Electro Spun Nanofibers for Biomedical Use: Fabrication Approaches and Functional Insights
1Department of Pharmaceutics, School of Pharmaceutical Sciences, Shri Guru Ram Rai University, Dehradun, Uttarakhand, India.
2Department of Pharmacology, School of Pharmaceutical Sciences, Shri Guru Ram Rai University, Dehradun, Uttarakhand, India.
3Department of Pharmacognosy, School of Pharmaceutical Sciences, Shri Guru Ram Rai University, Dehradun, Uttarakhand, India.
Corresponding Author E-mail:mittal.heena22@gmail.com
Download this article as:
ABSTRACT:Electrospinning is a versatile and efficient method for producing nanofibers that exhibit unique characteristics suitable for various medical uses. Electrospun nanofibers (EN) fabricated using biopolymers have revolutionized modern platforms for drug delivery due to their controlled drug release kinetics and structural mimicry of extracellular matrices. By understanding the intricacies of fabrication and functionality, we can better harness the potential of electrospun nanofibers in the biomedical field. This review aims to provide a comprehensive overview of electrospun nanofibers with particular emphasis on their fabrication approaches, use of polymers, and functional insights in the biomedical domain. Various fabrication approaches, including solution electrospinning, melt electrospinning, coaxial and triaxial electrospinning, and emulsion electrospinning, have been developed to optimize fiber morphology and impart multifunctionality. The choice of polymer—natural, synthetic, or blended—further enables tailoring of mechanical strength, biodegradability, and bioactivity. Functional modifications, such as surface coating, incorporation of bioactive molecules, and nanoparticle embedding, extend the scope of nanofibers toward drug delivery, wound healing, tissue engineering, and biosensing. In addition, researchers are advancing the design of multifunctional hybrid structures by integrating electrospinning with complementary fabrication techniques such as 3D printing, green electrospinning, and layer-by-layer assembly. These approaches allow for the creation of highly customized scaffolds with tailored properties and enhanced functionality. Overall, electrospun nanofibers represent a promising platform that bridges materials science and biomedical engineering, offering innovative solutions for next-generation therapeutics and regenerative medicine.
KEYWORDS:Biosensing; Electrospinning; Nanofibres; Polymers; Tissue engineering
Introduction
The last decade has witnessed a surge in advanced delivery of drugs as researchers strive to enhance therapeutic efficiency while reducing side effects. Nanotechnology is becoming one of the most revolutionary areas of contemporary biomedical research due to the ongoing hunt for cutting-edge biomaterials that can solve the expanding problems in healthcare. Using electrospun nanofibers made of naturally occurring biopolymers is one of the most promising approaches1,2. By leveraging the qualities of natural polymers nanofibers like biocompatibility, biodegradability, and their capacity to replicate the extracellular matrix-modern are emerging as a powerful tool in the delivery of drugs3. Although, concept of electrospinning can be traced back over a century, its full potential was only recognized in recent decades. The first patents related to electrospinning appeared in the early 20th century, but it wasn’t until the 1990s—with advancements in nanotechnology and microscopy—that interest in the process surged4. Today, electrospun nanofibres offer tunable physical, chemical, and biological characteristics tailored for specific applications5,6 (Fig. 1).
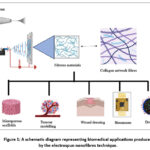 |
Figure 1: A schematic diagram representing biomedical applications produced by the electrospun nanofibres technique. |
Drug delivery systems have evolved to offer controlled, sustained, and targeted release to improve therapeutic outcomes. Electrospun nanofibers—fibers with diameters in the nanometer scale—present an innovative approach due to their porous structure and ease of fabrication7,8 (Fig. 2). Their use in localized and systemic drug delivery has gained substantial attention in recent years. Electrospun nanofibers offer a solution by providing high surface area, customizable porosity, and flexible drug loading strategies9.
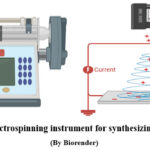 |
Figure 2: Electrospinning instrument for synthesizing nanofibers Click here to View Figure |
Electrospinning has emerged as a bridge between polymer science and biomedical engineering by providing a flexible platform to process diverse polymers—natural, synthetic, or their composites—into fibrous scaffolds. While synthetic polymers, such as polycaprolactone (PCL), polylactic acid (PLA), and poly(lactic-co-glycolic acid) (PLGA), offer controlled degradation and mechanical strength, natural polymers, including collagen, gelatin, chitosan, and silk fibroin, offer intrinsic biocompatibility and bioactivity. When made from biocompatible polymers, these fibers not only enhance therapeutic efficiency but also ensure safety and biodegradability10. The unique characteristics of nanofibers, including their porous structure, customizable functionalization, morphology, and ability to incorporate functional molecules, make them especially valuable in areas such as drug delivery, wound healing, tissue engineering, and biosensors. A major advantage of electrospinning lies in its ability to process the use of polymers, and even blends. Moreover, functional agents like proteins, API, or nanoparticles can be directly incorporated into the fibers during the electrospinning process, allowing for controlled and sustained release11,12.
Principle of Electrospun Nanofibers
Electrospinning is a fiber production technique that uses a high-voltage electric field to draw a charged solution of polymer or melt into fine fibers with diameters ranging from nanometers to micrometers. In this process, the solution of polymer is placed in a syringe fitted with a metallic needle, which is connected to a high-voltage power supply. When the electric field overcomes the surface tension of the polymer droplet at the needle tip forms, and a thin charged jet is ejected toward a grounded collector the solvent evaporates (or the melt solidifies), resulting in the deposition of continuous fibers13,14 (Fig 3).
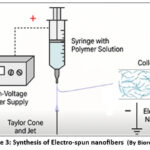 |
Figure 3: Synthesis of Electro-spun nanofibers |
Key Steps in Electrospinning Process
High-Voltage Application: A metallic needle holding the polymer solution is attached to a high-voltage power source, usually 10–30 kV.
Taylor Cone Formation: The polymer droplet’s surface tension is overcome by electrostatic repulsion, creating the Taylor cone, a conical shape.
Jet Ejection: From the cone’s tip, a jet which is charged is released and moves in the direction of the collector.
Jet Thinning and Solidification: The jet’s diameter is reduced to nanoscale as it moves because it expands and the solvent evaporates.
Fiber Deposition: The solidified nanofibers are collected as a non-woven mat on a grounded collector (usually a rotating or static plate)15,16.
Mechanisms for Release of Drug
Drug release from electrospun nanofibers generally occurs through multiple mechanisms, often operating simultaneously. The mechanisms are discussed below17 (Fig 4).
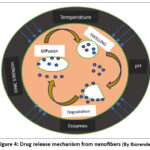 |
Figure 4: Drug release mechanism from nanofibers Click here to View Figure |
Diffusion-controlled release
Diffusion is one of the most basic processes, in which drug molecules move from the inside of the fibre to the surrounding media. The concentration differential between the surrounding fluid and the nanofiber matrix controls this process. Drug solubility, hydrophilicity, and polymer porosity all affect the rate of diffusion. While hydrophobic polymers, like polycaprolactone (PCL), slow down diffusion and lengthen release duration, hydrophilic fibres, like those formed of polyvinyl alcohol (PVA), allow faster diffusion, leading to rapid release18.
Degradation or erosion-controlled release
Drugs can be released through degradation or erosion of biodegradable polymers such as polylactic acid (PLA), polyglycolic acid (PGA), and their copolymer PLGA. During bulk erosion, water penetrates the whole fiber, causing uniform degradation, while during surface erosion, degradation occurs only on the surface of the fiber. As the polymer degrades, entrapped drugs are progressively liberated. The mechanism is particularly beneficial for long-term therapeutic applications requiring sustained drug release19.
Swelling-controlled release
Hydrophilic polymers, including gelatin, alginate, and chitosan, can absorb water and swell upon contact with aqueous environments. Swelling speeds up drug molecule mobility by increasing the free volume inside the fibres. When a quick therapeutic effect is sought, this mechanism is beneficial for localised therapies and wound dressings since it frequently offers a reasonably rapid release profile20.
Stimuli-responsive release
It is possible to construct sophisticated functional nanofibers that react to environmental stimuli like light, temperature, pH, and magnetic fields. Acidic or basic environments cause the polymer to dissolve or swell in pH-sensitive systems, releasing the medication. While photo- or magnetically responsive systems allow for on-demand, externally prompted release, thermo-responsive fibres release medications at predetermined temperatures. These intelligent systems hold great promise for intelligent wound dressings and targeted cancer treatment21.
Table 1: Drug fabrication Techniques for Electrospun Nanofibres
| Method | Working Principle | Key Features | Applications | References |
| Blend Electrospinning
|
Drug is mixed directly with the polymer solution to form a uniform blend before electrospinning. | Simple, scalable; may cause burst release; drug distributed throughout the fibre matrix. | Wound healing, local antibiotic delivery, anti-inflammatory drug release |
22,23 |
| Co-axial Electrospinning | Two separate solutions (core: drug; shell: polymer) are electrospun through a concentric nozzle, forming core–shell fibres. | Protects sensitive drugs; enables sustained/controlled release; reduces burst effect. | Protein/peptide delivery, dual-drug systems, tissue regeneration | 23,24 |
| Emulsion Electrospinning | Fibres with core-shell structures are created by electrospinning a drug and polymer-containing (W/O) or (O/W) emulsion.
|
Encapsulates hydrophilic drugs in hydrophobic polymers; moderate drug protection. | Hydrophilic drug delivery, tissue scaffolding, prolonged release systems |
23,25 |
| Surface Immobilization | Drug is loaded onto the surface of pre-formed fibres via physical adsorption or chemical conjugation (post-spinning). | Enables post-spinning functionalization; suitable for fragile drugs or fast local release. | Growth factors, enzymes, wound dressing, anti-infective surfaces |
26,27 |
Biopolymers in Electrospinning
Biopolymers are either naturally derived or synthetic materials that are biocompatible, biodegradable, and often bioactive.
Natural Biopolymers
Chitosan
Chitosan is widely used in electrospun nanofibres for its antimicrobial, biocompatible, and biodegradable properties. It promotes wound healing, supports cell adhesion, and is ideal for skin and bone tissue engineering. Its pH-responsive nature enables controlled drug release, making it effective in wound dressings, cancer therapy, and regenerative medicine applications28,29.
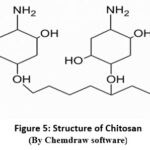 |
Figure 5: Structure of Chitosan (By Chemdraw software) Click here to View Figure |
Gelatin
Gelatin is commonly used in electrospun nanofibres because of their biocompatibility. Derived from collagen, it supports cell adhesion and proliferation, which is good for wound healing. Its ability to encapsulate drugs or bioactive agents also enables controlled delivery in biomedical applications30,31.
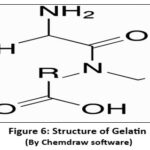 |
Figure 6 : Structure of Gelatin Click here to View Figure |
Alginate
Alginate is used in electrospun nanofibres for its biocompatibility, gel-forming ability, and moisture retention. Derived from seaweed, it supports wound healing and tissue regeneration. Often blended with other polymers due to poor spinnability alone, alginate-based nanofibres are ideal for wound dressings, drug delivery, and skin and cartilage engineering32,33.
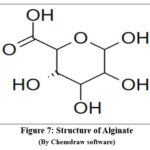 |
Figure 7: Structure of Alginate Click here to View Figure |
Silk fibroin
Silk fibroin is used in electrospun nanofibres for its exceptional mechanical strength, biocompatibility, and slow biodegradation. It supports cell attachment and proliferation, which is good for bone, skin, and nerve tissue engineering. Its qualities to incorporate the drugs or other factors enhance its role in regenerative medicine34,35.
Collagen
Collagen is widely used in electrospun nanofibres for its biocompatibility and for mimicking the extracellular matrix (ECM). It supports the applications especially in skin, bone, and cartilage regeneration. Its natural origin promotes integration with host tissues and accelerates healing36,37.
Synthetic Biopolymers
Polycaprolactone (PCL)
Polycaprolactone (PCL) is another polymer used in electrospun nanofibres because of their mechanical strength as well as slow biodegradability. It supports long-term tissue regeneration and is ideal for bone, vascular, and nerve engineering. PCL nanofibres can encapsulate drugs or biomolecules, enabling controlled release in drug delivery and wound healing applications38,39.
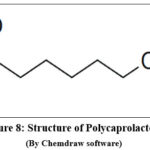 |
Figure 8: Structure of Polycaprolactone Click here to View Figure |
Polylactic acid (PLA)
Commonly used in electrospun nanofibres for its biodegradability, biocompatibility, and ease of processing. Derived from renewable sources, PLA supports cell growth and is ideal for scaffolds in bone, cartilage, and skin tissue engineering. Its fibres also enable controlled drug release for therapeutic applications40.
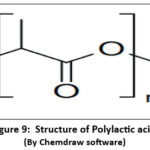 |
Figure 9: Structure of Polylactic acid Click here to View Figure |
Poly(lactic-co-glycolic acid)
Another synthetic polymer used in electrospun nanofibres for its tunable biodegradation rate, biocompatibility, and FDA approval. It supports tissue regeneration and enables sustained drug, gene, or protein delivery. PLGA nanofibres are ideal for wound healing, bone repair, and cancer therapy due to their controlled release capabilities41.
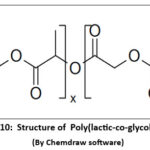 |
Figure 10: Structure of Poly(lactic-co-glycolic acid) Click here to View Figure |
Polyvinyl alcohol (PVA)
Commonly used in electrospun nanofibres for its excellent spinnability, biocompatibility, and water solubility. PVA is often blended with natural polymers to enhance mechanical properties and support cell adhesion and proliferation42,43.
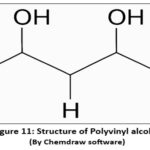 |
Figure 11: Structure of Polyvinyl alcohol Click here to View Figure |
Role of tissue engineering in modern therapeutics
Tissue engineering (TE) has emerged as a transformative approach in modern medicine, aiming to restore, maintain, or enhance the function of damaged tissues and organs. By integrating principles of biology, materials science, and engineering, tissue engineering creates biomimetic scaffolds which provide attachment of cell44.
In therapeutic applications, tissue engineering offers promising solutions for chronic diseases, trauma, congenital defects, and age-related degeneration. It addresses limitations associated with conventional treatments, like tissue shortages and dependence on drugs or prosthetics45.
With the introduction of electrospun nanofibers to tissue engineering, a new generation of biomimetic scaffolds has revolutionized modern therapeutics by closely resembling native extracellular matrix both structurally and functionally. They are perfect for promoting cell adhesion, proliferation, and differentiation because of their distinct nanoscale design, high surface-to-volume ratio, and adjustable porosity46,47.
In modern therapeutics, electrospun nanofibers are utilized not only as passive structural matrices but also as active biofunctional platforms. By adding bioactive substances to the nanofiber matrix or to their surface, such as growth factors, cytokines, or medications (via techniques like blend, coaxial, or surface-immobilization electrospinning), these scaffolds enable spatiotemporally controlled delivery, enhancing therapeutic outcomes while reducing systemic side effects48,49.
Applications span a wide range of tissues (Fig 12):
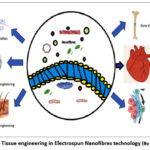 |
Figure 12: Tissue engineering in Electrospun Nanofibres technology. |
Skin tissue engineering
In skin tissue engineering, electrospun nanofibres are frequently utilised for medication administration, scaffolding, and wound dressings. They assist healing, encourage cell development, and imitate the extracellular matrix. Their high porosity and biocompatibility enable controlled release of bioactives, making them ideal for treating burns, ulcers, and chronic skin wounds50,51.
Bone tissue engineering
Electrospun nanofibres in bone tissue engineering provide a scaffold that mimics bone ECM, supporting osteoblast adhesion and proliferation. They can be reinforced with bioactive ceramics like hydroxyapatite and deliver growth factors such as BMP-2, promoting osteogenesis. Their porous structure aids vascularization, crucial for effective bone regeneration and healing52,53.
Cartilage tissue engineering
Electrospun nanofibres aid by mimicking the zonal structure of native cartilage and supporting chondrocyte attachment and growth. Their tunable mechanical properties and porosity enable nutrient diffusion and matrix deposition. Functionalization with bioactive molecules enhances chondrogenesis, making them ideal scaffolds for repairing articular cartilage defects and injuries54.
Nerve regeneration
Electrospun nanofibres support nerve regeneration by mimicking the extracellular matrix, guiding axonal growth with aligned fibres, and enhancing neuron adhesion. They can deliver neurotrophic factors like NGF and BDNF for sustained support. Their biocompatibility and customizable architecture make them ideal for repairing peripheral nerves and spinal cord injuries55.
Cardiac tissue engineering
Electrospun nanofibers enhance cardiomyocyte alignment, adhesion, and contraction by providing scaffolds that resemble the heart’s extracellular matrix. Conductive fibres enhance electrical signal transmission. They also enable delivery of growth factors and drugs, promoting vascularization and repair, making them ideal for myocardial patches and post-infarction cardiac regeneration56,57.
Cancer Therapy
In cancer treatment, electrospun nanofibres provide targeted, long-term medication delivery to tumour locations while reducing systemic toxicity. Their high surface area enhances drug loading, while targeting ligands enable site-specific action, making them effective platforms for post-surgical cancer treatment and recurrence prevention58,59.
Table 2: Applications of Nanofibres in drug delivery
| Type of Drug Delivery | Purpose | Target/Application Area | References |
| Topical drug delivery | Localized treatment, enhanced absorption | Wound healing, burns, skin infections | 60,61 |
| Transdermal delivery | Sustained drug release through skin | Pain relief, hormone therapy, anti-inflammatory drugs | 62 |
| Oral drug delivery | Controlled gastrointestinal release | Antibiotics, anticancer drugs | 63 |
| Implantable drug delivery systems | Long-term sustained release at target site | Cancer therapy, orthopedic implants | 64 |
| Inhalable nanofibres | Targeted pulmonary delivery | Respiratory diseases (e.g., tuberculosis, asthma) | 65 |
| Ocular drug delivery | Localized delivery to the eye | Glaucoma, infections, dry eye treatment | 66 |
| Vaginal/rectal delivery | Mucosal adhesion and localized therapy | Antiviral, HIV infections, contraceptive delivery | 67 |
| Cancer-targeted delivery | Site-specific drug release, reduced toxicity | Breast, skin, and colon cancer | 68 |
| Gene and protein delivery | Tissue regeneration, gene therapy | Bone repair, nerve regeneration, stem cell therapy | 69 |
| Antibiotic delivery | Prevent or treat infection | Post-surgical sites, bone infections, chronic wounds | 70 |
Recent Advances and Innovations
Nanofibre functionalization
Involves modifying the surface or composition of electrospun fibres to enhance their biological, chemical, or physical properties. This process can include chemical grafting, plasma treatment, or coating agents, or antibiotics71. Functionalization improves cell adhesion, targeted drug delivery, and antimicrobial activity, making nanofibres more effective for biomedical applications. For example, ligand-functionalized fibres can direct drug release to specific cells or tissues, while surface-immobilized proteins can promote tissue regeneration. Such modifications tailor nanofibres for advanced roles in tissue engineering, wound healing, and localized therapeutics with enhanced performance and biocompatibility72.
Smart Nanofibers
Advanced electrospun fibres known as “smart nanofibers” are designed to react to external stimuli like pH, temperature, or enzymes. These stimuli-responsive fibres are perfect for targeted therapy because they allow for controlled and on-demand medication release73. Smart nanofibers can also exhibit properties like self-healing, shape memory, or environmental sensing, increased their utility in wound healing, and precision medicine. By integrating functional polymers and bioactive agents, they offer dynamic and adaptable platforms for next-generation biomedical applications74.
Gene and Protein Delivery
Electrospun nanofibres offer a versatile platform for gene and protein delivery, enabling sustained and localized release of sensitive biomolecules. Because of their large surface area and adjustable shape, they can effectively encapsulate and shield proteins, RNA, or DNA from deterioration75. Techniques like coaxial electrospinning or surface immobilization ensure bioactivity retention and controlled release. These nanofibres support cell transfection, tissue regeneration, and therapeutic protein delivery, making them valuable in gene therapy, cancer treatment, and regenerative medicine applications76.
Green Electrospinning
Green electrospinning focuses on environmentally friendly practices by using non-toxic, biodegradable polymers and eco-friendly solvents like water or ethanol. It aims to reduce environmental impact while ensuring biocompatibility for biomedical applications. This sustainable approach supports safer fabrication of nanofibres for drug delivery, tissue engineering, and wound healing77,78.
3D Electrospun Structures
3D electrospun structures are engineered scaffolds with enhanced thickness, porosity, and mimicking extracellular matrix more effectively than 2D fibres79. Achieved through modified collectors or layered deposition, they support improved cell infiltration, vascularization for advanced tissue engineering applications80.
Conclusion and perspective
A flexible and effective method for creating nanofibrous scaffolds for a range of biological purposes is electrospinning. Electrospun nanofibers represent one of the most promising classes of biomaterials at the interface of nanotechnology, polymer science, and biomedical engineering. Over the past two decades, they have transitioned from a laboratory curiosity to a mainstream research focus due to their ability to mimic the natural extracellular matrix (ECM), provide controlled release of bioactive agents, and act as multifunctional scaffolds in tissue engineering, wound healing, drug delivery, and biosensing. The fabrication versatility of electrospinning—ranging from conventional solution and melt techniques to advanced coaxial, emulsion, and triaxial approaches—has enabled the design of highly tunable nanofibers with tailored morphology, porosity, and surface chemistry. By judicious selection of polymers, additives, and fabrication parameters, researchers have been able to achieve scaffolds that meet specific mechanical, biological, and therapeutic requirements.
The functional insights derived from these nanofibers highlight their ability to act as more than just structural scaffolds. They can be functionalized with drugs, proteins, growth factors, nanoparticles, or signaling molecules, thereby extending their role into therapeutic delivery and biosensing. The range of nanofibre applications in tissue engineering, wound healing, drug administration, and regenerative medicine has increased due to recent developments such as stimuli-responsive smart fibres, 3D electrospun architectures, and green electrospinning techniques. Despite remarkable progress, challenges remain in terms of scalability, regulatory approval, and clinical translation.
Future research should focus on integrating advanced materials, improving biological performance, and exploring personalized nanofibre-based therapies. With ongoing innovation, electrospun nanofibres hold immense promise as next-generation biomaterials for improving patient outcomes in modern medicine.
Acknowledgment
We are thankful to SGRR University for their encouragement and support for the submission of this article and also providing their valuable insights and suggestions during revision of the manuscript.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Shastri, S.S.; Varma. P.; Kandasubramanian, B., Biomedical Materials & Devices., 2024, 3, 1-24.
CrossRef - Hiwrale, A.; Bharati, S.; Pingale, P.; Rajput, A., , 2023, 9(9).
CrossRef - Das, A.; Shetty, S.; Shetty, R.; Suranjan Salins, S., Engineering., 2024,11(1), 2433147.
CrossRef - Barhoum, A.; Rasouli, R.; Yousefzadeh, M.; Rahier, H.; Bechelany, M., Handbook of nanofibers., 2019,3-43.
CrossRef - Keirouz, A.; Wang, Z.; Reddy, V.S.; Nagy, Z.K.; Vass, P.; Buzgo, M.; Ramakrishna, S.; Radacsi, N., Materials. Tech., 2023, 8(11), 2201723.
CrossRef - Babar, A.A.; Iqbal, N.; Wang, X.; Yu, J.; Ding, B., Electrospinning: Nanofabrication and Applications., 2019, 3-20.
CrossRef - Xue, J.; Wu, T.; Dai, Y.; Xia, Y., reviews., 2019, 119(8), 5298-5415.
CrossRef - Tören, E.; Buzgo, M.; Mazari, A.A.; Khan, M.Z., Polymers for Advance Technologies., 2024, 35(3), 6309.
CrossRef - Chinnappan, A.; Baskar, C.; Baskar, S.; Ratheesh, G.; Ramakrishna, S., Journal of Materials Chem., 2017, 5(48), 12657-12673.
CrossRef - Shitole, M.M.; Dugam, S.S.; Desai, N.D.; Tade, R.S.; Nangare, S.N., Asian Journal of Pharmacy and Technologies., 2020, 10(3), 187-201.
CrossRef - SIRIN, S.; CETINER, S.; Sarac, A.S., Kahramanmaras Sutcu Imam University Journal of Engi. Sciences., 2013, 16(2).
- Duygulu, N.E.; Balkas, M.; Ciftci, F.; Kucak, M., Inorganic Chemistry Communications., 2025,
- Zdraveva, E.; Fang, J.; Mijovic, B.; Lin, T., Structure and properties of high-performance fibers., 2017, 267-300.
CrossRef - Ji, D.; Lin, Y.; Guo, X.; Ramasubramanian, B.; Wang, R.; Radacsi, N.; Jose, R.; Qin, X.; Ramakrishna, S., Nature Reviews Methods Primers., 2024, 4(1), 1.
CrossRef - Lukáš, D.; Sarkar, A.; Martinová, L.; Vodsed’álková, K.; Lubasová, D.; Chaloupek, J.; Pokorný, P.; Mikeš, P.; Chvojka, J.; Komárek, M., Textile progress., 2009, 41(2), 59-140.
CrossRef - Bölgen, N.; Demir, D.; Aşık, M.; Sakım, B.; Vaseashta, A., Electrospun Nanofibers: Principles, Technology and Novel Applications., 2022, 3-34. Springer International Publishing.
CrossRef - Abdul Hameed, M.M.; Mohamed Khan, S.A.P.; Thamer, B.M.; Rajkumar, N.; El‐Hamshary, H.; El‐Newehy, M., Polymers for Advanced Technologies., 2023, 34(1), 6-23.
CrossRef - Zandi, N.; Lotfi, R.; Tamjid, E.; Shokrgozar, M.A.; Simchi, A., Materials Science and Engineering: C., 2020,108, 110432.
CrossRef - Dong, Y.; Liao, S.; Ngiam, M.; Chan, C. K.; Ramakrishna, S., Tissue Engineering Part B: Reviews., 2009, 15(3), 333-351.
CrossRef - Wang, Y.; Li, Z.; Shao, P.; Hao, S.; Wang, W.; Yang, ; Wang, B., Materials Science and Engineering: C., 2014, 44, 109-116.
CrossRef - Huang, C.; Soenen, S. J.; Rejman, J.; Lucas, B.; Braeckmans, K.; Demeester, J.; De Smedt, S.C., Chemical Society Reviews., 2011, 40(5), 2417-2434.
CrossRef - Valizadeh, A.; Mussa, Farkhani, S., IET nanobiotechnology., 2014,8(2), 83-92.
CrossRef - Buzgo, M.; Mickova, A.; Rampichova, M.; Doupnik, M., Core-shell nanostructures for drug delivery and theranostics., 2018, 325-347.
CrossRef - Han, D.; Steckl, A.J., ChemPlus Chem., 2019, 84(10), 1453-1497.
CrossRef - Zhang, C.; Feng, F.; Zhang, H., Trends in Food Science & Technology., 2018, 80, 175-186.
CrossRef - Wang, Z.G.; Wan, L.S.; Liu, Z.M.; Huang, X.J.; Xu, Z.K., Journal of Molecular Catalysis B: Enzymatic., 2009, 56(4), 189-195.
CrossRef - Rather, A.H.; Khan, R.S.; Wani, T.U.; Beigh, M.A.; Sheikh, F.A., Biotechnology and Bioengineering., 2022, 119(1), 9-33.
CrossRef - Kalantari, K.; Afifi, A.M.; Jahangirian, H.; Webster, T.J., Carbohydrate polymers., 2019, 207, 588-600.
CrossRef - Sun, K.; Li, Z.H., Express Polymer Letters., 2011, 5(4).
CrossRef - Huang, Z.M.; Zhang, Y.Z.; Ramakrishna, S.; Lim, C.T., , 2004, 45(15), 5361-5368.
CrossRef - Songchotikunpan, P.; Tattiyakul, J.; Supaphol, P., International Journal of Biological Macromolecules., 2008, 42(3), 247-255.
CrossRef - Wróblewska-Krepsztul, J.; Rydzkowski, T.; Michalska-Pożoga, I.; Thakur, V.K., , 2019,9(3), 404.
CrossRef - Taemeh, M.A.; Shiravandi, A.; Korayem, M.A.; Daemi, H., Carbohydrate polymers., 2020, 228, 115419.
CrossRef - Kim, S.H.; Nam, Y.S.; Lee, T.S.; Park, W.H., Polymer Journal., 2003, 35(2), 185-190.
CrossRef - Zhang, J.G.; Mo, X.M., Frontiers of Materials Science., 2013,7(2), 129-142.
CrossRef - Matthews, J.A.; Wnek, G.E.; Simpson, D.G.; Bowlin, G.L., , 2002,3(2), 232-238.
CrossRef - Yang, L.; Fitié, C.F.; Van Der Werf, K.O.; Bennink, M.L.; Dijkstra, P.J.; Feijen, J., , 2008, 29(8), 955-962.
CrossRef - Azari, A.; Golchin, A.; Maymand, M.M.; Mansouri, F.; Advanced Pharmaceutical Bulletin., 2021,12(4), 658.
- Reneker, D.H.; Kataphinan, W.; Theron, A.; Zussman, E.; Yarin, A.L., , 2002, 43(25), 6785-6794.
CrossRef - Maleki, H.; Azimi, B.; Ismaeilimoghadam, S.; Danti, S., Applied Sciences., 2022, 12(6), 3192.
CrossRef - Herrero-Herrero, M.; Gomez-Tejedor, J.A.; Valles-Lluch, A., , 2021, 13(5), 695.
CrossRef - Ding, B.; Kim, H.Y.; Lee, S.C.; Shao, C.L.; Lee, D.R.; Park, S.J.; Kwag, G.B.; Choi, K.J., Journal of Polymer Science Part B: Polymer Physics., 2002, 40(13), 1261-1268.
CrossRef - Yang, E.; Qin, X.; Wang, S., Materials Letters., 2008,62(20), 3555-3557.
CrossRef - Vacanti, C.A., Journal of cellular and molecular medicine., 2007, 10(3), 569.
CrossRef - Lavik, E.; Langer, R., Applied microbiology and biotechnology., 2004, 65(1), 1-8.
CrossRef - Kishan, A.P.; Cosgriff‐Hernandez, E.M., Journal of Biomedical Materials Research Part A., 2017,105(10), 2892-2905.
CrossRef - Sill, T.J.; Von Recum, H.A., , 2008, 29(13), 1989-2006.
CrossRef - Chen, K.; Li, Y.; Pan, W.; Tan, G., Macromolecular Bioscience., 2023, 23(2), 2200380.
CrossRef - Alessandrino, A.; Marelli, B.; Arosio, C.; Fare, S.; Tanzi, M.C.; Freddi, G., Engineering in life sciences., 2008, 8(3), 219-225.
CrossRef - Law, J.X.; Liau, L.L.; Saim, A.; Yang, Y.; Idrus, R., Tissue engineering and regenerative medicine., 2017, 14(6), 699-718.
CrossRef - Hernández‐Rangel, A.; Martin‐Martinez, E.S., Journal of Biomedical Materials Research Part A., 2021, 109(9), 1751-1764.
CrossRef - Shin, S.H.; Purevdorj, O.; Castano, O.; Planell, J.A.; Kim, H.W., Journal of tissue engineering., 2012, 3(1).
CrossRef - Linder, H.R.; Glass, A.A.; Day, D.E.; Sell, S.A., , 2020, 7(4), 165.
CrossRef - Liu,, Y.; Liu, L.; Wang, Z.; Zheng, G.; Chen, Q.; Luo, E., Current Stem Cell Research & Therapy., 2018, 13(7), 526-532.
CrossRef - Behtaj, S.; Ekberg, J.A.; St John, J.A.; , 2022,14(2), 219.
CrossRef - Sell, S.A.; McClure, M.J.; Garg, K.; Wolfe, P.S.; Bowlin, G.L., Advanced drug delivery reviews., 2009,61(12), 1007-1019.
CrossRef - Kitsara, M.; Agbulut, O.; Kontziampasis, D.; Chen, Y.; Menasché, P., Acta biomaterialia., 2017, 48, 20-40.
CrossRef - Contreras-Cáceres, R.; Cabeza, L.; Perazzoli, G.; Díaz, A.; López-Romero, J.M.; Melguizo, C.; Prados, J., , 2019,9(4), 656.
CrossRef - Chen, Z.; Zhang, A.; Hu, J.; Wang, X.; Yang, Z., Biomaterials science., 2016, 4(6), 922-932.
CrossRef - Tomar, Y.; Pandit,, N.; Priya S.; Singhvi, G., ACS omega., 2023, 8(21), 18340-18357.
CrossRef - Tören, E., Biomedical Materials & Devices., 2025, 1-24.
- Esentürk, İ.; Erdal, M.S.; Güngör, S., Journal of Faculty of Pharmacy of Istanbul University., 2016, 46(1), 49-69.
- Akhgari, A.; Shakib, Z.; Sanati, S., Nanomedicine Journal., 2017, 4(4).
- Valizadeh,, A.; Asghari, S.; Abbaspoor, S.; Jafari, A.; Raeisi, M.; Pilehvar, Y., Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology., 2023, 15(6), e1909.
CrossRef - Marulanda, K.; Mercel,, A.; Gillis, D.C.; Sun, K.; Gambarian, M.; Roark, J.; Weiss, J.; Tsihlis, N.D.; Karver, M.R.; Centeno, S.R.; Peters, E.B., Advanced healthcare materials., 2021, 10(13), 2100302.
CrossRef - Uzel,, E.; Durgun, M.E.; Esentürk-Güzel, İ.; Güngör, S.; Özsoy, Y., , 2023, 15(4), 1062.
CrossRef - Mesquita,, L.; Galante, J.; Nunes, R.; Sarmento, B.; das Neves, J., , 2019, 11(3), 1-20.
CrossRef - Norouzi, M.; Nazari, B.; Miller, D.W., Electrospun materials for tissue engineering and biomedical applications., 2017,337-356.
CrossRef - Lee, S.; Jin, G.; Jang, J.H., Journal of biological engineering., 2014, 8(1), 1-30.
CrossRef - Silva, G.L.G.; de Bustamante, M.S.D.S.; Dias, M.L.; Costa, A.M.; Rossi, A.M.; dos Santos Matos, A.P.; Santos-Oliveira, R.; Ricci-Junior, E., Arabian Journal of Chemistry., 2023, 16(1), 104392.
CrossRef - Zamel, D.; Khan, A.U., Polymers for Advanced Technologies., 2021, 32(12), 4587-4597.
CrossRef - Lou, L.; Osemwegie, O.; Ramkumar, S.S., Industrial & Engineering Chemistry Research., 2020,59(13), 5439-5455.
CrossRef - Chamanehpour, E.; Thouti, S.; Rubahn, H.G.; Dolatshahi‐Pirouz, A.; Mishra, Y.K., Advanced Materials Technologies., 2024,9(3), 2301392.
CrossRef - Tien, N.D.; Lyngstadaas, S.P.; Mano, J.F.; Blaker, J.J.; Haugen, H.J., , 2021, 26(9), 2683.
CrossRef - Riley, M.K.; Vermerris, W., , 2017, 7(5), 1-19.
CrossRef - Todorova, R., Drug Delivery., 2011, 18(8), 586-598.
CrossRef - Berdimurodov, E.; Dagdag, O.; Berdimuradov, K.; Wan Nik, W.M.N.; Eliboev, I.; Ashirov, M.; Niyozkulov, S.; Demir, M.; Yodgorov, C.; Aliev, N., , 2023, 11(5), 150.
CrossRef - Cho, Y.; Beak, J.W.; Sagong, M.; Ahn, S.; Nam, J.S.; Kim, I.D., Advanced Materials., 2025,
- Keirouz, A.; Chung, M.; Kwon, J.; Fortunato, G.; Radacsi, N., Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology., 2020, 12(4), e1626.
CrossRef - Chen, Y.; Dong, X.; Shafiq, M.; Myles, G.; Radacsi, N.; Mo, X., Advanced Fiber Materials., 2022, 4(5), 959-986.
CrossRef
Abbreviations List
EN: Electrospun nanofibers
PCL: Polycaprolactone
PLA: Polylactic acid
PLGA: Poly(lactic-co-glycolic acid)
PGA: Polyglycolic acid
PVA: Polyvinyl alcohol
ECM: Extracellular matrix
TE: Tissue engineering
RNA: Ribonucleic acid
DNA: Deoxyribonucleic acid
Accepted on: 15 Sep 2025
Second Review by: Dr. Farnaz Fattahi
Final Approval by: Dr. Charanjeet Kaur

















