Identifying the Active Chemical Ingredients in Extracts of Certain Plants with Medicinal Properties Used for the Treatment of Intestinal Diseases
1Department of Chemistry, College of Science, University of Tabuk, Tabuk, Saudi Arabia
2Department of Medical Microbiology, Faculty of Medicine, University of Tabuk, Tabuk, KSA
3Molecular Microbiology and Infectious Diseases Research Unit, University of Tabuk, Tabuk, KSA
Corresponding Author E-mail: khalid.mustafa938@gmail.com
Download this article as:
ABSTRACT:Traditionally, in many places, including Saudi Arabia, the plants Salvia officinalis, Artemisia herba alba, and Teucrium polium have been used to treat different diseases, especially intestinal ones. All three plant-specific water extracts were analysed using FTIR and GC-MS. Several flavonoids, phytosterol and phenolic acids have been identified. In a time-dependent growth suppression experiment, Teucrium polium (O1), Salvia officinalis (O2), and Artemisia herba-alba (O3) were examined for antibacterial activity. The growth curves of Pseudomonas aeruginosa and Staphylococcus aureus at O1, O2, and O3 revealed that these extracts may suppress bacterial growth and reproduction. O1, O2, and O3 had concentration-dependent effects on test microorganisms, with O1 inhibiting P. aeruginosa at 87% and S. aureus at 74% at the minimum inhibitory dose. At 0.5xMIC O1, very equal inhibition was observed. At MIC, the O₂ extract reduces 88%, but at 0.5xMIC, it reduces 63% and 60%. O₃ showed a 10% and 22% difference against P. aeruginosa and S. aureus at MIC and 0.5xMIC, respectively. It has also extended the plant's therapeutic advantages, opening up new applications.
KEYWORDS:Antibacterial Activity; Diseases; HPLC; Time-Dependent Growth; Biofilm Assays
Introduction
Teucrium polium L.
Teucrium polium L. (O1), sometimes known as Germander, is a perennial member of the Lamiaceae family.1,2 This plant originates in the Mediterranean, Southwest Asia, Turkey, and North Africa3,4 and is also used in traditional medicine due to its medicinal characteristics. Antibacterial, anticancer, antioxidant, and analgesic. Its low-glycaemic properties make it an ideal diabetes treatment. Flavonoids, terpenoids, and essential oils all contribute to the plant’s medicinal benefits. Antioxidants and anti-inflammatory substances enhance the plant’s therapeutic properties.5-8
Salvia officinalis
Salvia officinalis (O2), also known as Sage, has been used for decorative, medicinal, and culinary purposes. This Lamiaceae plant has received much research owing to its biological functions and complicated chemical composition. Salvia officinalis is a versatile medicinal plant. Because of its complex chemical structure, it possesses antibacterial, antioxidant, antidiabetic, anti-inflammatory, and neuroprotective activities. Even though the current study has supported Salvia officinalis’ traditional use.9-13
Artemisia herba-alba.
Artemisia herba-alba (O3), often known as desert or white wormwood, is a perennial that has silvery-grey leaves and a pleasant odour. The plant grows 20–40 cm, depending on the environment. Artemisia herba-alba grows in dry and semi-arid regions of southern Europe, the Middle East, and North Africa. Morocco, Algeria, Tunisia, Libya, Egypt, Israel, Jordan, Syria, Iraq, Saudi Arabia, and Spain often have it. The plant grows on hot, well-drained soils with minimal rainfall. Artemisia herba-alba is used in traditional medicine in North Africa and the Middle East. It treats parasites, skin, respiratory, and digestive problems. Plant flavonoids, volatile oils, and other bioactive compounds might explain its medicinal properties. Artemisia herba-alba is used in traditional medicine to alleviate diarrhoea, bloating, and stomach pain. Anthelmintic: kills parasitic worms. The plant’s antimicrobial properties may help to heal skin and wounds. It alleviates coughs and bronchitis. Artemisia herba-alba has antibacterial, anti-inflammatory, and antioxidant activities that support its traditional use.4-18
Materials and Methods
Chemical analysis begins with the collection and preparation of plant samples. The Tabuk region is utilised to collect different components of Salvia officinalis, Artemisia herba alba, and Teucrium polium, such as leaves, fruits, seeds, and roots, for the research.19 To increase extraction efficiency and improve preservation, plant material is frequently dried after collection to reduce moisture content.20 Researchers employed the oven-drying approach to speed up the drying process by working at controlled temperatures (such as 40-60°C). To increase the surface area for solvent extraction, the dried plant material is ground into a fine powder. Each extract sample was prepared for examination by dissolving 80 milli grammes (80 mg) of dried or finely powdered plant material in 250 millilitres of water as a solvent. Until further study, the powdered material is stored in airtight containers that are protected from moisture and light.
HPLC and gas chromatography-mass spectrometry (GC-MS) have been employed to identify bioactive components.21 The researchers employed the GC-MS [Shimadzu of the United States] Method 8260 to analyse volatile organic chemicals and the Method 8270 to investigate semi-solid organic compounds.
Bioactivity
Bacterial Strains Used
We use P aeruginosa and S aureus from laboratory stock S. aureus (ATCC 25923) as standard strains to use control for various experiments. We selected well-characterised, pre-identified strains, cefoxitin-resistant, ESBL positive P. aeruginosa, and methicillin-resistant S. aureus from the stock culture. The P. aeruginosa was reassessed as cefoxitin resistant and ESBL positive, while S. aureus was for methicillin resistance using CLSI guidelines. The standard strains used were P. aeruginosa ATCC 27853 and S aureus ATCC 25923.
Biofilm Formation in 96-well Microtiter Plates
Biofilm formation was examined quantitatively as described elsewhere 22 using the method in 96-well flat bottom plates. Biofilm assays were performed in triplicate, and the mean biofilm absorbance value was determined. Biofilm formed were classified as weak (OD590 0.1 to ≤0.400), moderate (OD590 > 0.400) and strong (OD590 > 0.800).
Minimum Inhibitory Concentration (MIC) Activity of Extracts
MIC of Teucrium polium (O1), Salvia officinalis (O2), and Artemisiaherba alba (O3) against drug-resistant biofilm-positive strains of P. aeruginosa and S. aureus was determined using the standard micro-broth dilution method of CLSI.23 The extract having an initial concentration of 99000 mg/L for O1, 167000 mg/L for O2 and 33000 mg/L for O3. To examine the bacterial growth curve and inhibition in microtiter plate, inoculations were given from fresh colonies on MHA plates into 10 ml of Luria Bertaini (LB) culture medium. Growth was allowed till the optical density reached 0.1 at 580 nm (OD of 0.1 corresponds to 108 CFU/ml of medium). Subsequently, 2×108 CFU/ml from above was added to 1.5 ml of liquid LB media supplemented with 0, 0.5x, 0.25x, 0.125x and 0.0625x of initial Concentrations of Teucrium polium (O1), Salvia officinalis (O2), and Artemisiaherba alba (O3) extracts. The bacterial growth was determined by measuring optical density after every 2 h. The lowest concentration at which there was no exponential phase after 18-22 h incubation at 37°C at 580nm was considered for antibiofilm experiment at this concentration (MIC) and 0.5xMIC with slight modification.
Effect of Extract on Biofilm Formation
In the inhibition tests, bacteria inoculated in microtiter plates were exposed to MIC and 0.5xMIC. The treated mixtures were incubated for 48 hours at 37 °C. To quantify biofilm inhibition, the methods described in the previous section were employed. Positive control wells contained no extracts, while negative control wells were filled only with Tryptic Soy Broth (TSB). After the incubation, the plates were rinsed gently with 1X phosphate-buffered saline (PBS, pH 7.4) and stained for 30 minutes at room temperature using 100 μl of 0.1% crystal violet solution (Sigma-Aldrich, St. Louis, MO). The crystal violet was then solubilized in 95% ethanol, and any excess dye was removed through washing. Lastly, the biofilm’s optical density was measured at 590 nm (OD590 nm) of the supernatant.22-23 All experiment were performed in triplicate.
Results and Discussions
The FTIR Results and Discussions
The FTIR study of these three plants constantly reveals considerable functional groups that reflect their phytochemical richness. Common outcomes include:
Alkane (C-H) stretches at 2918-2851 cm⁻¹ indicate the presence of saturated hydrocarbons and fatty acids.
Carbonyl (C=O) stretches at 1729 cm⁻¹ represent esters, ketones, and conjugated carbonyls.
Amine and amide groups (N-H) exist with varied frequency depending on hydrogen bonding patterns.
Identify aromatic compounds by C=C stretching vibrations in the 1500-1600 cm⁻¹ range.
Identify the phenolic and alcoholic (O-H) groups. Broad absorption bands at 3000-3500 cm⁻¹.24
FT-IR spectra results
 |
Figure 1: FT-IR spectra of O1 (Teucrium Polium Plant), O2 (Salvia_officinalis Plant), O3 (Artemisia_herba-alba plant) Click here to View Figure |
FTIR spectroscopy detects functional groups, revealing the chemical profiles of Teucrium polium, Salvia officinalis, and Artemisia herba alba solutions. Organic compounds with unique resonance patterns show strong absorption bands in FTIR spectra. Plant extracts commonly have O-H stretching vibrations between 3700-3200 cm⁻¹, which indicate the presence of alcohols and phenols. C-H stretching vibrations in alkanes and aliphatic molecules range from 3000-2850 cm⁻¹. Carbonyl groups (C=O stretch) in compounds including aldehydes, ketones, carboxylic acids, and esters display strong absorption bands around 1750-1650 cm⁻¹, whereas C=C stretching vibrations emerge at 1680-1580 cm⁻¹ in alkenes and aromatic rings. Figure – 1 illustrates one of these examples.
Each plant has distinctive spectrum characteristics that reflect its chemical content. For example, Salvia officinalis extracts often exhibit peaks corresponding to carboxylic acids, alcohols, phenols, esters, ethers, and aldehydes, suggesting a diverse range of biomolecules implicated in the antioxidant capabilities. Teucrium polium, Salvia officinalis, and Artemisia herba alba’s chemical profiles may be distinguished by the presence or absence of certain bands, as well as differences in their strength. These observed functional groupings correspond to recognised phytochemical classes. For example, O-H bonds are found in flavonoids and phenolic acids, C=O bonds are found in numerous terpenoids and flavonoids, and C-H bonds are required for alkaloids, terpenoids, and fatty acids, all of which are widely found in medicinal plants.25
GC- MS Results and Discussions
GC-MS examination of Teucrium polium, Salvia officinalis, and Artemisia herba-alba plants revealed several chemical constituents. Terpenoids, lipid compounds, alcohol compounds, and ketones were among the volatile and non-volatile chemical substances discovered in this investigation at varying concentrations. One may be seen in Tables 1 to 6.
The chemical composition of Teucrium polium is mostly established by GC-MS analysis, which displays many terpenoids. Plant biology makes use of monoterpenes and sesquiterpenes. Teucrium polium’s essential oils include α-pinene, germacrene D, and β-pinene. Compounds including caryophyllene, δ-cadinene, and 3-carene have been used to treat gastrointestinal disorders and inflammation.
The GC-MS analysis of Salvia officinalis (common sage) consistently reveals a diversified monoterpene and diterpene-dominated essential oil composition. Understanding its many traditional and therapeutic use requires a complicated chemical fingerprint. Salvia officinalis essential oils include α-thujone, camphor, 1,8-cineole, and borneol, which have antibacterial, antioxidant, and memory-enhancing properties. This chemical richness is linked to the plant’s culinary and medicinal functions.
The GC-MS analysis of Artemisia herba alba essential oil often yields oxygenated monoterpenes and sesquiterpenes. This comprises key compounds such cis-chrysanthenyl acetate, α-thujone, camphor, chrysanthenone, 1,8-cineole, and trans-dihydroterpineol. Its antibacterial and antifungal characteristics make it ideal for therapeutic use.26
The FTIR and GC-MS analysis reveal that these three plant species contain an extensive range of bioactive compounds with great therapeutic potential. Functional group identification via FTIR reveals the presence of phenolic compounds, flavonoids, and terpenoids, while GC-MS identifies specific chemicals quantitatively. These findings support the plants’ historic use while also emphasising their potential for pharmacological and nutraceutical uses.27-28
Table 1: Volatile organic compounds (VOCS) analysis Sample code: Q1 Teucrium polum
| S.No. | Compounds detected | Results &unit: mg /l |
| 1 | Benzene | 0.5877 |
| 2 | Ethane, 1,1-dichloro- | 0.5797 |
| 3 | p-Isopropylbenzene (o-Cymene) | 0.5007 |
| 4 | Ethylbenzene | 0.403 |
| 5 | Ethylene, 1,2-dichloro-, (Z)- | 0.3915 |
| 6 | o-Xylene | 0.3742 |
| 7 | Benzene, 1,2-dichloro- | 0.0411 |
| 8 | Propane, 1,2,3-trichloro- | 0.0001 |
| 9 | Methane, bromodichloro- | 0.0001 |
| 10 | Methane, dibromo- | 0.0001 |
| 11 | Methane, dibromochloro- | 0.0001 |
| 12 | Methylene chloride | 0.0001 |
| 13 | m-Xylene | 0.0001 |
| 14 | Naphthalene | 0.0001 |
| 15 | Propane, 1,2-dibromo-3-chloro- | 0.0001 |
| 16 | Isopropylbenzene | 0.0001 |
| 17 | Propane, 1,2-dichloro- | 0.0001 |
| 18 | Propane, 1,3-dichloro– | 0.0001 |
| 19 | Propene, 1,1-dichloro- | 0.0001 |
| 20 | p-Xylene | 0.0001 |
| 21 | Sec- butylbenzene | 0.0001 |
| 22 | Trichloroethylene | 0.0001 |
| 23 | Toluene | 0.0001 |
| 24 | trans-1,2-dichloro Ethene | 0.0001 |
| 25 | trans-1,3-Dichloropropene | 0.0001 |
| 26 | Methylene chloride | 0.0001 |
| 27 | Ethene, 1,1-dichloro- | 0.0001 |
| 28 | Hexachlorobutadiene | 0.0001 |
| 29 | Benzene, chloro- | 0.0001 |
| 30 | 2-Chlorotoluene | 0.0001 |
| 31 | 2-Pentanone | 0.0001 |
| 32 | Benzene, 1,2,3-trichloro- | 0.0001 |
| 33 | Benzene, 1,2,4-trichloro- | 0.0001 |
| 34 | Benzene, 1,2,4-trimethyl- | 0.0001 |
| 35 | Benzene, 1,2,5-trimethyl- | 0.0001 |
| 36 | Benzene, 1,3-dichloro- | 0.0001 |
| 37 | Benzene, bromo- | 0.0001 |
| 38 | Benzene, propyl- | 0.0001 |
| 39 | cis-1,3-Dichloro-Propene | 0.0001 |
| 40 | Benzene, propyl- | 0.0001 |
| 41 | Benzene, tert-butyl- | 0.0001 |
| 42 | Bromoform | 0.0001 |
| 43 | Chloroform | 0.0001 |
| 44 | Cyclopentane, methyl- | 0.0001 |
| 45 | Ethane, 1,1,1-trichloro- | 0.0001 |
| 46 | Ethane, 1,1,2-trichloro- | 0.0001 |
| 47 | Ethane, 1,2-dibromo- | 0.0001 |
| 48 | Tetrachloroethylene | 0.0001 |
Table 2: Semi Volatile Organic Compounds (SVOCs) Analysis Report Q1 – Teucrium polum
| S. No. | Compounds | Concentration (mg/l) |
| 1 | Benzo[a]pyrene | 0.0271 |
| 2 | Benzo[k]fluoranthene | 0.0135 |
| 3 | Dimethyl phthalate | 0.0102 |
| 4 | Di-n-octyl phthalate | 0.0099 |
| 5 | Naphthalene, 2-methyl- | 0.0093 |
| 6 | Diethyl Phthalate | 0.0083 |
| 7 | Anthracene | 0.0049 |
| 8 | 4-Nitroaniline | 0.0044 |
| 9 | Azobenzene | 0.0012 |
| 10 | 2-Nitroaniline | 0.0015 |
| 11 | Carbazole | 0.0013 |
| 12 | Phenol, 2-nitro- | 0.0014 |
| 13 | Dibutyl phthalate | 0.0010 |
| 14 | 2,4-Dinitrotoluene | 0.0010 |
| 15 | Isophorone | 0.0008 |
| 16 | Phenol, 4-methyl (p-Cresol) | 0.0007 |
| 17 | Phenanthrene | 0.0007 |
| 18 | Naphthalene | 0.0006 |
| 19 | Benzene, 1,2-dichloro- | 0.0003 |
| 20 | Benzene, 1,4-dichloro- | 0.0003 |
| 21 | Bis(2-ethylhexyl) phthalate | 0.0003 |
| Undetectable Compounds (<0.0001 mg/l) | < 0.0001 | |
| 22 | 1,1′-Biphenyl, 2-methyl- | < 0.0001 |
| 23 | 2,6-Dinitrotoluene | < 0.0001 |
| 24 | 3-Nitroaniline | < 0.0001 |
| 25 | 4-Bromophenyl ether | < 0.0001 |
| 26 | 4-Chloroaniline | < 0.0001 |
| 27 | Acenaphthene | < 0.0001 |
| 28 | Acenaphthylene | < 0.0001 |
| 29 | Benz[a]anthracene | < 0.0001 |
| 30 | Benzene, 1,3,4-trichloro- | < 0.0001 |
| 31 | Benzene, 1,3-dichloro- | < 0.0001 |
| 32 | Benzene, 1-chloro-3-phenoxy- | < 0.0001 |
| 33 | Benzene, hexachloro- | < 0.0001 |
| 34 | Benzene, nitro- | < 0.0001 |
| 35 | Benzo[b]fluoranthene | < 0.0001 |
| 36 | Benzo[ghi]perylene | < 0.0001 |
| 37 | Benzyl butyl phthalate | < 0.0001 |
| 38 | Bis(2-chloroethyl) ether | < 0.0001 |
| 39 | Chrysene | < 0.0001 |
| 40 | Dibenzofuran | < 0.0001 |
| 41 | Ethane, hexachloro- | < 0.0001 |
| 42 | Fluoranthene | < 0.0001 |
| 43 | Fluorene | < 0.0001 |
| 44 | Hexachlorobutadiene | < 0.0001 |
| 45 | Hexachlorocyclopentadiene | < 0.0001 |
| 46 | Indeno[1,2,3-cd]pyrene | < 0.0001 |
| 47 | Methane, bis(2-chloroethoxy)- | < 0.0001 |
| 48 | Naphthalene, 2-chloro- | < 0.0001 |
| 49 | Pentacene | < 0.0001 |
| 50 | Phenol | < 0.0001 |
| 51 | Phenol, 2,4,5-trichloro- | < 0.0001 |
| 52 | Phenol, 2,4,6-trichloro- | < 0.0001 |
| 53 | Phenol, 2,4-dichloro- | < 0.0001 |
| 54 | Phenol, 2,4-dimethyl- | < 0.0001 |
| 55 | Phenol, 2,4-dinitro- | < 0.0001 |
| 56 | Phenol, 2-chloro- | < 0.0001 |
| 57 | Phenol, 2-methyl- | < 0.0001 |
| 58 | Phenol, 2-methyl, 4,6-dinitro- | < 0.0001 |
| 59 | Phenol, 4-chloro-3-methyl- | < 0.0001 |
| 60 | Phenol, pentachloro- | < 0.0001 |
| 61 | Pyrene | < 0.0001 |
| 62 | Tetradecane | < 0.0001 |
Table 3: Volatile Organic Compounds (VOCs) Analysis Report Q2 – Salvia officinalis
| S.No. | Compounds detected | Result (mg/l) |
| 1 | Benzene | 0.5867 |
| 2 | Ethane, 1,1-dichloro- | 0.5787 |
| 3 | p-Isopropylbenzene (o-Cymene) | 0.5015 |
| 4 | Benzene, tert-butyl- | 0.4739 |
| 5 | Benzene, propyl- | 0.4294 |
| 6 | Ethylbenzene | 0.4034 |
| 7 | o-Xylene | 0.3748 |
| 8 | Propane, 1,2,3-trichloro- | 0.2658 |
| 9 | 2-Chlorotoluene | 0.2445 |
| 10 | Benzene, 1,2,5-trimethyl- | 0.1202 |
| 11 | Benzene, 1,2-dichloro- | 0.0403 |
| 12 | Benzene, chloro- | 0.0219 |
| 13 | Propane, 1,2-dichloro- | <0.0001 |
| 14 | Propane, 1,3-dichloro- | <0.0001 |
| 15 | Propene, 1,1-dichloro- | <0.0001 |
| 16 | p-Xylene | <0.0001 |
| 17 | Sec- butylbenzene | <0.0001 |
| 18 | Tetrachloroethylene | <0.0001 |
| 19 | Toluene | <0.0001 |
| 20 | trans-1,2-dichloro Ethene | <0.0001 |
| 21 | trans-1,3-Dichloropropene | <0.0001 |
| 22 | Trichloroethylene | <0.0001 |
| 23 | Ethene, 1,1-dichloro- | <0.0001 |
| 24 | cis-1,3-Dichloro-Propene | <0.0001 |
| 25 | 2-Pentanone | <0.0001 |
| 26 | Benzene, 1,2,3-trichloro- | <0.0001 |
| 27 | Benzene, 1,2,4-trichloro- | <0.0001 |
| 28 | Benzene, 1,2,4-trimethyl- | <0.0001 |
| 29 | Benzene, 1,3-dichloro- | <0.0001 |
| 30 | Benzene, bromo- | <0.0001 |
| 31 | Benzene, n-butyl- | <0.0001 |
| 32 | Bromoform | <0.0001 |
| 33 | Chloroform | <0.0001 |
| 34 | Cyclopentane, methyl- | <0.0001 |
| 35 | Ethane, 1,1,1-trichloro- | <0.0001 |
| 36 | Ethane, 1,1,2-trichloro- | <0.0001 |
| 37 | Ethane, 1,2-dibromo- | <0.0001 |
| 38 | Ethylene, 1,2-dichloro-, (Z)- | <0.0001 |
| 39 | Hexachlorobutadiene | <0.0001 |
| 40 | Isopropylbenzene | <0.0001 |
| 41 | Methane, bromochloro- | <0.0001 |
| 42 | Methane, bromodichloro- | <0.0001 |
| 43 | Methane, dibromo- | <0.0001 |
| 44 | Methane, dibromochloro- | <0.0001 |
| 45 | Methylene chloride | <0.0001 |
| 46 | m-Xylene | <0.0001 |
| 47 | Naphthalene | <0.0001 |
| 48 | Propane, 1,2-dibromo-3-chloro- | <0.0001 |
Table 4: Semi Volatile Organic Compounds (SVOCs) Analysis Report Q2 – Salvia officinalis
| S. No. | Compounds | Concentration (mg/l) |
| 1 | Di-n-octyl phthalate | 0.0338 |
| 2 | Dimethyl phthalate | 0.0046 |
| 3 | 4-Nitroaniline | 0.0021 |
| 4 | Carbazole | 0.0018 |
| 5 | Fluoranthene | 0.0007 |
| 6 | 2-Nitroaniline | 0.0006 |
| 7 | Pyrene | 0.0006 |
| 8 | Naphthalene | 0.0005 |
| 9 | Bis(2-ethylhexyl) phthalate | 0.0005 |
| 10 | Diethyl Phthalate | 0.0004 |
| 11 | Phenol, 2-nitro- | 0.0003 |
| 12 | Dibutyl phthalate | 0.0003 |
| 13 | Benzene, 1,2-dichloro- | 0.0002 |
| 14 | Benzene, 1,4-dichloro- | 0.0002 |
| 15 | Isophorone | 0.0002 |
| 16 | Benzo[k]fluoranthene | <0.0001 |
| 17 | Benzene, 1,3-dichloro- | <0.0001 |
| 18 | Acenaphthylene | <0.0001 |
| 19 | Anthracene | <0.0001 |
| 20 | Azobenzene | <0.0001 |
| 21 | Dibenzofuran | <0.0001 |
| 22 | Benz[a]anthracene | <0.0001 |
| 23 | Chrysene | <0.0001 |
| 24 | Benzene, 1,3,4-trichloro- | <0.0001 |
| 25 | Ethane, hexachloro- | <0.0001 |
| 26 | Benzo[ghi]perylene | <0.0001 |
| 27 | Bis(2-chloroethyl) ether | <0.0001 |
| 28 | Benzene, 1-chloro-3-phenoxy- | <0.0001 |
| 29 | Benzene, hexachloro- | <0.0001 |
| 30 | Benzene, nitro- | <0.0001 |
| 31 | Benzyl butyl phthalate | <0.0001 |
| 32 | Benzo[a]pyrene | <0.0001 |
| 33 | Acenaphtherne | <0.0001 |
| 34 | Benzo[b]fluoranthene | <0.0001 |
| 35 | Fluorene | <0.0001 |
| 36 | 4-Chloroaniline | <0.0001 |
| 37 | Phenol, 2,4-dinitro- | <0.0001 |
| 38 | Hexachlorocyclopentadiene | <0.0001 |
| 39 | Indeno[1,2,3-cd]pyrene | <0.0001 |
| 40 | Methane, bis(2-chloroethoxy)- | <0.0001 |
| 41 | Naphthalene, 2-chloro- | <0.0001 |
| 42 | Naphthalene, 2-methyl- | <0.0001 |
| 43 | Pentacene | <0.0001 |
| 44 | Phenanthrene | <0.0001 |
| 45 | Phenol, 2,4,5-trichloro- | <0.0001 |
| 46 | Phenol, 2,4,6-trichloro- | <0.0001 |
| 47 | Phenol, 2,4-dichloro- | <0.0001 |
| 48 | Phenol, 2,4-dimethyl- | <0.0001 |
| 49 | Phenol, 2-chloro- | <0.0001 |
| 50 | Hexachlorobutadiene | <0.0001 |
| 51 | Phenol, 2-methyl- | <0.0001 |
| 52 | Phenol, 2-methyl, 4,6-dinitro- | <0.0001 |
| 53 | Phenol, 4-chloro-3-methyl- | <0.0001 |
| 54 | Phenol, 4-methyl (p-Cresol) | <0.0001 |
| 55 | Phenol, pentachloro- | <0.0001 |
| 56 | Tetradecane | <0.0001 |
| 57 | Phenol | <0.0001 |
| 58 | 1,1′-Biphenyl, 2-methyl- | <0.0001 |
| 59 | 2,4-Dinitrotoluene | <0.0001 |
| 60 | 2,6-Dinitrotoluene | <0.0001 |
| 61 | 3-Nitroaniline | <0.0001 |
| 62 | 4-Bromophenyl ether | <0.0001 |
Table 5: Volatile Organic Compounds (VOCs) Analysis Q3 – Artemisiaherba alba
| S. No. | Compounds detected | Result(mg/l) |
| 1 | Benzene | 0.5841 mg/l |
| 2 | Isopropylbenzene | 0.4666 mg/l |
| 3 | Ethylbenzene | 0.4007 mg/l |
| 4 | o-Xylene | 0.3726 mg/l |
| 5 | Benzene, 1,2,5-trimethyl- | 0.121 mg/l |
| 6 | Benzene, 1,2-dichloro- | 0.0367 mg/l |
| 7 | Ethene, 1,1-dichloro- | 0.0001 mg/l |
| 8 | cis-1,3-Dichloro-Propene | 0.0001 mg/l |
| 9 | 2-Chlorotoluene | 0.0001 mg/l |
| 10 | 2-Pentanone | 0.0001 mg/l |
| 11 | Benzene, 1,2,3-trichloro- | 0.0001 mg/l |
| 12 | Benzene, 1,2,4-trichloro- | 0.0001 mg/l |
| 13 | Benzene, 1,2,4-trimethyl- | 0.0001 mg/l |
| 14 | Benzene, 1,3-dichloro- | 0.0001 mg/l |
| 15 | Benzene, bromo- | 0.0001 mg/l |
| 16 | Benzene, chloro- | 0.0001 mg/l |
| 17 | Benzene, n-butyl- | 0.0001 mg/l |
| 18 | Benzene, propyl- | 0.0001 mg/l |
| 19 | Benzene, tert-butyl- | 0.0001 mg/l |
| 20 | Bromoform | 0.0001 mg/l |
| 21 | Chloroform | 0.0001 mg/l |
| 22 | Cyclopentane, methyl- | 0.0001 mg/l |
| 23 | Ethane, 1,1,1-trichloro- | 0.0001 mg/l |
| 24 | Ethane, 1,1,2-trichloro- | 0.0001 mg/l |
| 25 | Ethane, 1,1-dichloro- | 0.0001 mg/l |
| 26 | Ethane, 1,2-dibromo- | 0.0001 mg/l |
| 27 | Ethylene, 1,2-dichloro-, (Z)- | 0.0001 mg/l |
| 28 | Hexachlorobutadiene | 0.0001 mg/l |
| 29 | Methane, bromochloro- | 0.0001 mg/l |
| 30 | Methane, bromodichloro- | 0.0001 mg/l |
| 31 | Methane, dibromo- | 0.0001 mg/l |
| 32 | Methane, dibromochloro- | 0.0001 mg/l |
| 33 | Methylene chloride | 0.0001 mg/l |
| 34 | m-Xylene | 0.0001 mg/l |
| 35 | Naphthalene | 0.0001 mg/l |
| 36 | p-Isopropylbenzene (o-Cymene) | 0.0001 mg/l |
| 37 | Propane, 1,2,3-trichloro- | 0.0001 mg/l |
| 38 | Propane, 1,2-dibromo-3-chloro- | 0.0001 mg/l |
| 39 | Propane, 1,2-dichloro- | 0.0001 mg/l |
| 40 | Propane, 1,3-dichloro- | 0.0001 mg/l |
| 41 | Propene, 1,1-dichloro- | 0.0001 mg/l |
| 42 | p-Xylene | 0.0001 mg/l |
| 43 | Sec-butylbenzene | 0.0001 mg/l |
| 44 | Tetrachloroethylene | 0.0001 mg/l |
| 45 | Toluene | 0.0001 mg/l |
| 46 | trans-1,2-dichloro Ethene | 0.0001 mg/l |
| 47 | trans-1,3-Dichloropropene | 0.0001 mg/l |
| 48 | Trichloroethylene | 0.0001 mg/l |
Table 6: Semi volatile Organic Compounds (SVOCs) Analysis Report Sample Code: Q3 – Artemisia Erba alba.
| S. No. | Compounds detected | Result (mg/l) |
| 1 | Nitroaniline | 0.0402 |
| 2 | Naphthalene,2-methyl- | 0.0400 |
| 3 | Fluorene | 0.0275 |
| 4 | Di-n-octyl phthalate | 0.0225 |
| 5 | Diethyl Phthalate | 0.0216 |
| 6 | Isophorone | 0.0111 |
| 7 | Phenol, 2-nitro- | 0.0108 |
| 8 | Acenaphthylene | 0.0088 |
| 9 | Dibutyl phthalate | 0.0050 |
| 10 | Azobenzen | < 0.0044 |
| 11 | Phenol, 4methyl (p-Cresol) | < 0.0027 |
| 12 | Phenol | 0.0022 |
| 13 | chloroaniline | 0.0022 |
| 14 | Dimethyl phthalate | 0.0017 |
| 15 | Phenol, 2,4-dimethyle | 0.0017 |
| 16 | Benzene, nitro- | 0.0013 |
| 17 | Bis(2-ethylhexyl) phthalate | 0.0013 |
| 18 | Naphthalene, 2-chloro- | 0.0013 |
| 19 | 2,4-Dinitrotoluene | 0.0012 |
| 20 | Anthracene | 0.0004 |
| 21 | Bis(2-chloroethyle) ether | 0.0003 |
| 22 | Naphthalene | 0.0003 |
| 23 | Benzyl butyl phthalate | 0.0001 |
| 24 | 1,1′-Biphenyl, 2-methyl- | < 0.0001 |
| 25 | 2,6-Dinitrotoluene | < 0.0001 |
| 26 | 2-Nitroaniline | < 0.0001 |
| 27 | 3- Nitroaniline | < 0.0001 |
| 28 | 4-Bromophenyl ether | < 0.0001 |
| 29 | Acenaphthene | < 0.0001 |
| 30 | Acenaphthylene | < 0.0001 |
| 31 | Benz[a]anthrance | < 0.0001 |
| 32 | Benzene, 1,2-dichloro- | < 0.0001 |
| 33 | Benzene, 1,3,4-trichloro- | < 0.0001 |
| 34 | Benzene, 1,3-dichloro- | < 0.0001 |
| 35 | Benzene, 1,4-dichloro- | < 0.0001 |
| 36 | Benzene, 1-chloro-3-phenoxy | < 0.0001 |
| 37 | Benzene, hexachloro- | < 0.0001 |
| 38 | Benzo[a] pyrene | < 0.0001 |
| 39 | Benzo[b] fluoranthene | < 0.0001 |
| 40 | Benzo[ghi] perylene | < 0.0001 |
| 41 | Benzo[k] fluoranthene | < 0.0001 |
| 42 | Carbazole | < 0.0001 |
| 43 | chrysene | < 0.0001 |
| 44 | Dibenzofuran | < 0.0001 |
| 45 | Ethane, hexachloro | < 0.0001 |
| 46 | fluoranthene | < 0.0001 |
| 47 | Hexachlorobutadiene | < 0.0001 |
| 48 | Hexachlorocyclopentadiene | < 0.0001 |
| 49 | Indeno[1m2,3-cd]pyrene | < 0.0001 |
| 50 | Methane, bis(2-chloroethoxy)- | < 0.0001 |
| 51 | Naphthalene,2-methyl- | < 0.0001 |
| 52 | Pentacene | < 0.0001 |
| 53 | phenanthrene | < 0.0001 |
| 54 | Phenol, 2,4,5-trichloro- | < 0.0001 |
| 55 | Phenol, 2,4,6-trichloro- | < 0.0001 |
| 56 | Phenol, 2,4-dichloro- | < 0.0001 |
| 57 | Phenol, 2,4-dinitro- | < 0.0001 |
| 58 | Phenol, 2-chloro- | < 0.0001 |
| 59 | Phenol, 2-methyl- | < 0.0001 |
| 60 | Phenol, 2-methyl, 4,6-dinitro- | < 0.0001 |
| 61 | Phenol, 4-chloro-3-methyl- | < 0.0001 |
| 62 | Phenol, pentachloro | < 0.0001 |
| 63 | Pyrene | < 0.0001 |
| 64 | Tetradecane | < 0.0001 |
Bioactivity result and discussion
Antibacterial effect
The antibacterial efficacy of Teucrium polium (O1), Salvia officinalis (O2), and Artemisia herba-alba (O3) was assessed using a time-dependent growth inhibition experiment. The growth curves of Pseudomonas aeruginosa and Staphylococcus aureus subjected to O1, O2, and O3 demonstrated that these extracts possess the capability to limit bacterial growth and reproduction. Table – 7 and Figure-2 illustrates the proliferation of P. aeruginosa in LB broth inoculated with 10^8 CFU/mL, subjected to 0.5x, 0.25x, 0.125x, and 0.0625x concentrations of O1 (99,000 mg/L), O2 (167,000 mg/L), and O3 (33,000 mg/L). All concentrations exhibited a growth delay in the tested strains. The growth curves displayed three stages: lag, exponential, and stabilisation; however, decline phases were absent, as OD580 values included both viable and non-viable bacteria. In the absence of extract, P. aeruginosa swiftly entered the exponential growth phase; however, treatment with 0.5x O1, 0.125x O2, and 0.0625x O3 postponed growth for as long as 4 hours, demonstrating significant bacteriostatic action at lower doses and bactericidal effects at elevated concentrations. Likewise, S. aureus exhibited three phases, characterised by rapid exponential development in the control group. Treatments with 0.5x O1, 0.25x O2, and 0.25x O3 successfully suppressed growth for 3–6 hours, whereas lower concentrations initially delayed growth before transitioning to exponential growth, indicating substantial bacteriostatic activity at reduced doses and bactericidal activity at elevated doses [Table – 8] and (Figure -2).
 |
Table 7: Pseudomonas aeruginosa growth time assay with MIC analysis. Click here to View Table |
 |
Table 8: S aureus growth time assay with MIC analysis. Click here to View Table |
 |
Figure 2: Pseudomonas aeruginosa and S aureus growth curve over time when exposed to various concentrations with MIC. Click here to View Figure |
Antibiofilm Effect
The potential of herbal extract of Teucrium polium (O1), Salvia officinalis (O2), and Artemisia herba-alba (O3) to inhibit the biofilm formation at MIC and 0.5xMIC from the antimicrobial growth curve. The results of the biofilm inhibition study are presented in Figure. 3. Concentration-dependent effect of O1, O2 and O3 was observed against test bacteria and maximum inhibition was 87% by O1 against P aeruginosa and 74% for S aureus at MIC. Whereas almost similar inhibition was recorded at 0.5xMIC of O1. O2 extract shows 88% for both at MIC and 63% and 60% reduction at 0.5xMIC. Whereas, there was a difference of 10% and 22% at MIC and 0.5xMIC of O3 against P aeruginosa and S aureus. Can be seen in the Table -9.
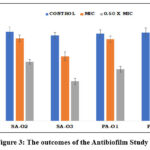 |
Figure 3: The outcomes of the Antibiofilm Study Click here to View Figure |
Table 9: Pseudomonas aeruginosa, and S aureus Biofilm Inhibition assay in microtiter plate.
| NO GRWOTH BY MIC AT CONCENTRATION | ||||||
| PS | SA | |||||
| C1 | 0.5x MIC | 0.5x MIC | ||||
| C2 | 0.125x MIC | 0.25x MIC | ||||
| C3 | 0.0625x MIC | 0.25x MIC | ||||
| Percent (%) reduction of biofilm PRODUCTION | ||||||
| Mean | Mean | Mean | SD | SD | SD | |
| CONTROL | MIC | 0.50 X MIC | CONTROL | MIC | 0.50 X MIC | |
| SA-O1 | 91 | 74 | 54 | 3.21 | 2.95 | 2.9 |
| SA-O2 | 95 | 88 | 63 | 5.21 | 3.45 | 2.56 |
| SA-O3 | 91 | 69 | 42 | 3.14 | 4.58 | 2.99 |
| PA-O1 | 93 | 87 | 55 | 4.12 | 3.54 | 3.01 |
| PA-O2 | 94 | 88 | 60 | 5.01 | 2.98 | 1.98 |
| PA-O3 | 96 | 79 | 64 | 3.54 | 4.32 | 3.74 |
PA: Pseudomonas aeruginosa
SA: S aureus
Conclusions
The FTIR and GC-MS analyses show that Salvia officinalis, Artemisia herba alba, and Teucrium polium contain a wide variety of bioactive chemicals with high medicinal potential. Functional group identification by FTIR indicates the existence of phenolic compounds, flavonoids, and terpenoids, while GC-MS identifies individual chemicals quantitatively.
P. aeruginosa immediately underwent exponential growth without extract, whereas 0.5x O1, 0.125x O2, and 0.0625x O3 reduced growth for 4 hours, exhibiting bacteriostatic and bactericidal effects at lower dosages and greater concentrations. S. aureus has three phases, including rapid exponential growth in controls. 0.5x O1, 0.25x O2, and 0.25x O3 inhibited growth for 3–6 hours, although lower doses delayed exponential growth, demonstrating bacteriostatic and bactericidal effects. The concentration-dependent biofilm inhibition investigation indicated that O1 inhibited P. aeruginosa 87% and S. aureus 74% at the MIC. Similar suppression was seen at 0.5x O1 MIC. O2 extract reduces by 88% at MIC, 63% at 0.5xMIC, and 60% at The MIC and 0.5xMIC of O3 varied by 10% and 22% against P. aeruginosa and S. aureus, respectively.
This study reveals the bioactive compounds, molecular processes, and health benefits of Salvia officinalis, Artemisia herba alba, and Teucrium polium. The finding has broadened the understanding of the plant’s bioactivities and medicinal applications, especially for the treatment of intestinal diseases.
Acknowledgement
It is with great pleasure that the authors would like to express their gratitude to the Department of Chemistry, Department of Medical Microbiology and Molecular Microbiology and Infectious Diseases Research Unit, University of Tabuk, K. S. A.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Yassine, Kalbaza Ahmed, Houari, H., Eddine, Rezgui Houssam, Younes, Hamouta, Mokhtar, Benchohra, and Karim, Amara. 2023. “Evaluation of Wound Healing Activity of Teucrium polium on Excisional Wounds in Wistar Rats”. Agricultural Science Digest, volume 44 issue 4-2024 : 745-750,. https://doi.org/10.18805/ag.df-520
CrossRef - Subhan, Ghani, Iqbal, Zafar, Mateen, Abdul, Akbar, Maaz, and Ullah, Faseeh. “Phytosociological Analysis and Vegetation Structure in Tehsil Timergara, District Dir Lower, Pakistan: A Comprehensive Study of Five Distinct Plant Communities”. Ecology and Evolutionary Biology. 2024.Vol- 9,issue-3 https://doi.org/10.11648/j.eeb.20240903.11
CrossRef - Pulat, Esra, YILDIZBA, Avni, and Yaman, Barbaros. “Wood Anatomical Characteristics of Felty Germander (Teucrium polium L. subsp. polium) in Two Different Habitats”. Bartn University. Journal of Bartin Faculty of Forestry. 2024, Volume: 26 Issue: 1, 110 – 118 https://doi.org/10.24011/barofd.1379079
CrossRef - Ghassa, Ali, Ismail, Mousa, and Alani, Leen Alabd Alrazak. “Teucrium polium: new toxic effects added to the literature: a case report”. Annals of Medicine and Surgery. 2024 ;86 (5):3005–3008. https://doi.org/10.1097/MS9.0000000000001760
CrossRef - Al Naemi, Hamda A., Al asmar, Reem Moath, and Al-Ghanim, Kaltham. “Alcoholic extracts of Teucrium polium exhibit remarkable anti-inflammatory activity: In vivo study”. Biomol Biomed. 2024 Feb 1;24(1):82–88. https://doi.org/10.17305/bb.2023.9239
CrossRef - Soussa, Amel, Dridi, Amina, Otmani, Ines, Daas, Hiba, and Kerkoub, N. “Evaluation of Biological Activity of Essential Oil from Teucrium polium L.”. Agricultural Science Digest. 2024. https://doi.org/10.18805/ag.df-625
CrossRef - Maodaa, S.N., Al-quraishy, S., Abdel-Gaber, R., Alatawi, A., Alawwad, S., Alhomoud, D., and Al-Shaebi, E. “Protective Effects of Teucrium polium Leaves Ethanolic Extract against Eimeria papillata-Induced Behavioral Changes in Mice”. Indian Journal of Animal Research. vol 58 (10). 2024: 1722-1732, https://doi.org/10.18805/ijar.bf-1802
CrossRef - Aljaber, A. Y., Alatawi, Z., Asiri, M., Alomrani, Naser A., Al-Aoh, Hatem A., and Mustafa, Syed Khalid.. “A Study of Chemical Composition and Bioactivity of Folk Medicinal Plant (Teucrium polium)”. Asian Journal of Chemical Sciences. 2024. https://doi.org/10.9734/ajocs/2024/v14i6341
CrossRef - Assaggaf, H., et al… “Chemical Analysis and Investigation of Biological Effects of Salvia officinalis Essential Oils at Three Phenological Stages”. Molecules. 2022. https://doi.org/10.3390/molecules27165157
CrossRef - Akacha, Boutheina Ben, Hsouna, Anis Ben, Mekini, Ivana Generali, Belgacem, Amni Ben, Saad, Rania Ben, Mnif, W., Kaniov, M., and Garzoli, S. “Salvia officinalis L. and Salvia sclarea Essential Oils: Chemical Composition, Biological Activities and Preservative Effects against Listeria monocytogenes Inoculated into Minced Beef Meat”. Plants. 2023. https://doi.org/10.3390/plants12193385
CrossRef - Ovidi, E., Masci, V. Laghezza, Zambelli, Marta, Tiezzi, A., Vitalini, S., and Garzoli, S. “Laurus nobilis, Salvia sclarea and Salvia officinalis Essential Oils and Hydrolates: Evaluation of Liquid and Vapor Phase Chemical Composition and Biological Activities”. Plants. 2021. https://doi.org/10.3390/plants10040707
CrossRef - Kaniov, M., et al. “Chemical composition and biological activity of Salvia officinalis essential oil”. Acta Horticulturae et Regiotecturae. 2021. https://doi.org/10.2478/ahr-2021-0028
CrossRef - Soltanbeigi, Amir, Yldz, Mustafa, Draman, Harun, Terzi, Hakan, Sakartepe, Erhan, and Yldz, Emel. “Growth responses and essential oil profile of Salvia officinalis L. Influenced by water deficit and various nutrient sources in the greenhouse”. Elsevier BV. 2021. https://doi.org/10.1016/j.sjbs.2021.08.034
CrossRef - H. Mohsen and F. Ali.Study of genetic polymorphism of Artemisia herba-alba from Tunisia using ISSR markers, African J. of Biotechnol., 2008. 7(1), 44-50. DOI:10.4314/AJB.V7I1.58313
- T. Efferth. Antiplasmodial and antitumor activity of artemisinin-from bench to besides, Planta Med.; 2007. .73(4):299-309. doi: 10.1055/s-2007-967138
CrossRef - H. Wong and G. D. Brown, Germacranolides from Artemisia myriantha and their conformation, Phytochemistry, 2002 ;59 (5):529-36. doi: 10.1016/s0031-9422(01) 00479-4.
CrossRef - J. H. Kwak, W. Y. Jang, O. P. Zee and R. K. Lee. A new Coumarin-Monoterpene ether from Artemisia keiskeana, Planta Med., 1997. 63, 474-476. DOI:10.1055/s-2006-957741
CrossRef - O. Bergendorff and Sterner O. Spasmolytic flavonols from Artemisia abrotanum, Planta Med.1995. 61, 370-371. doi: 10.1055/s-2006-958106.
CrossRef - Elaloui, M., Ghazghazi, H., Ennajah, A., Manaa, S., Guezmir, W., Karray, N., and Laamouri, A. “Phenolic profile, antioxidant capacity of five Ziziphus spina-christi (L.) Willd provenances and their allelopathic effects on Trigonella foenum-graecum L. and Lens culinaris L. seeds”. Natural Product Research. 2017. 48(3):1600-1612. DOI:10.15835/nbha48312025
CrossRef - Rialdi, Azzahra Putri, Prangdimurti, E., and Saraswati, S. “Effect of Different Solvent on The Antioxidant Capacity of Bidara Leaves Extract (Ziziphus Spina-Christi)”. Devotion Journal of Community Service. 2023. Vol. 4 No. 6 . https://doi.org/10.59188/devotion.v4i6.483
CrossRef - Hussain, M. A., Nathar, V. N., and Mushtaq, Roohi. “In vitro plant regeneration and metabolite profiling of an aromatic medicinal plant Ruta graveolens L. by using GC-MS”. Current Botany. 2022. https://doi.org/10.25081/cb.2022.v13.6856
CrossRef - Zubair, M.; Fatima, F.; Rahman, S.; Alrasheed, T.; Alatawy, R.;Mesaik, M.A. Disruption of Biofilm Formation by Dead Sea Soil Extracts: A Novel Approach Against Diabetic Foot Wound Isolates. Microbiol. Res.2024(a), 15, 2535–2554.
CrossRef - Zubair M, Husain FM, Al-Amri M, Hasan I, Hassan I, Albalawi T, Fatima F, Khan A, Arshad M, Alam P, Ahmad N, Alatawy R, Begum S, Mir R, Alshadfan H, Ansari AA, Al-Anazi ABAA. In vitro inhibition of biofilm and virulence factor production in azole-resistant strains of Candida albicans isolated from diabetic foot by Artemisia vulgaris stabilized tin (IV) oxide nanoparticles. Front Cell Infect Microbiol. 2024 (b) 25;13:1322778.
CrossRef
- M. Bouafia, F. Amamou, M. Gherib, M. Benaissa, R. Azzi, and S. Nemmiche, “Ethnobotanical and ethnomedicinal analysis of wild medicinal plants traditionally used in nama, southwest algeria,” Vegetos- An International Journal of Plant Research, 2021 ;34(3):654-662. https://doi.org/10.1007/s42535-021-00229-7
CrossRef - K. Belhouala and B. Benarba, “Medicinal plants used by traditional healers in algeria: A multiregional ethnobotanical study,” Frontiers Media, 2021. https://doi.org/10.3389/fphar.2021.760492
CrossRef - T. Benali et al., “Phytochemical analysis and study of antioxidant, anticandidal, and antibacterial activities of teucrium polium subsp. Polium and micromeria graeca (lamiaceae) essential oils from northern morocco,” Hindawi Publishing Corporation, 2021. https://doi.org/10.1155/2021/6641720
CrossRef - G. Catinella, N. Badalamenti, V. Ilardi, S. Rosselli, L. D. Martino, and M. Bruno, “The essential oil compositions of three teucrium taxa growing wild in sicily: HCA and PCA analyses,” Molecules, 2021. 27(5), 1580;https://doi.org/10.3390/molecules27051580
CrossRef - H. Houti et al., “Moroccan endemic artemisia herba-alba essential oil: GC-MS analysis and antibacterial and antifungal investigation,” Multidisciplinary Digital Publishing Institute, 2023. https://doi.org/10.3390/separations10010059
CrossRef
Second Review by: Dr. K. Koteswara Rao
Final Approval by: Dr. Murat Hatipoglu

















