhad, F., Allahdou, M., Mehravaran, L., & Naderi, S. Discover Applied Sciences, 2025, 7(3), 196. https://doi.org/10.1007/s42452-025-06623-z
CrossRef - Chabattula, S. C., Gupta, P. K., Tripathi, S. K., Gahtori, R., Padhi, P., Mahapatra, S., Biswal, B. K, Today Chem., 2021, 22, 100618. https://doi.org/10.1016/j.mtchem.2021.100618
CrossRef - Rauf, M. A.; Oves, M.; Rehman, F. U.; Khan, A. R.; Husain, N. Pharmacother. 2019, 116, 108983. https://doi.org/10.1016/j.biopha.2019.108983
CrossRef - Agarwal, H.; Menon, S.; Shanmugam, V. K. Interfaces 2020, 19, 100521. https://doi.org/10.1016/j.surfin.2020.100521
CrossRef - Ganesan, L.; Arumugam, M.; Maluventhen, V. Mater. Devices 2025. https://doi.org/10.1007/s44174-025-00346-w
CrossRef - Xia, Y.; Yang, P.; Sun, Y.; Wu, Y.; Mayers, B.; Gates, B.; Yin, Y.; Kim, F.; Yan, H. Mater. 2003, 15, 353–389. https://doi.org/10.1002/adma.200390087
CrossRef - Lahoti, R.; Carroll, D. Next Res. 2025, 1, 100164. https://doi.org/10.1016/j.nexres.2025.100164
CrossRef - Babayevska, N., Przysiecka, Ł., Iatsunskyi, I., Nowaczyk, G., Jarek, M., Janiszewska, E., & Jurga, S. Rep., 2022, 12(1), 8148. https://doi.org/10.1038/s41598-022-12134-3
CrossRef - Gaddeyya, G.; Kumar, R. P. K. Crop Weed 2015, 11, 47–54.
- Falade, T.; Ishola, I. O.; Akinleye, M. O.; Oladimeji-Salami, J. A.; Adeyemi, O. O. Ethnopharmacol. 2019, 238, 111831. https://doi.org/10.1016/j.jep.2019.111831
CrossRef - Poddar, S.; Ghosh, P.; Sarkar, T.; Sarkar, A.; Choudhury, S.; Chatterjee, S. Pharm. Sci. Res. 2020, 12(7), 899–903.
- Ghosh, P.; Chatterjee, S.; Das, P.; Banerjee, A.; Karmakar, S.; Mahapatra, S. J. Pharm. Sci. Res. 2019, 10(4), 1605–1612. https://doi.org/10.13040/IJPSR.0975-8232.10(4).1605-12
CrossRef - Yamaki, J.; Nagulapalli Venkata, K. C.; Mandal, A.; Bhattacharyya, P.; Bishayee, A. Integr. Med. 2016, 14(2), 84–99. https://doi.org/10.1016/S2095-4964(16)60247-9
CrossRef - Jaiswal, V.; Lee, H.-J. Plants 2025, 14(3), 349. https://doi.org/10.3390/plants14030349
CrossRef - Chowdhury, V.; Shristy, N. T.; Rahman, M. H.; Chowdhury, T. A. Dhaka Univ. J. Pharm. Sci. 2022, 21(1), 33–43. https://doi.org/10.3329/dujps.v21i1.60394
CrossRef - Laasya, T. P. S.; Thapliyal, S.; Goel, K. K.; Kumar, B.; Poduri, R.; Joshi, G. Sci. Plus 2024, 7(2), 2300140. https://doi.org/10.1002/sscp.202300140
CrossRef - Okolo, S. C.; Habila, J. D.; Hamisu, I. J. Chem. Res. 2024, 29(2), 115–121. https://doi.org/10.4314/njcr.v29i2.4
CrossRef - Saputra, I. S.; Nurfani, E.; Fahmi, A. G.; Saputro, A. H.; Apriandanu, D. O. B.; Annas, D.; Yulizar, Y. Vacuum 2024, 227, 113434. https://doi.org/10.1016/j.vacuum.2024.113434
CrossRef - Arefi, M. R.; Rezaei-Zarchi, S. R. J. Mol. Sci. 2012, 13(4), 4340–4350. https://doi.org/10.3390/ijms13044340
CrossRef - Cao, D.; Gong, S.; Shu, X.; et al. Nanoscale Res. Lett. 2019, 14, 210. https://doi.org/10.1186/s11671-019-3038-3
CrossRef
Accepted on: 07 Aug 2025
Second Review by: Dr. Paresh Patel
Final Approval by: Dr. Charanjeet Kaur

















