Molecular Docking Evaluation of Flavonoids from Fenugreek (Trigonella foenum-graecum) as Potential DPP-4 Inhibitors for Antidiabetic Activity
1Department of Pharmaceutics, College of Pharmacy, Jazan University, Jazan, KSA
2Department of Pharmaceutical Chemistry, K.B.H.S.S Trust's, Institute of Pharmacy, Malegaon, Nashik, Maharashtra, India.
3Department of ECE, Loyola Institute of Technology and Science, Loyola Nagar, Thovalai, Kanyakumari, India.
4Department of Pharmaceutical Sciences, Maharshi Dayanand University, Rohtak Haryana, India.
5Department of Quality Assurance, Aura Biosciences, Guest Street, Boston, MA.
6Department of Pharmaceutical Technology, Brainware University, Barasat, Kolkata, West Bengal.
7Department of Pharmacy, Anuradha college of pharmacy, Chikhli, Maharashtra, India.
8Advanced Institute of Pharmacy, 70 KM Stone, Delhi Mathura Road, Near Aurangabad, Palwal, Haryana, India.
Corresponding Author E-mail:armandalal1997@gmail.com
Download this article as:
ABSTRACT:Type 2 Diabetes Mellitus is an extensive metabolic disorder, and dipeptidyl peptidase-4 is an important target for therapeutic strategies (World Health Organization, 2021). This study uses in silico molecular docking to examine the potential of four flavonoids from Trigonella foenum-graecum (fenugreek) seeds: rutin, quercetin, vitexin, and isovitexin, as natural dipeptidyl peptidase-4 (DPP-4) inhibitors for antidiabetic action. AutoDock Vina was used to dock the chosen ligands with the DPP-4 enzyme (PDB ID: 2OQV) while minimizing their energy. The binding interactions were then examined. At important catalytic residues, rutin showed significant hydrogen bonding and hydrophobic interactions, outperforming the co-crystallized inhibitor with the greatest binding affinity (−9.4 kcal/mol). Rutin's molecular complementarity was found to be favorable when the interaction profiles and binding orientations were examined using Discovery Studio Visualizer. According to the study, rutin's better binding affinity and interaction network suggest that it may have natural DPP-4 inhibitory properties. These findings offer valuable insights into fenugreek’s antidiabetic efficacy and support its traditional usage, suggesting further experimental validation of rutin for drug development.
KEYWORDS:DPP-4 Inhibitors; Discovery Studio; Flavonoids; Incretin Hormones; Molecular Docking; Rutin
Introduction
Diabetes mellitus is a chronic metabolic condition characterized by high blood glucose levels, due to both inadequate insulin synthesis, impaired responsiveness to insulin, or both. It creates a huge worldwide health concern in the modern period. Diabetes is referred to into three main types: Type 1 diabetes mellitus (T1DM), a form of autoimmune disease labeled by limited or absent insulin production; Type 2 diabetes mellitus (T2DM), most strongly prominent form, comprising insulin resistance and inadequate insulin secretion; and gestational diabetes, that arises during pregnancy.1 As per the World Health Organization, global incidence of diabetes among adults has risen considerably since 1980. In 2021, around 537 million adults aged 20–79 were living with diabetes, with forecasts expecting this number to climb to 643 million by 2030, 783 million by 20452,3.
Diabetes puts a heavy financial strain on healthcare systems around the world and is a contributing factor to consequences such cardiovascular diseases, neuropathy, nephropathy, and retinopathy. The hunt for novel medicines and other approaches to better handle and treat diabetes has accelerated as a result of these expanding trends4.
For the treatment of type 2 diabetes, dipeptidyl peptidase-4 (DPP-4), a crucial regulator of glucose metabolism, is an essential pharmacological target. Incretin hormones that stimulate insulin secretion in a glucose-dependent manner, such as gastric inhibitory polypeptide (GIP) and glucagon-like peptide-1 (GLP-1), are quickly deactivated by this enzyme5. DPP-4 shortens the half-life of these hormones and lessens their insulinotropic properties by breaking them down. Consequently, DPP-4 inhibition increases incretin activity, which improves glycemic control, lowers glucagon release, and increases insulin secretion6.
Currently licensed for clinical usage are a number of synthetic DPP-4 inhibitors, including vildagliptin, saxagliptin, and sitagliptin7. However, the long-term safety of these agents is yet unknown, and they are frequently linked to negative side effects include joint pain, pancreatitis, and allergic responses. As a result of their potential for safer therapeutic results, interest in DPP-4 inhibitors produced from plants has grown8.
A common leguminous plant in South Asia, the Mediterranean, and North Africa, fenugreek (Trigonella foenum-graecum) has long been used in Ayurvedic and Unani medicine to treat a variety of illnesses, most notably diabetes9. Its hypoglycemic, hypolipidemic, antioxidant, anti-inflammatory, and gastroprotective benefits have all been confirmed by recent pharmacological research10. Fenugreek’s extensive phytochemical composition, which includes flavonoids, alkaloids, fibers, amino acids (particularly 4-hydroxyisoleucine), and saponins, is thought to be responsible for its antidiabetic properties11.
Bioactive polyphenols called flavonoids, which include quercetin, rutin, vitexin, and isovitexin, are present in fenugreek and are thought to play a major role in its antidiabetic effects. With its three benzene rings and five hydroxyl groups, quercetin is a strong antioxidant that can scavenge free radicals and inhibit enzymes12. It has been demonstrated that the quercetin glycoside rutin inhibits DPP-4 and promotes vascular health. Flavone glucosides with structural similarities, vitexin and isovitexin, have hypoglycemic and anti-inflammatory effects. Through π-π stacking, hydrophobic forces, and hydrogen bonding, their C6-C3-C6 backbone enables a variety of interactions with proteins13,14.
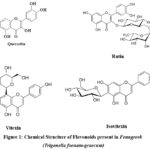 |
Figure 1: Chemical Structure of Flavonoids present in Fenugreek (Trigonella foenum-graecum) Click here to View Figure |
Considering the biological importance of these flavonoids, in silico molecular docking methods offer a swift and economical means to assess their binding affinities and interaction patterns with particular protein targets such as DPP-4. This study utilised AutoDock Vina, a prevalent docking software, to predict the binding conformation and affinity of quercetin, rutin, vitexin, and isovitexin with the DPP-4 enzyme (PDB ID: 2OQV). The 2OQV structure is a thoroughly characterised crystal structure of human DPP-4 co-crystallized with a recognised inhibitor, offering a dependable model for virtual screening. Ligand compounds underwent energy minimisation and were transformed into appropriate forms via Open Babel and AutoDock Tools, while the protein structure was constructed by eliminating water molecules, including polar hydrogens, and delineating the active site grid. Eleven
Subsequent to docking, the resultant complexes were assessed via Discovery Studio Visualiser, which facilitated a comprehensive examination of molecular interactions. The docking scores and interaction profiles were subsequently compared with those of the conventional inhibitor to evaluate the relative efficacy of the chosen flavonoids. This approach facilitates the identification of promising drug development leads and clarifies structure-activity correlations that can inform the synthesis of more potent analogues. 12
This research investigates the antidiabetic potential of flavonoids from fenugreek seeds by in silico docking studies focused on the DPP-4 enzyme. This study integrates phytochemistry with computational biology, and addressing the global need for safer and more effective antidiabetic medicines. The results are anticipated to provide an understanding of the molecular mechanisms underlying fenugreek’s traditional application and facilitate the creation of innovative plant-based DPP-4 inhibitors. Fourteen
Materials and Methods
Ligand Selection
The four flavonoid compounds—quercetin, rutin, vitexin, and isovitexin—were chosen due to their documented occurrence in Trigonella foenum-graecum and established antidiabetic properties. Each compound was transformed into PDBQT format via AutoDock Tools 1.5.7, subsequent to energy minimisation employing the MMFF94 force field. Partial charges were allocated, torsions were modified, and non-polar hydrogens were consolidated during the ligand preparation stage.15
Preparation of Protein Targets
The crystal structure of human dipeptidyl peptidase-4 (DPP-4) complexed with a known inhibitor (PDB ID: 2OQV) was sourced from the RCSB Protein Data Bank . The structure was imported into AutoDock Tools for its processing. Water molecules and heteroatoms were eliminated, polar hydrogens were included, and Kollman charges were allocated. The active site was delineated according to the region where the CCL inhibitor binds. The finalised protein structure was stored in PDBQT format for docking purposes.16
Grid Box Configuration
A grid box was established to include the active site of DPP-4; utilising coordinates derived from the inhibitor binding area in the 2OQV crystal structure. Seventeen. The grid dimensions were established as follows:
Centre:X=40.123,Y=38.456,Z=50.278
Dimensions:X=25Å,Y=25Å,Z=25Å
These dimensions guaranteed that the binding pocket and adjacent residues were sufficiently encompassed to facilitate appropriate ligand flexibility and orientation.
Molecular Docking Utilising Auto Dock Vina
Molecular docking studies were performed using Auto Dock Vina (v1.1.2) for its rapidity and precision in predicting binding affinity. The docking was executed in command-line mode utilising default parameters, with an exhaustiveness level configured to 8 to enhance sampling precision. Each ligand was docked separately to the DPP-4 protein. The binding affinities (in kcal/mol) and ligand conformations were documented. The highest-ranked conformation with the minimal binding energy for each ligand was chosen for subsequent investigation.18
Visualisation and Interaction Assessment
A post-docking analysis was performed utilising Discovery Studio Visualiser (v2021) to investigate the characteristics of ligand–protein interactions. Crucial chemical interactions, including hydrogen bonding, hydrophobic interactions, π–π stacking, van der Waals forces, and electrostatic contacts, were discerned between the ligands and amino acid residues in the DPP-4 active site. Ligand orientation, bond lengths, and interacting residues were documented to evaluate binding affinity and compare with the reference inhibitor.19
Oversight and Verification
The docking process was validated by redocking the co-crystallized inhibitor from the 2OQV structure to confirm the precision of the docking method. The root mean square deviation (RMSD) between the docked and crystallised conformations was computed, with values under 2.0 Å being acceptable, signifying reliable docking parameters.20
Table 1: Docking Score and Interaction Summary
| Compound | Binding Affinity (kcal/mol) | Key Hydrophobic Interactions | Key Hydrophilic Interactions | Key Interacting Residues |
| Rutin | −9.4 | Val207, Tyr547, Trp629 | Glu205, Ser630, Asp708, Glu206 | Tyr662, Glu205, Glu206, Ser630, Trp629 |
| Quercetin | −8.7 | Val207, Tyr547, Ile472 | Glu205, Ser630 | Tyr547, Glu205, Ser630, Trp629 |
| Vitexin | −8.3 | Ile472, Tyr662 | Glu205, Ser630, Asp739 | Tyr662, Ser630, Glu205 |
| Isovitexin | −8.1 | Ile472, Val207 | Glu205, Ser209 | Val207, Ser209, Glu205 |
| Co-crystallized Ligand | −9.1 | Val207, Tyr547, Trp629 | Glu206, Glu205, Ser630, Tyr662 | Glu205, Glu206, Tyr662, Trp629 |
Results and Discussion
Overview of Docking Affinity
The molecular docking simulation performed using AutoDock Vina revealed favourable binding affinities for all four selected flavonoids from Trigonella foenum-graecum with the DPP-4 enzyme (PDB ID: 2OQV). Rutin exhibited the highest binding affinity of −9.4 kcal/mol, surpassing the binding energy of the co-crystallized reference inhibitor, which was −9.1 kcal/mol. Quercetin demonstrated a binding energy of −8.7 kcal/mol, but vitexin and isovitexin exhibited lower binding energies of −8.3 and −8.1 kcal/mol, respectively. The negative findings indicate spontaneous binding and strong molecular interactions with the active site of the DPP-4 enzyme, suggesting the potential of these flavonoids as natural DPP-4 inhibitors.21
Interaction Profile of Rutin
Rutin, a flavonoid glycoside consisting of quercetin and the disaccharide rutinose, exhibited a robust interaction profile with the DPP-4 protein. The compound exhibited both hydrophobic and hydrophilic interactions, enhancing its binding affinity.22
Hydrophobic interactions were detected with residues Val207, Tyr547, and Trp629, situated in the S1 pocket of the enzyme. These residues are crucial for stabilising ligand ingress via π–π stacking and van der Waals interactions. The hydrophilic contacts comprised Glu205, Ser630, Asp708, and Glu206, which constitute the enzyme’s active catalytic triad. The hydrogen interaction between the numerous hydroxyl groups of rutin and these residues increases binding stability, particularly owing to their polar characteristics.23
Analysis using Discovery Studio verified that rutin establishes 5 hydrogen bonds, 3 hydrophobic interactions, and participates in π–π interactions with Tyr662, a crucial binding determinant inside the DPP-4 active site. This intricate network of contacts enhances rutin’s substantial docking affinity and suggests favourable structural complementarity with the DPP-4 binding region. Twenty-four.
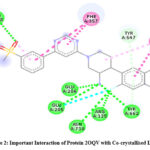 |
Figure 2: Important Interaction of Protein 2OQV with Co-crystallised Ligand Click here to View Figure |
Co-crystallized Ligand vs. Rutin
The correctness of the docking approach was confirmed by the binding score of -9.1 kcal/mol obtained from the docking of the co-crystallized ligand. The docking approach successfully replicated the native binding orientation, as evidenced by the re-docked ligand’s root mean square deviation (RMSD) of less than 1.8 Å from its initial crystallographic pose. Remarkably, the ligand established crucial connections with residues Glu205, Glu206, Tyr662, and Ser630, which are the same as those found in the binding profile of rutin. This overlap highlights how rutin may resemble the binding patterns of well-known DPP-4 inhibitors.
Furthermore, because of its extra sugar moiety, which allowed for the creation of a wider hydrogen bond network, rutin showed an extended interaction network. Compared to lesser flavonoids or synthetic analogs, rutin may be able to block the DPP-4 active site more successfully due to its structural advantage, which could improve enzyme inhibition.
Other Flavonoids: Quercetin, Vitexin, and Isovitexin
The docking score for quercetin, the rutin aglycone, was -8.7 kcal/mol. Quercetin interacted well with important amino acids such as Tyr547, Glu205, and Ser630 despite not having a sugar group. Although it may have fewer hydrogen bonds due to its lower molecular size, it has more conformational flexibility inside the binding pocket. Its somewhat lower affinity is probably caused by the smaller number of hydrogen bond donors, even though its binding profile is very similar to that of rutin. Two C-glycosylated flavones, vitexin and isovitexin, showed modest binding affinities (−8.3 and −8.1 kcal/mol, respectively). Both substances demonstrated good compatibility with the DPP-4 binding site by forming hydrogen bonds with Glu205 and Ser630. Their lower binding energies, however, may be explained by their restricted capacity to generate extensive polar contacts due to their more stiff molecular architectures and small number of active hydroxyl groups.
Pharmacological Implications
Rutin’s high binding affinity suggests that it could be a useful natural DPP-4 inhibitor. With the extra benefit of being plant-derived, it may have a safer pharmacological profile than synthetic inhibitors while still having the same capacity to engage important catalytic residues. Fenugreek seeds’ traditional usage in the treatment of diabetes is supported by the presence of rutin in them. Additionally, when combined with other fenugreek components like quercetin, saponins, and dietary fibers, rutin’s synergistic actions may help create a more comprehensive and successful glycemic management plan.
Considering the positive docking results, more experimental verification is necessary. To verify rutin’s therapeutic effectiveness, in vitro tests such as cellular models and enzyme inhibition studies are crucial, as are in vivo research employing diabetic animal models. To assess its pharmacokinetic appropriateness for clinical usage, ADMET (Absorption, Distribution, Metabolism, Excretion, and Toxicity) profiling should also be carried out26.
Limitations and Future Scope
While molecular docking is a robust preliminary screening method, it has inherent limitations. The current docking simulation does not account for protein flexibility, solvent effects. .
Moreover, flavonoids such as rutin often suffer from poor bioavailability due to limited intestinal absorption and rapid metabolism. Future studies should focus on developing rutin-based nanoformulations or glycosylation strategies to enhance its therapeutic effectiveness.
This in silico docking study demonstrates that rutin, a major flavonoid in fenugreek, shows strong potential as a natural inhibitor of the DPP-4 enzyme, with better docking affinity than the co-crystallized synthetic ligand. The interaction with key catalytic residues supports its role in modulating incretin activity and thus lowering blood glucose levels. The findings reinforce the antidiabetic efficacy of Trigonella foenum-graecum and highlight flavonoids as promising candidates for future drug development targeting T2DM.
Conclusion
This in silico study sought to assess the antidiabetic efficacy of specific flavonoid compounds—rutin, quercetin, vitexin, and isovitexin—derived from Trigonella foenum-graecum (fenugreek), through molecular docking with the enzyme dipeptidyl peptidase-4 (DPP-4), a recognised therapeutic target in the treatment of type 2 diabetes mellitus (T2DM). The docking simulations were conducted with AutoDock Vina, and Discovery Studio was utilised for the analysis of protein-ligand interactions.
The study demonstrated that all four flavonoids displayed advantageous binding interactions with the DPP-4 enzyme, with rutin showing the highest binding affinity (−9.4 kcal/mol), exceeding that of the co-crystallized inhibitor (−9.1 kcal/mol). Rutin exhibited robust interactions with critical amino acid residues including Tyr547, Glu205, Ser630, and Trp629, establishing both hydrogen bonds and hydrophobic interactions. These findings indicate a stable and efficient binding conformation in the active region of DPP-4, suggesting possible inhibitory activity.
The results are markedly important given the adverse effects and financial constraints linked to current synthetic DPP-4 inhibitors. Rutin and associated flavonoids from fenugreek offer a natural and potentially safer alternative, reinforcing the historic application of this herb in diabetes control. This study emphasises the significance of employing computational techniques as initial tools in drug development, conserving both time and resources prior to doing biological experiments.
This study provides significant insight into the molecular mechanisms that underpin the antidiabetic effects of fenugreek and emphasises the therapeutic potential of plant-derived DPP-4 inhibitors, especially flavonoids. It further substantiates the notion that dietary phytochemicals might affect metabolic pathways and provide promise for nutraceutical advancement.
Future Research Directions
Notwithstanding the encouraging results, numerous measures are required to further authenticate and enhance the therapeutic potential of rutin and other flavonoids as DPP-4 inhibitors:
In Vitro Validation: The docking outcomes must be corroborated using enzymatic inhibition experiments utilising pure DPP-4 to verify genuine inhibitory effects. Research using pancreatic β-cells and intestinal L-cells will evaluate the effects on incretin hormones and insulin secretion.
In Vivo Studies: Animal models of Type 2 Diabetes Mellitus should be employed to assess the pharmacological effects of rutin on glucose regulation, insulin sensitivity, and β-cell preservation. These investigations are essential for comprehending its efficacy and safety within a complicated biological system.
Bioavailability Enhancement: Given that flavonoids such as rutin frequently exhibit suboptimal oral bioavailability, it is imperative to investigate formulation options including nanoencapsulation, liposomes, or phytosomes to improve their systemic absorption and therapeutic efficacy.
Structure–Activity Relationship (SAR) Analysis: Additional chemical modifications of rutin may be produced to enhance binding efficacy and pharmacokinetic characteristics.
Toxicity and ADMET Profiling: A thorough assessment of absorption, distribution, metabolism, excretion, and toxicity is crucial to guarantee safety, particularly for prolonged usage in diabetic populations.
Synergistic Effects: Examining the combined impact of rutin with other fenugreek components (e.g., saponins, alkaloids, dietary fibre) or with current antidiabetic medications may uncover synergistic effects, resulting in multi-targeted therapy.
Clinical Evaluation: Following successful preclinical validation, clinical studies should be structured to assess the efficacy of rutin-rich formulations in human diabetes cohorts, emphasising indicators such as HbA1c, fasting glucose, and insulin resistance.
This work establishes a solid basis for future investigations into plant-based antidiabetic treatments and identifies rutin as a prominent contender for further advancement. Combining computational docking with laboratory and clinical validation helps expedite the transition from conventional knowledge to evidence-based phytopharmaceuticals.
Acknowledgment
We acknowledge the use of computational resources from [insert institution or facility name] for molecular docking and analysis.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Sarker, D. K.; Ray, P.; Rouf, R.; et al. In Silico Molecular Docking and Dynamic Investigations of Bioactive Phytoconstituents from Fenugreek Seeds as a Potent Drug against DPP IV Enzyme. ACS Food Sci. Technol. 2023, 3(9), 1423–1439.
CrossRef - Bhatti, J. S.; Sherawat, A.; Mishra, J.; et al. Oxidative Stress in the Pathophysiology of Type 2 Diabetes and Related Complications: Current Therapeutic Strategies and Future Perspectives. Free Radic. Biol. Med. 2022, 184, 114–134.
CrossRef - Fatima, H.; Shahid, M.; et al. HPLC DAD MS Characterization, Antioxidant Activity, α-Amylase Inhibition, Molecular Docking, and ADMET of Flavonoids from Fenugreek Seeds. 2024, 28(23), 7798.
CrossRef - He, Y.; Ding, C.; Wang, X.; et al. Using Response Surface Methodology to Optimize Separation of Polyphenol Compounds from Fenugreek Seeds. Liq. Chromatogr. Relat. Technol. 2022, 38, 29–35.
CrossRef - Huang, P. K.; Lin, S. R.; Chang, C. H.; et al. Natural Phenolic Compounds Potentiate Hypoglycemia via Inhibition of Dipeptidyl Peptidase IV. Rep. 2019, 9, 15585.
CrossRef - Dias, M. C.; Pinto, D. C. G. A.; Silva, A. M. S. Plant Flavonoids: Chemical Characteristics and Biological Activity. 2021, 26(26), 5377.
CrossRef - Hussain, T.; Tan, B.; Murtaza, G.; et al. Flavonoids and Type 2 Diabetes: Evidence of Efficacy in Clinical and Animal Studies and Delivery Strategies to Enhance Their Therapeutic Efficacy. Res. 2020, 152, 104629.
CrossRef - Proença, C.; Ribeiro, D.; Freitas, M.; Fernandes, E. Flavonoids as Potential Agents in the Management of Type 2 Diabetes through Modulation of α-Amylase and α-Glucosidase Activity: A Review. Rev.. Food Sci. Nutr. 2021, 62(22), 3137–3207.
CrossRef - Pan, J., Zhang, Q., Zhang, C., Yang, W., Liu, H., Lv, Z., Liu, J. and Jiao, Z. Inhibition of dipeptidyl peptidase-4 by flavonoids: Structure–activity relationship, kinetics and interaction mechanism. Front Nutr. 2022, 9, 892426.
CrossRef - Alghamdi, S. S.; Suliman, R. S.; Alsaeed, A. S.; et al. Novel Anti-Tubulin Compounds from Trigonella foenum-graecum Seeds; Insights into In Vitro and Molecular Docking Studies. Drug Des. Devel. Ther. 2021, 15, 4195–4211.
CrossRef - Benayad, Z.; Gómez Cordovés, C.; Es Safi, N. E. Characterization of Flavonoid Glycosides from Fenugreek Crude Seeds by HPLC DAD ESI/MS Analysis. J. Mol. Sci. 2014, 15(11), 20668–20685.
CrossRef - Idris, S.; Mishra, A.; Khushtar, M. Recent Therapeutic Interventions of Fenugreek Seed: A Mechanistic Approach. Drug Res. (Stuttg.) 2021, 71(4), 180–192.
CrossRef - Alu’datt, M. H.; Rababah, T.; Al Ali, S.; et al. Current Perspectives on Fenugreek Bioactive Compounds and Their Potential Impact on Human Health: A Review of Recent Insights into Functional Foods and Other High-Value Applications. Food Sci. 2024, 89(4), 1835–1864.
CrossRef - Kan, R.; Ren, P.; Wu, A.; et al. Identification and Molecular Docking Study of Sugarcane Leaf-Derived Compounds as Potent Dipeptidyl Peptidase IV, α-Glucosidase, and α-Amylase Inhibitors. Sci. Food Agric. 2023, 103(11), 5388–5400.
CrossRef - He, Y.; Ding, C.; Wang, X.; et al. Identification of Curcumin as a Potential α-Glucosidase and DPP 4 Inhibitor: Molecular Docking, In Vitro and In Vivo Evaluation. Pharm. Pharmacol. 2021, 73(4), 487–495.
- Sarker, D. K.; Ray, P.; Rouf, R.; et al. In Silico Molecular Docking and Dynamic Investigations of Bioactive Phytoconstituents from Fenugreek Seeds as a Potent Drug against DPP IV Enzyme. ACS Food Sci. Technol. 2023, 3(9), 1423–1439.
CrossRef - Luo, W.; Deng, J.; He, J.; et al. Integration of Molecular Docking, Molecular Dynamics, and Network Pharmacology to Explore the Multi-Target Pharmacology of Fenugreek against Diabetes. Cell. Mol. Med. 2023, 27(14), 1959–1974.
CrossRef - Haxhiraj, M.; White, K.; Terry, C. The Role of Fenugreek in the Management of Type 2 Diabetes. J. Mol. Sci. 2024, 25(13), 6987.
CrossRef - Chhabria, S., Mathur, S., Vadakan, S., Sahoo, D.K., Mishra, P. and Paital, B., 2022. A review on phytochemical and pharmacological facets of tropical ethnomedicinal plants as reformed DPP-IV inhibitors to regulate incretin activity. Front Endocrinol. 2022,13, 1027237.
CrossRef - Abdulai, I.L., Kwofie, S.K., Gbewonyo, W.S., Boison, D., Puplampu, J.B. and Adinortey, M.B., 2021. Multitargeted effects of vitexin and isovitexin on diabetes mellitus and its complications. The Scientific World Journal. 2021, 2021(1), 6641128.
CrossRef - Khenifi, M.L., Serseg, T., Migas, P., Krauze-Baranowska, M., Özdemir, S., Bensouici, C., Alghonaim, M.I., Al-Khafaji, K., Alsalamah, S.A., Boudjeniba, M. and Yousfi, M., 2023. HPLC-DAD-MS characterization, antioxidant activity, α-amylase inhibition, molecular docking, and ADMET of flavonoids from fenugreek seeds. Molecules. 2023, 28(23), 7798.
CrossRef - Kumar, A.; Singh, V. K.; Kayastha, A. M. Molecular Modeling, Docking and Dynamics Studies of Fenugreek (Trigonella foenum-graecum) α-Amylase. Biomol. Struct. Dyn. 2022, 41(19), 9297-312.
CrossRef - Tao, M., Li, R., Zhang, Z., Wu, T., Xu, T., Zogona, D., Huang, Y., Pan, S. and Xu, X., 2022. Vitexin and isovitexin act through inhibition of insulin receptor to promote longevity and fitness in Caenorhabditis elegans. Nutr. Food. Res. 2022, 66(17), 2100845.
CrossRef - Dias, M. C.; Pinto, D. C. G. A.; Silva, A. M. S. Plant Flavonoids: Chemical Characteristics and Biological Activity. Molecules, 2021, 26(26), 5377.
CrossRef - Benayad, Z.; Gómez Cordovés, C.; Es Safi, N. E. Flavonoid Glycosides from Fenugreek Seeds by HPLC DAD ESI/MS Analysis. J. Mol. Sci. 2014, 15(11):20668-85.
CrossRef - Hussain, T.; Tan, B.; Murtaza, G.; et al. Flavonoids and Type 2 Diabetes: Efficacy and Delivery Strategies. Res. 2020, 152 (2020), 104629.
CrossRef
Accepted on: 13 Sep 2025
Second Review by: Dr. Nitin Pandey
Final Approval by: Dr. Ravindra M Kumbhare

















