Development and Characterization of Biogenic Aurum Nanoparticles from a Southeast Asian Herb with Antioxidant Potential
1Centre of Diagnostic, Therapeutic and Investigative Studies, Faculty of Health Sciences, Universiti Kebangsaan Malaysia, Jalan Raja Muda Abdul Aziz, Kuala Lumpur, Malaysia
2Science Forensic Programme, Faculty of Health Sciences, Universiti Kebangsaan Malaysia, Bangi, Selangor
Corresponding Author E-mail:atikahnasir@ukm.edu.my
DOI : http://dx.doi.org/10.13005/ojc/420113
ABSTRACT:This study explores the sustainable synthesis of Aurum nanoparticles (AuNPs) using Biokesum®, a Southeast Asian Herb called Polygonum minus leaves extract serves as both reducing and capping agent. Ultraviolet visible spectroscopy confirmed surface plasmon resonance peaks at 530–545 nm indicating well-formed AuNPs. Dynamic light scattering and zeta potential analysis revealed hydrodynamic diameters of 66 to 112 nm and moderate stability from −8.88 to −20 mV, respectively. Transmission electron microscopy and field emission scanning electron microscopy paired with energy dispersive spectroscopy revealed the morphology of AuNPs is spherical to quasi-spherical particles less than 20 nm, with phytochemical capping evidenced by carbon and oxygen signatures. X-ray fluorescence analysis confirmed the successful formation of AuNPs by identifying and quantifying a significant amount of AuNPs, residual chloride, and trace phytochemical. Antioxidant assays (DPPH) revealed pH-dependent activity, with maximal activity at pH 4 (~35%), while Biokesum® extract (~95%) and quercetin (~97%) maintained consistently high activity across all pH values. Sample A5 (66 nm, −14.6 mV) emerged as the most optimal formulation. These findings highlight Biokesum® as an effective, eco-friendly alternative for AuNPs synthesis for biomedical and pharmaceutical applications.
KEYWORDS:Aurum nanoparticles (AuNPs); Antioxidant activity; Green synthesis; Polygonum Minus; Phytochemical capping
Introduction
Oxidative stress results from an imbalance between reactive oxygen species (ROS) generation and endogenous antioxidant defences. This imbalance plays a central role in cellular damage and the progression of chronic diseases such as cardiovascular disorders, neurodegeneration, diabetes, and cancer 1. Environmental pollutants and modern lifestyle factors further elevate ROS levels, emphasizing the need for effective antioxidant agents. Although natural antioxidants mitigate ROS by donating electrons to stabilize reactive species 2,their therapeutic use is limited by instability, low solubility, and susceptibility to degradation 3.
Nanomaterials have gained increasing attention as alternative antioxidants because of their high surface-to-volume ratios, tunable physicochemical properties, and intrinsic redox activities. Organic nanomaterials, metal oxides, and noble metal nanoparticles including Aurum nanoparticles (AuNPs) exhibit radical scavenging and enzyme-mimetic behaviors 4,5. AuNPs are particularly attractive for biomedical applications because of their biocompatibility, stability, and low toxicity 6. However, conventional AuNP synthesis often requires hazardous chemicals and energy-intensive procedures, limiting sustainability.
Green nanotechnology addresses these limitations by utilizing phytochemicals as reducing and stabilizing agents, enabling eco-friendly and cost-effective nanoparticle production7. Polygonum minus also known as kesum, a Southeast Asian herb rich in phenolic acids, flavonoids, and terpenoids, exhibits antioxidant, anti-inflammatory, antimicrobial, and anticancer properties8,9. Its abundant flavonoids contribute significantly to these bioactivities10 and standardized extracts such as Biokesum® serve to further validate its functional potential11. Nevertheless, phytochemical instability and low bioavailability limit direct therapeutic application12.
Therefore, this study aims to develop biogenic AuNPs using Biokesum® leaf extract through an environmentally friendly synthesis approach and to comprehensively characterize the resulting nanoparticles in terms of their optical properties, particle size distribution, surface charge, morphology, and elemental composition. In addition, the antioxidant potential of the synthesized AuNPs is evaluated under different pH conditions to elucidate their functional performance. By correlating physicochemical characteristics with bioactivity outcomes, this work seeks to establish the structure–stability–antioxidant relationship of Biokesum®-derived AuNPs and to highlight their promise as sustainable nanomaterials for biomedical and pharmaceutical applications.
Materials and Methods
Materials
Aurum (III) chloride trihydrate, AuCl4H7O3 (99.9%) purchased from Shanghai Macklin Biochemical. The Polygonum minus water extracts used in this study were from Biotropics Malaysia under the trade names of Biokesum® (batch number: KE 220103). Biokesum® is a water extract of the leaves of Polygonum minus, standardized based on a specification of not less than 0.45% of quercetin-3-glucuronide and not less than 100 mg GAE/g dE total phenolic content. The high-performance liquid chromatography (HPLC) fingerprint of Biokesum® was obtained according to the HPLC-diode array detection method using a Kinetex 1.7 μm C18 column (2.1 × 150 mm). The mobile phase consisted of solvent A: 0.10% formic acid in water and B: 0.10% formic acid in acetonitrile mixed according to a linear gradient program with a flow rate of 0.4mL/min. The solvent composition was set to 11% B from t= 0-18 min followed by 95% B from t = 20-23 min, before final t = 25 min at 11% B. Injection volume was 2.0 μl and the chromatogram was recorded at 366 nm.
Preparation of Stock Solutions
Initially, an Au precursor stock solution of 1 mM AuCl4H7O3 was prepared by mixing 39.383 mg into 100 mL of pure deionized water (HPLC grade) to ensure purification. The Au precursor stock solution was labelled and stored in an aluminium wrap Schott bottle at 4°C until further use. Biokesum® stock solution (0.1%) was prepared by adding 0.1g of powdered Biokesum® into 100ml deionized water. The mixture was stirred slowly for 10 min to form a homogeneous solution. Later, this stock solution was filtered by a Teflon membrane 0.04 μm to remove any impurities. Finally, the filtrate was labelled and kept in a Schott bottle at 4°C until further use. The stock solutions of Au precursor and Biokesum® were used as control solutions to monitor the production of AuNP colloidal solutions. Both of the stock solutions were labelled as Control 1 and Control 2, respectively.
Synthesis of AuNPs
To study the ability of Biokesum® extract as a bioreducing agent to produce AuNPs, various volumes of Biokesum® extract were used using a fixed volume of 1 mM AuCl4H7O3. A total of 5 mL of AuCl4 stock solution was added dropwise to a set of volumes of Biokesum® extract solution as determined in Table 1. The solution mixture was stirred continuously using a magnetic stirrer at room temperature. Initial observations were recorded through the color change of the solution from pale yellow to purple, then to ruby red. The formation of AuNPs colloidal solution was considered occurred when the mixed solution changed from pale yellow to purple to ruby red. The colour change can be observed with the naked eye. Further analysis was performed using an ultraviolet visible (UV-VIS) spectroscopy technique with a wavelength range of 400 to 800 nm. All experiments were performed in triplicate to verify the validity.
Table 1: Formulation parameters for Au nanoparticle synthesis using Biokesum® extract
|
Sample |
Volume 1 mM AuCl4H7O3 | Volume 0.1% Polygonum minus stock solution |
Ratio of ACl4H7O3 : Polygonum minus |
|
Control 1 |
5 | 0 | 5:0 |
| Control 2 | 0 | 5 |
0:5 |
|
A1 |
5 | 1 | 5:1 |
| A2 | 5 | 2 |
5:2 |
|
A3 |
5 | 3 | 5:3 |
| A4 | 5 | 4 |
5:4 |
|
A5 |
5 | 5 | 5:5 |
| A6 | 5 | 6 |
5:6 |
Characterization of AuNPs
AuNPs formation was monitored using an ultraviolet/visible/near infrared spectrometer (Perkin Elmer Lambda 950) over 400–800 nm at 1 nm intervals with deionized water as baseline. Hydrodynamic diameter and zeta potential were measured with a nanoparticle size analyzer (Malvern Zetasizer Nano ZS, measurement range 0.3 nm–10 μm, zeta potential range –150 mV to +150 mV) after dilution in deionized water at 25 °C in triplicate. Transmission electron microscopy (TEM) was performed on a Talos L120C microscope (120 kV, 0.2 nm resolution, 35×–910× magnification) by drop‑casting the colloid onto carbon‑coated copper grids. Field emission scanning electron microscopy coupled with energy‑dispersive X‑ray spectroscopy was carried out on a ZEISS MERLIN field emission scanning electron microscope at 15 kV with elemental mapping. X‑ray fluorescence analysis of lyophilized nanoparticle powder was conducted on a Rigaku ZSX Primus IV wavelength dispersive X‑ray fluorescence spectrometer.
Radical Scavenging Activity
The antioxidant activity of the 2,2-diphenyl-1-picrylhydrazyl (DPPH) method was used to evaluate the radical scavenging activity of AuNPs. The dark purple DPPH free radical changed to a yellow stable molecule when the electrons of the DPPH free radical interact with the hydrogen of the free radical scavenging antioxidant. Therefore, the degree of DPPH color change can indicate the antioxidant scavenging potential of the extract. To measure the antioxidant activity, the decrease in color intensity was monitored over time at a wavelength of 517 nm. This wavelength corresponds to the strongest absorption for DPPH. All measurements were performed in triplicate.
Cytotoxicity Assay
V79-4 cells were cultured in DMEM supplemented with 10% FBS, 1 mM sodium pyruvate and 1% antibiotics, and maintained at 37 ± 1 °C in 5 ± 1% CO₂. Confluent cells were detached by trypsinisation, counted, and seeded into 96-well plates at 10,000 cells/well, then incubated for at least 12 hours to reach 70–80% confluency. The test item was sterilised by 0.2 μm filtration, prepared at 50% v/v, and serially diluted to obtain additional concentrations of 25%, 12.5%, 6.25%, 3.13% and 1.56% v/v. Cells were exposed to each concentration in triplicate for 24 h ± 30 min under standard incubation conditions. After exposure, 5 mg/ml MTT solution was added to each well and incubated for 4 h ± 30 min, followed by removal of the reagent and solubilisation of the formazan crystals. Absorbance was measured to determine cell viability.
Results and Discussion
HPLC Chromatogram Analysis of Biokesum® and Reference Standards
The HPLC analysis of Biokesum® (Figure 1a) revealed two major peaks corresponding to quercetin-3-O-glucuronide and quercitrin, as confirmed by comparison with retention times (RT) of authentic standards (Figure 1b). The elution profiles of these compounds in Biokesum® (RT 7.332 min and 14.632 min) closely align with those of the standards (RT 7.335 min and 14.597 min), strongly supporting their identification. Retention time matching is a widely accepted method for preliminary compound identification in HPLC13. The minor discrepancies in retention times (~0.003–0.035 min) are likely due to slight variations in experimental conditions such as column temperature or mobile phase composition14.
 |
Figure 1: High-performance liquid chromatography (HPLC) chromatogram of Biokesum® extract and reference standards; (a) Biokesum® chromatogram showing quercetin-3-O-glucuronide (RT 7.332 min) and quercitrin (RT 14.632 min). (b) Reference standards of quercetin-3-O-glucuronide (RT 7.335 min) and quercitrin (RT 14.597 min) 12. |
The quantification of quercetin-3-O-glucuronide (1.3%) and quercitrin (0.4%) in Biokesum® highlights the significant presence of these bioactive flavonoids. Quercetin derivatives, particularly glucuronides, are renowned for their antioxidant and anti-inflammatory properties, as demonstrated in preclinical models15. Additionally, glucuronidation often enhances solubility and bioavailability, as observed in pharmacokinetic studies of quercetin derivatives16, suggesting that Biokesum® could offer improved in vivo efficacy. The identified concentrations (1.3% and 0.4%) are pharmacologically relevant, as similar levels of quercetin glycosides in plant extracts have demonstrated bioactivity in reducing oxidative stress 17. However, the presence of unresolved minor peaks in the chromatogram (Figure 1a) implies that other phytoconstituents, such as phenolic acids or additional flavonoids, may contribute synergistically to Biokesum®’s bioactivity.
Ultraviolet-visible Spectrum and Surface Plasmon Resonance (SPR) Analysis
The successful biosynthesis of AuNPs was confirmed using ultraviolet‑visible (UV-Vis) spectroscopy (Figure 2a), which detects the characteristic surface plasmon resonance (SPR) band arising from the oscillation of surface electrons18. All samples (A2–A6) showed distinct surface plasmon resonance (SPR) peaks between 534 to 568 nm (Figure 2b), indicating the reduction of Au³⁺ to Au⁰ and the formation of spherical or quasi-spherical nanoparticles. A systematic blue shift in the surface plasmon resonance maximum was observed with increasing Biokesum® extract concentration, shifting from 568 nm (A2) to 534 nm (A6). This progressive shift toward shorter wavelengths is a well-established indicator of reduced average particle size 19. The sharp, intense, and symmetric peak observed for samples A4 to A6 suggests the formation of small, monodisperse, and well-stabilized nanoparticles.
This result can be attributed to the high content of bioactive phytochemicals in Biokesum®, particularly quercetin derivatives, which act as strong reducing and capping agents 17,20. At the optimal concentration in A6, these compounds promote rapid nucleation and effective surface passivation, preventing ripening and aggregation 21. In contrast, lower extract concentrations (A2, A3) produced broader, weaker peaks at 565–568 nm, indicating polydispersity, possible aggregation, and incomplete reduction due to limited reducing agents5,7. The absence of secondary peaks in A4–A6 confirms minimal anisotropic structures or aggregation22. Similar trends were reported for Zingiber officinale-mediated AuNPs, where higher extract levels resulted in a blue shift and smaller particles19. Overall, extract concentration is a key determinant of nanoparticle size and stability, with A4-A6 representing the optimal Biokesum®- AuNPs ratio.
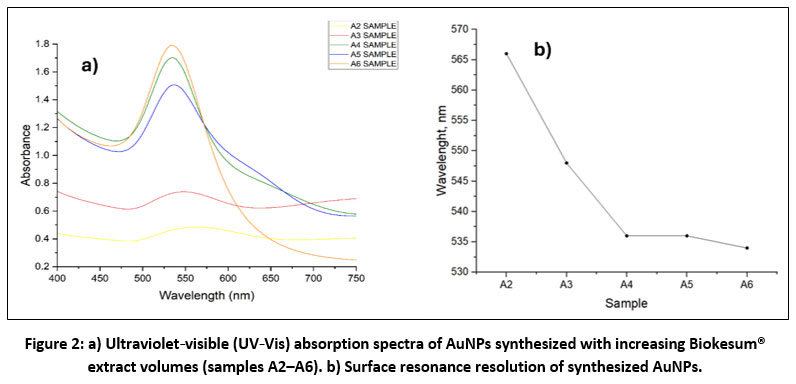 |
Figure 2: a) Ultraviolet‑visible (UV‑Vis) absorption spectra of AuNPs synthesized with increasing Biokesum® extract volumes (samples A2–A6). b) Surface resonance resolution of synthesized AuNPs. |
Zeta Potential Analysis
As summarized in Table 2, dynamic light scattering (DLS) and zeta potential collectively provided key insights into colloidal stability and size distribution, demonstrating the strong influence of extract concentration on nanoparticle characteristics. Zeta potential analysis showed moderate stability for samples A5 (−14.6 mV) and A4 (−13 mV), though the values remained below the ±30 mV threshold associated with long-term stability25. Sample A6 (−8.88 mV) aggregated rapidly despite its small size, indicating that high Biokesum® concentrations enhance reduction but may yield insufficient surface passivation26. The relatively high zeta potential of sample A2 (−20 mV) likely reflects the presence of charged aggregates rather than stable nanoparticles. The stability trend (A5 > A4/A3 > A6 > A2) reflects a balance between reduction kinetics and capping efficiency. Rapid nucleation at higher extract concentrations is facilitated by flavonoids such as quercetin-3-O-glucuronide21, but excessive extract can saturate reduction sites without adequate surface stabilization. In contrast to the previous study, where increasing plant extract concentration led to red-shifted SPR bands indicative of nanoparticle aggregation5, the present findings suggest that the distinct flavonoid composition of Biokesum® promotes efficient Au3+ reduction while exerting a differential influence on AuNPs stabilization dynamics. Overall, sample A5 represents the most promising formulation, balancing small size, monodispersed, and moderate stability.
Table 2: Stability characteristics of synthesized AuNPs based on DLS and zeta potential analyses
|
Sample |
DLS Size (nm) | Zeta Potential (mV) |
| A6 | 67.65 |
-8.88 |
|
A5 |
66 | -14.6 |
| A4 | 105 |
-13.0 |
|
A3 |
112 | -13.0 |
| A2 | 250 |
-20.0 |
High-Resolution Transmission Electron Microscopy (HRTEM) Analysis
Transmission Electron Microscopy (TEM) analysis of sample A5 revealed predominantly quasi-spherical, irregularly shaped nanoparticles, indicative of isotropic growth facilitated by plant-derived reducing and capping agents (Figure 3). The particles were well-dispersed with minor aggregation, likely due to partial surface capping or limited phytochemical stabilization, a phenomenon similarly observed in Azadirachta indica–mediated AuNPs27. The majority of nanoparticles measured below 20 nm, confirming their nanometric size and suitability for biomedical, catalytic, and environmental applications. These findings align with previous studies where Hibiscus rosa-sinensis–synthesized AuNPs exhibited sizes of 10–30 nm28, while Azadirachta indica–derived nanoparticles averaged 36.4 nm27.
 |
Figure 3: Transmission electron microscopy (TEM) images of sample A5 AuNPs (Talos L120C, 120 kV). (a) Particle distribution (500 nm scale). (b) Quasi‑spherical morphology with sizes below 20 nm (20 nm scale). |
Field Emission Scanning Electron Microscopy with Energy-Dispersive X-ray (FESEM-EDX) Analysis
Figure 4 shows FESEM analysis of sample A5 revealed predominantly quasi-spherical, irregularly shaped nanoparticles that were relatively well-dispersed with slight aggregation, a common feature in plant-mediated green synthesis27,28. Elemental mapping confirmed dense Au distributions at particle sites, while carbon (C) and oxygen (O) were more homogeneously distributed, likely originating from Biokesum® phytochemicals acting as reducing and stabilizing agents. EDX spectra supported these observations, showing characteristic peaks for Au (~2.3 keV), C (~0.28 keV), and O (~0.52 keV), consistent with typical biosynthesized AuNPs. These findings align with previous reports indicating that green-synthesized AuNPs are coated with plant-derived organic molecules, which enhance stability and prevent aggregation27,28. Overall, the results confirm that Biokesum® effectively facilitated the green synthesis of nanoscale AuNPs with good dispersion and biomolecule capping.
 |
Figure 4: Field‑emission scanning electron microscopy with energy‑dispersive X‑ray spectroscopy (FESEM‑EDX) of sample A5. (a) EDX spectrum showing Au and (b) individual elemental distribution of Au. |
X-ray Fluorescence (XRF) Analysis
X-ray fluorescence (XRF) analysis was conducted to confirm the elemental composition of the Biokesum® mediated AuNPs. The result of sample A5 indicated the presence of Au (intensity 4.6624), confirming the successful reduction of Au3+ precursor into elemental Au by Biokesum® extract. The residual Cl (intensity 0.2307) likely originates from incomplete reduction of the Au precursor, most commonly chloroauric acid (HAuCl₄), a typical feature in green synthesis18. Similar observations were reported by19, where Zingiber officinale–mediated AuNPs retained Cl due to partial reduction. The trace Al, K, and S with intensity 0.1423, 0.1929 and 0.1510 respectively, are characteristic of phytochemicals and organic acids in plant extracts, supporting the role of biomolecules in capping and stabilizing the nanoparticles29. Although XRF does not provide oxidation state information, the confirmed presence of Au strongly supports nanoparticle formation, particularly when interpreted alongside complementary characterization techniques such as UV–Vis spectroscopy or XRD. Overall, the XRF findings verify that Biokesum® extract effectively facilitates the formation of Au nanoparticles while maintaining a relatively clean elemental profile, consistent with previously reported green synthesis approaches.
Antioxidant Activity Analysis (DPPH Assay)
Quercetin, a well-known flavonoid antioxidant, is most stable and effective in mildly acidic conditions15. Similarly, the biosynthesized AuNPs, sample A5 exhibited maximal DPPH scavenging (~35%) at pH 4, likely because the Biokesum®-derived phytochemical capping remains protonated and can donate hydrogen atoms to neutralize radicals. From Figure 5, at pH 5 and 7, activity dropped sharply (~17–19%), possibly due to deprotonation of phenolic groups and changes in nanoparticle surface charge, rather than quercetin degradation, which occurs only above pH 930. Under moderately alkaline conditions (pH 9–11), activity increased slightly (~19–23%), reflecting enhanced nanoparticle formation and greater surface area for biomolecule adsorption. At pH 13, scavenging dropped drastically (~6.5%) due to phytochemical degradation or desorption, consistent with flavonoid instability under strong alkalinity 31
In contrast, Biokesum® extract (~95%) and quercetin (~97%) maintained high antioxidant activity across all pH levels, highlighting their inherent stability. The lower activity of AuNPs aligns with reports that plant metabolites consumed during Au³⁺ reduction reduce the available pool of bioactive compounds20. However, some studies report higher AuNPs activity than crude extracts, depending on phytochemical capping and synthesis conditions22. Overall, these results demonstrate that pH significantly influences the antioxidant performance of Biokesum®-mediated AuNPs, with mildly acidic conditions favoring maximal activity, moderate alkalinity supporting nanoparticle formation, and extreme alkalinity compromising stability.
 |
Figure 5: DPPH radical scavenging activity of sample A5, Biokesum® extract, and quercetin standard. Click here to View table |
Cytotoxicity Evaluation of Biokesum® on V79-4 Cells
The cytotoxicity of sample A5 was evaluated using the MTT assay on V79-4 fibroblast cells to assess biocompatibility (Table 3). Cell viability after 24 h exposure to Biokesum® (1.56–50 % v/v) remained high, ranging from 101% to 87%, with no treatment falling below the ISO 10993-5 cytotoxicity threshold of 70% (ISO 10993-5:2009). The MTT assay measures mitochondrial activity via reduction of MTT to formazan, reflecting cellular metabolic integrity. These results indicate that Biokesum® does not interfere with mitochondrial function, consistent with reports for green-synthesized AuNPs capped with plant-derived biomolecules, which typically maintain >80 % viability19,22.
Table 3: V79-4 cell viability after 24-hour exposure to sample A5
|
|
Optical Density, OD (570 nm) | Standard Deviation (SD) | Cell Viability (%) | ||||
|
Replicate 1 |
Replicate 2 | Replicate 3 |
Average |
||||
| Negative Control | 3.214 | 3.051 | 3.032 | 3.099 | 0.10 |
100 |
|
|
Test Item Concentration (% v/v) |
1.56 |
2.999 | 3.119 | 3.269 | 3.129 | 0.14 | 101 |
| 3.13 | 3.452 | 3.426 | 2.822 | 3.233 | 0.36 |
104 |
|
|
6.25 |
3.341 | 3.443 | 2.911 | 3.232 | 0.28 | 104 | |
| 12.5 | 2.716 | 2.878 | 2.660 | 2.751 | 0.11 |
89 |
|
|
25 |
2.919 | 2.788 | 2.897 | 2.868 | 0.07 | 93 | |
| 50 | 2.709 | 2.719 | 2.679 | 2.702 | 0.02 |
87 |
|
|
Positive Control (10 mM) |
0.184 |
0.186 | 0.165 | 0.178 | 0.01 |
6 |
|
The non-cytotoxicity may be attributed to flavonoids such as quercetin-3-O-glucuronide and quercitrin, which scavenge ROS and protect cells from oxidative stress15,17. These phytochemicals also serve as nanoparticle capping agents, reducing membrane interactions and potential oxidative or inflammatory responses. Potential optical interference from AuNPs in the MTT assay is noted, and complementary assays (LDH release, Neutral Red uptake, Annexin V/PI staining) are recommended to confirm viability.
Overall, the A5 formulation, with stable size (66 nm, −14.6 mV) and moderate antioxidant activity, is non-cytotoxic and biocompatible, supporting its safe application in nanobiotechnology. These findings align with other green AuNP systems, such as Zingiber officinale and Azadirachta indica, which demonstrate high cellular viability and low inflammatory potential19,27.
Conclusions
In conclusion, this study demonstrates that Biokesum® is an effective bio-reducing and stabilizing agent for the green synthesis of AuNPs. Its rich phytochemical content, particularly quercetin derivatives, enabled the formation of nanoparticles with defined morphological and optical characteristics. Characterization by UV-Vis, DLS, zeta potential, TEM, and FESEM-EDX showed that sample A5 achieved an optimal balance of particle size (~66 nm), surface plasmon resonance (~530–535 nm), and moderate colloidal stability (−14.6 mV), making it the most promising formulation. The AuNPs exhibited pH-dependent antioxidant activity, with maximal DPPH scavenging at acidic pH (4) and decreased efficacy at neutral and highly alkaline conditions. In contrast, Biokesum® extract and quercetin maintained high activity (>95%), indicating that phytochemical consumption during nanoparticle synthesis limits free radical scavenging. Overall, Biokesum®-mediated AuNPs offer a sustainable, biocompatible platform with potential for mitigating oxidative stress-related disorders. Challenges such as moderate stability and variable antioxidant performance warrant further optimization through surface functionalization, long-term stability assessment, and in vivo validation. Future studies should refine synthesis parameters, enhance stability using polymeric or biomolecular coatings, and evaluate pharmacological safety and efficacy in relevant biological models.
Acknowledgement
We would like to express our gratitude to the Universiti Kebangsaan Malaysia, Selangor, Malaysia for the Geran Galakan Penyelidik Muda (GGPM) code GGPM-2023-039 and Geran Galakan Fakulti Sains Kesihatan NN-2025-037.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Sies H, Berndt C, Jones DP. Oxidative Stress. Annu Rev Biochem. 2017 Jun 20;86:715-748. doi: 10.1146/annurev-biochem-061516-045037.
CrossRef - Pham-huy, L. A.; He, H.; Pham-huy, C. Free Radicals , Antioxidants in Disease and Health. Int J Biomed Sci. 2008, 4 (2), 89–96.
CrossRef - Muscolo, A.; Mariateresa, O.; Giulio, T.; Mariateresa, R. Oxidative Stress: The Role of Antioxidant Phytochemicals in the Prevention and Treatment of Diseases. Int. J. Mol. Sci. 2024, 25, 3264. https://doi.org/10.3390/ijms25063264.
CrossRef - Ge X, Cao Z, Chu L. The Antioxidant Effect of the Metal and Metal-Oxide Nanoparticles. Antioxidants. 2022; 11(4):791. https://doi.org/10.3390/antiox11040791
CrossRef - Singh, S. Nanomaterials Exhibiting Enzyme-Like Properties ( Nanozymes ): Current Advances and Future Perspectives. Frontiers in Chemistry, 2019, 7 (February), 1–10. https://doi.org/10.3389/fchem.2019.00046.
CrossRef - Dykman, L. A.; Khlebtsov, N. G. Immunological Properties of Gold Nanoparticles. Chem. Sci. 2017, 8 (3), 1719–1735. https://doi.org/10.1039/c6sc03631g.
CrossRef - Pattoo, T. A. Flora to Nano : Sustainable Synthesis of Nanoparticles via Plant-Mediated Green Chemistry. Plant Science Archives, 2023, 8(1). https://doi.org/10.51470/PSA.2023.8.1.12
CrossRef - Christapher, P.; Parasuraman, S.; Christina, J.; Asmawi, M. Z.; Vikneswaran, M. Review on Polygonum Minus. Huds, a Commonly Used Food Additive in Southeast Asia. Pharmacognosy Res. 2015, 7 (1), 1–6. https://doi.org/10.4103/0974-8490.147125.
CrossRef - Seimandi, G.; Álvarez, N.; Stegmayer, M.I.; Fernández, L.; Ruiz, V.; Favaro, M.A.; Derita, M. An Update on Phytochemicals and Pharmacological Activities of the Genus Persicaria and Polygonum. Molecules 2021, 26, 5956. https://doi.org/10.3390/molecules26195956.
CrossRef - Al-Snafi, P. D. A. E. A Review on Fagopyrum Esculentum: A Potential Medicinal Plant. IOSR Journal of Pharmacy (IOSRPHR) 2017, 07 (03), 21–32. https://doi.org/10.9790/3013-0703012132.
CrossRef - Williams, M.; ND; RH (AHG); Jenks, A. Kesum Persicaria Minor Plant Family: Polygonaceae. HerbalGram, 2024, 139, 6–12.
- Chinnappan, S. M.; George, A.; Choudhary, Y. K.; Godavarthi, A.; Teng, C.-L.; Jin, W. H. In Vitro and In Vivo Investigations on Anti-Hyperuricemic Activity of Commercial Preparations of Persicaria Minor (Biokesum®) and Eurycoma Longifolia (Physta®). Current Traditional Medicine 2022, 9 (5). https://doi.org/10.2174/2215083809666221213142127.
CrossRef - Arumugam, B.; Manaharan, T.; Heng, C. K.; Kuppusamy, U. R.; Palanisamy, U. D. Antioxidant and Antiglycemic Potentials of a Standardized Extract of Syzygium Malaccense. Lwt 2014, 59 (2P1), 707–712. https://doi.org/10.1016/j.lwt.2014.06.041.
CrossRef - Popovici, L.-F.; Brinza, I.; Gatea, F.; Badea, G.I.; Vamanu, E.; Oancea, S.; Hritcu, L. Enhancement of Cognitive Benefits and Anti-Anxiety Effects of Phytolacca americana Fruits in a Zebrafish (Danio rerio) Model of Scopolamine-Induced Memory Impairment. Antioxidants, 2025, 14, 97. https://doi.org/10.3390/antiox14010097.
CrossRef - Azeem, M.; Hanif, M.; Mahmood, K.; Ameer, N.; Chughtai, F. R. S.; Abid, U. An Insight into Anticancer, Antioxidant, Antimicrobial, Antidiabetic and Anti-Inflammatory Effects of Quercetin: A Review. Polymer Bulletin 2023, 80 (1), 241–262. https://doi.org/10.1007/s00289-022-04091-8.
CrossRef - Hai, Y.; Zhang, Y.; Liang, Y.; Ma, X.; Qi, X.; Xiao, J.; Xue, W.; Luo, Y.; Yue, T. Advance on the Absorption, Metabolism, and Efficacy Exertion of Quercetin and Its Important Derivatives: Absorption, Metabolism and Function of Quercetin. Food Front. 2020, 1 (4), 420–434. https://doi.org/10.1002/fft2.50.
CrossRef - Alharbi, H. O. A.; Alshebremi, M.; Babiker, A. Y.; Rahmani, A. H. The Role of Quercetin, a Flavonoid in the Management of Pathogenesis Through Regulation of Oxidative Stress, Inflammation, and Biological Activities. Biomolecules 2025, 15 (1). https://doi.org/10.3390/biom15010151.
CrossRef - Ghosh, S. K.; Pal, T. Interparticle Coupling Effect on the Surface Plasmon Resonance of Gold Nanoparticles: From Theory to Applications. Chem. Rev. 2007, 107 (11), 4797–4862. https://doi.org/10.1021/cr0680282.
CrossRef - Fouda, A.; Eid, A. M.; Guibal, E.; Hamza, M. F.; Hassan, S. E. D.; Alkhalifah, D. H. M.; El-Hossary, D. Green Synthesis of Gold Nanoparticles by Aqueous Extract of Zingiber Officinale: Characterization and Insight into Antimicrobial, Antioxidant, and In Vitro Cytotoxic Activities. Applied Sciences (Switzerland) 2022, 12 (24). https://doi.org/10.3390/app122412879.
CrossRef - Huang, X.; Devi, S.; Bordiga, M.; Brennan, C. S.; Xu, B. Phenolic Compounds Mediated Biosynthesis of Gold Nanoparticles and Evaluation of Their Bioactivities: A Review. Int. J. Food Sci. Technol. 2023, 58 (4), 1673–1694. https://doi.org/10.1111/ijfs.16346.
CrossRef - Azam, A. “A Review of Plant-Based Green Nanoparticle Synthesis: Mechanisms, Metals, and Applications in Nanotechnology.” Revista Electronica De Veterinaria 2024, 25 (1), 3576–3584. https://doi.org/10.69980/redvet.v25i1.1657.
CrossRef - dos Santos Corrêa, A.; Contreras, L. A.; Keijok, W. J.; Barcelos, D. H. F.; Pereira, A. C. H.; Kitagawa, R. R.; Scherer, R.; de Oliveira Gomes, D. C.; da Silva, A. R.; Endringer, D. C.; de Oliveira, J. P.; Guimarães, M. C. C. Virola Oleifera-Capped Gold Nanoparticles Showing Radical-Scavenging Activity and Low Cytotoxicity. Materials Science and Engineering C 2018, 91 (May), 853–858. https://doi.org/10.1016/j.msec.2018.06.027.
CrossRef - María, A.; Nogales, J. A Rigorous Exploration of the Plexcitonic Phenomena: Hybridized Light-Matter States Using Gold Nanoparticles and J-Aggregates, UNIVERSITY OF THE BASQUE COUNTRY, SPAIN, 2024, Vol. 2024.
- Pratap, D.; Soni, S. Review on the Optical Properties of Nanoparticle Aggregates Towards the Therapeutic Applications. Plasmonics 2021, 1495–1513. https://doi.org/10.1007/s11468-021-01443-4.
CrossRef - Dissanayake, K.; Midekessa, G.; Godakumara, K.; Ord, J.; Viil, J.; La, F.; Kopanchuk, S.; Rinken, A.; Andronowska, A.; Bhattacharjee, S.; Rinken, T.; Fazeli, A. Zeta Potential of Extracellular Vesicles : Toward Understanding the Attributes That Determine Colloidal Stability. ACS Omega, 2020. https://doi.org/10.1021/acsomega.0c01582.
CrossRef - Baek, W.; Chang, H.; Bootharaju, M. S.; Kim, J. H.; Park, S.; Hyeon, T. Recent Advances and Prospects in Colloidal Nanomaterials. JACS Au 2021, 1 (11), 1849–1859. https://doi.org/10.1021/jacsau.1c00339.
CrossRef - Gopalakrishnan, V.; Singaravelan, R. Enhanced Antidiabetic and Antioxidant Properties of Gold Nanoparticles Green Synthesized Using Blossom Extract of Azadirachta Indica: In Vitro Studies. Inorg. Chem. Commun. 2023, 158 (pt 2), 111609. https://doi.org/10.1016/j.inoche.2023.111609.
CrossRef - Kumar, B.; Smita, K.; Angulo, Y.; Debut, A.; Cumbal, L. China Rose/Hibiscus rosa-sinensis Pollen-Mediated Phytosynthesis of Silver Nanoparticles and Their Catalytic Activity. J. Compos. Sci. 2022, 6, 322. https://doi.org/10.3390/jcs6110322.
CrossRef - Zhang, J.; Cui, Y.; Zhou, T.; Wang, J.; Zhu, Z.; Liu, F.; Xiao, G. Development of Certified Reference Materials of Trace Elements in Gold Solution ( GBW02797 – GBW02800 ). Accreditation and Quality Assurance 2023, 28 (6), 299–310. https://doi.org/10.1007/s00769-023-01557-8.
CrossRef - Kellil, A.; Grigorakis, S.; Loupassaki, S.; Makris, D.P. Empirical Kinetic Modelling and Mechanisms of Quercetin Thermal Degradation in Aqueous Model Systems: Effect of pH and Addition of Antioxidants. Appl. Sci. 2021, 11, 2579. https://doi.org/10.3390/app11062579
CrossRef - Jurasekova, Z.; Domingo, C.; Garcia-Ramos, J. V.; Sanchez-Cortes, S. Effect of PH on the Chemical Modification of Quercetin and Structurally Related Flavonoids Characterized by Optical (UV-Visible and Raman) Spectroscopy. Physical Chemistry Chemical Physics 2014, 16 (25), 12802–12811. https://doi.org/10.1039/c4cp00864b.
CrossRef
Accepted on: 20 Feb 2026
Second Review by: Dr. Sara R. A.
Final Approval by: Dr. Tanay Pramanik

















