Chemical Speciation and Bioavailability of Nutrients in Soils Under Different Management Practices
1Department of Agricultural Chemistry and Soil Science, R. B. S. College Bichpuri, Agra (Dr. B. R. A. University, Agra), U. P., India
2Department of Soil Science and Agricultural Chemistry, Bihar Agricultural University-Irrigation Research Station, Bikramganj, Rohtas, (Bihar), India.
3Krishi Vigyan Kendra Kendra , Saraiya, Muzaffarpur, Bihar Under Dr. Rajendra Prasad Central Agricultural University, Pusa, Samastipur, Bihar, India
4Department of Microbiology, Punjab Agricultural University, Ludhiana, Panjab, India.
5Bihar Agricultural University, Sabour, Bhagalpur, Bihar, India
6 College of Agriculture (OUAT), Chiplima, Sambalpur, India
7School of Agriculture Sciences K.R Mangalam University Sohna Gurugram, Haryana, India
8On Farm Research Centre, Onattukara Regional Agricultural Research Station, Kayamkulam, Kerala Agricultural University, Kerala, India
Corresponding Author E-mail: beerendrasoil@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/420124
ABSTRACT:The bioavailability of essential nutrients and the mobility of contaminants in soil are governed not by their total concentrations but by their specific chemical forms—their speciation. This review examines the intricate dynamics of trace metals (iron, zinc, copper) and macronutrients like calcium within the soil matrix, focusing on the master variables of pH, redox potential (Eh), and dissolved inorganic carbon (DIC) that dictate speciation. We analyze how contrasting agricultural management practices—conventional intensive systems versus sustainable approaches—fundamentally alter these biogeochemical parameters. Conventional practices, such as high-input ammonium fertilization and intensive tillage, drive acidification, deplete exchangeable calcium, and can induce aluminum toxicity, while irrigation with bicarbonate-rich water promotes calcite precipitation and micronutrient deficiency. Conversely, sustainable practices like liming, organic amendment application, and conservation tillage work to buffer pH, enhance soil organic matter (SOM), and increase cation exchange capacity (CEC). These practices stabilize beneficial nutrient speciation, fostering organo-mineral complexes and chelation processes that optimize the narrow window between nutrient deficiency and toxicity. By integrating principles of chemical speciation, this synthesis argues for a paradigm shift in soil fertility management: from a focus on bulk nutrient inputs to the strategic "engineering" of soil chemistry. This approach is critical for enhancing nutrient use efficiency, mitigating environmental risks from heavy metals, building long-term soil health, and achieving sustainable agricultural productivity in the face of global food security and climate challenges.
KEYWORDS:Bioavailability; Chelation; Chemical Speciation; pH Buffering; Soil Biogeochemistry
Introduction
The Global Imperative for Sustainable Soil Fertility Management
The global food security is based on the narrow and alive mantle of soil that is spread upon the terrestrial surface of the earth. This essential commodity faces unprecedented stress due to an amalgamation of forces: a human population of, by 2050, projected to reach 10 billion, requiring a corresponding boost in food output; the growing effects of climate change1 in the form of changed rainfall patterns, higher occurrence of extreme weather patterns, and an ever-growing decline in soil quality; and the inefficient consumption of major agricultural resources, especially phosphate rock and nitrogen-based dungurhins,2,3 which are energy intensive to manufacture. In the past, the management of soil fertility was based on a paradigm that is linear input-based, with macronutrients (Nitrogen, Phosphorus and Potassium) being the major drivers of yield. Although it had been effective in the Green Revolution in the short-term, it has frequently overlooked the ecological complexity of the holistic ecology of the soil system, causing widespread deficits in the micronutrients, loss of soil organic matter, acidification, salinization and contamination. The result is a vicious cycle of diminishing returns with larger and larger fertilizer inputs giving smaller and smaller productivity returns and at the same time reducing the long-term biological health and buffering capacity of the soil. In this way sustainable soil fertility management is a radical [4shift in this linear model to the cyclical, systems-based model. It focuses on the development and sustainability of the natural ability of the soil to provide nutrients, healthy plant growth and environmental fluxes. This necessity is not only agronomic, but is inseparably connected with the United Nations Sustainable Development Goals (SDGs) Zero Hunger (SDG 2), Clean Water and Sanitation (SDG 6), Climate Action (SDG 13), and Life on Land (SDG 15). Sustainable management aims at ensuring efficiency in nutrient utilization, reduction of environmental externalities, like greenhouse gas emissions and eutrophy of water bodies, and development of resilient agroecosystems which can absorb climatic shocks. At the core of this transition is a more detailed, more sophisticated conceptualization of the biogeochemistry of the soils, beyond the simple approaches of quantifying the total nutrient content of the soils to a more dynamic view of nutrient speciation, mobility and ultimate bioavailability to plants and soil microbes. This change of opinion is particularly urgent when it comes to such essential trace metals as iron (Fe), zinc (Zn), and copper (Cu), the reactions in which occur in a complicated and sensitive chain of chemical reactions, but not just through bulk concentration.3,4
The Paradox of Trace Metal Deficiency and Toxicity
The agronomic and ecological problem of trace metals is summed up in a basic paradox the same elements that are essential to the growth of plants in minute amounts are dangerous toxins at only a little greater concentrations.5,6 This gives an extremely small “range of bioavailability” within which the best plant health can be found. Micronutrient deficiency, especially that of Zn and Fe is a common constraint to soil production of crops in the world, with more than half of the land areas used to grow cereals worldwide being affected by it.7 A typical symptom of high-pH calcareous soils, which includes interveinal yellowing of new leaves, is known as iron chlorosis making large portions of agricurally valuable soil unproductive despite having high total Fe concentration. Both intensive soils and high-phosphorus soils, as well as calcareous soils, are widespread in zinc deficiency, which slows the growth of plants and decreases their production and nutritional value.8,9 Copper deficiency, which is less pronounced, may be serious on organic soils and very weathered sandy soils. On the contrary, trace metal toxicity is a great threat to acidic soil, or in regions where mining and industry are evident, or where prolonged use of metal-containing sewage sludges, animal manures, or fungicides (e.g., fungicides containing copper in vineyards and orchards) has caused dangerous levels. Free ionic Cu2+, which is toxic, can cause severe root membrane damage, microorganism inhibition, and plant metabolism. This is the duality of deficiency and toxicity, which is not necessarily a characteristic of the total quantity of the metal in the soil, but of its chemical form or speciation.10 A soil can have large amounts of total Fe, but the plants can die of inadequate bioavailable species. A different soil can be having only moderately high total Cu, yet, when conditions are favorable to the release of free Cu2+ ions, toxicity may then occur. This paradox highlights the severe weakness of conventional soil analyses which determine the total or generally available (through strong extractants) levels of metals. These measurements do not always predict well plant uptake or phytotoxicity since they do not measure the dynamic equilibrium between various metal species of the soil solution and soil solid. To solve this paradox, there is the need to switch to a more speciation-aware system, where management practices are used to vary metal chemistry, away to the extremes of deficiency and toxicity, to the best region of its bioavailability.
Core Concepts: Chemical Speciation, Bioavailability, and Chelation
A clear understanding of three interrelated central concepts, chemical speciation, bioavailability, and chelation, is required to manoeuvre through the paradox of trace metals.11 The specific form, oxidation state and molecular environment in which an element appears in a system is called the chemical speciation. In the case of trace metals in soil, it includes a very large number of species: free hydrated ions ( e.g. Fe3+(aq), Zn2+(aq), Cu2+(aq) ), inorganic complexes ( e.g. Fe(OH)2 +, ZnCl+, CuCO3(aq)) and organic complexes of different strengths ( e.g. metal-citrate, metal-fulvate ) as well as adsorbed to the surfaces of clays, oxides or organic matter. It is different to total concentration which is a bulk analysis of the amount of all forms of the element present regardless of their solubility and reactivity.12 The most important understanding is that, it is almost entirely the speciation, and not the total concentration, of a metal, which determines the biological and environmental behavior of a metal, its mobility, toxicity, and nutritional value. One gram of Fe tied up in an insoluble crystalline oxide mineral is ecologically and nutritionally insignificant in contrast to one microgram of soluble Fe3+-siderophore complex.13
The concept of bioavailability is functional and refers to the availability or the amount of a nutrient that can be absorbed and absorbed by a living organism within a given period of time. In the case of plants, it is not a pool but a continuum, which is characterized by kinetic and dynamic processes. The path of the metal starts with the metal in the soil solution, the immediate liquid phase around soil particles and roots. Species will only be accessible to uptake in this solution. Nevertheless, the soil solution is in dynamic equilibrium with the “labile pool” -metal species loosely attached to solid surfaces which can be replenished easily as roots absorb nutrients. A non-labile pool is comprised of deeper reserves in the mineral structures or well-bound complexes. Bioavailability therefore entails sequential: Desorption/ dissolution of the solid phase into the soil solution, Diffusion as well as mass transportation of the soluble species to the root surface, Uptake by particular transport proteins in the root plasma membrane, and 4) Internal translocation and use in the plant. Anything that influences these processes (including soil pH, moisture, temperature, root structure, microbial activity, etc.) has a direct impact on bioavailability.15
At this point, the phenomenon of chelation comes into existence as a trace metal master key. A particular form of complexation is called chelation, in which a organic molecule, the ligand or the chelating agent, binds a central metal ion in a number of coordinate covalent bond forming a ring-like structure called a chelate (named after the Greek word chele, meaning claw). The essence of this process is to change the properties of the metal. Chelation elevates the water-solubility of the metal ion many folds, blocking the precipation of the insoluble hydroxides (particularly of Fe3+, which can occur in alkaline soils) or phosphates. It protects the charged metal ion making it less reactive to sites of soil adsorption and thereby increasing its mobility and persistence in the soil solution. More importantly, it is also capable of shielding the plant against toxicity by regulating the level of highly reactive free metal ion. There are two main types of Chelating agents, namely synthetic (e.g., EDTA, DTPA, EDDHA) and natural ones (e.g., organic acids such as citrate, microbial siderophores, plant-derived phytosiderophores, and humic/fulvic compounds).16,17 Highly stable synthetic chelates are designed and utilized as effective fertilizer additives to rectify acute deficiencies. Natural chelates are part and parcel of the ecological nutrient cycling system in the soil, which is made by roots and microbes to act as active scavengers and mobilize the metals. The interactions of these chelating agents with the soil matrix and physiology of plants reside at the core of the contemporary, sustainable trace metal management.18,19
Focus Metals: Physiological Roles of Fe, Zn, and Cu in Plants
The choice of iron (Fe), zinc (Zn), and copper (Cu) as metals of interest is not random, as these are the most distinctive and non-interchangeable metals in plant biochemistry, and they have general limitations in bioavailability in soils, as well as influence on crop yield and human diet. One of the most important elements in plant metabolism is iron, which is mainly because of its reversible redox reactions between Fe3 (ferric) and Fe2 (ferrous). This is what makes it a necessary part of electron transport chains. It is a core element of heme groups, including those within the cytochromes of photosynthesis and respiration and non-heme iron-sulfur (Fe-S) clusters of important enzymes such as ferredoxin, nitrite reductase and succinate dehydrogenase. Most famously,20,21 Fe is needed in chlorophyll synthesis but iron does not belong to the chlorophyll molecule itself its loss has a direct effect on chloroplast development resulting in chlorosis. Plants have developed complex, genetically controlled mechanisms (Strategy I in dicots and Strategy II in graminaceous grasses) of obtaining Fe, frequently by active acidification of the rhizosphere and exudation of chelating compounds or reductants to liberate insoluble soil Fe.22
Zinc plays a major role as a structural and catalytic element of an enormous number of enzymes. It is a major subunit of more than 300 enzymes in all the major metabolic pathways including RNA polymerase, carbonic anhydrase, superoxide dismutase (Cu/Zn-SOD), and alcohol dehydrogenase.23,24 However, Zn is not involved in redox reactions; it is in the non-redox state Zn 2. It has a structural nature, and it stabilizes the tertiary and quaternary structure of proteins and nucleic acids (zinc finger proteins). It is required in protein synthesis, carbohydrate metabolism, auxin (IAA) metabolism, and membrane integrity. Zn deficiency quickly interferes with these processes, which are shown by slowed growth (in extreme cases, it is called rosetting), decreased internodal extension, and tiny and deformed leaves. Zn nutrition is also associated with plant resilience to abiotic stresses, such as drought and heat, because of its functions in antioxidant defense (via SOD).25,26
Copper, just as Fe, is a redox-active metal, being alternated between the states of Cu+ and Cu2+. This characteristic is utilized in enzymes that are related to essential electron transfer and oxidation-reduction reactions. The most important cuproenzymes are plastocyanin (a component of photosynthetic electron transport in the chloroplast), cytochrome c oxidase (the final enzyme in you mitochondrial respiratory chain), superoxide dismutase ( Cu/Zn-SOD ), and laccase (part of the lignin biosynthesis).27 Cu is thus essential in the energy production, antioxidant provision and cell wall integrity. Deficiency causes chloroses, necrosis of young leaf tips, and hindrance of lignification, wilting and weakening of the shoot. Nevertheless, because of its reactivity.28 Cu is also very toxic in its free ionic state, and it has to be strictly maintained in the cell as a tight homeostasis, often including chaperone proteins and chelation in the plant. The unique but essential roles of Fe, Zn, and Cu render their sufficient bioavailability an uncompromising condition to the health, production, and nutritional values of plants. Their common propensity to constitute insoluble precipitates or powerful complexes in soils especially at the neutral to alkaline pH poses a common agronomic problem. The role of various soil management regimes in controlling the complex speciation chemistry of these metals and especially the use of chelation approach in understanding the relationship between the various regimes with respect to the generation of the next generation of sustainable soil fertility methods is therefore paramount in the development of the next generation of sustainable soil fertility methods.29 This investigation is the main thesis of this work, which attempts to mediate the gap between basic soil chemistry and regenerative agriculture that is useful in the real world.
Chemical speciation is a fundamental principle of environmental chemistry, geochemistry, and toxicology and is defined as the particular form, oxidation state or molecular geometry in which an element is present in a particular system. The specific determination and measurement of the various chemical and physical manifestations of an element that make up its total concentration are what contribute to overall concentration. This is an essential distinction since the mobility, bioavailability, reactivity as well as the toxicity of an element is nearly solely reliant on its speciation, rather than its total concentration. As an example, chromium( VI ) is a very strong carcinogen and very soluble in water whereas chromium(III) is a necessary nutrient, and it readily precipitates. On the same note, inorganic mercury (Hg2+) is also toxic but its methylated counterpart, methylmercury is a neurotoxic that is disastrous and bioaccumulates along the food chain. In this way, the speciation analysis goes beyond the simplistic question of how much to consider the much more important questions of in what form, with what binding, and what potential consequences.30
There are three dimensions of speciation; chemical forms, physical phases and interfacial interactions. Chemical forms encompass the oxidation state of the element (e.g. Fe2 + vs. Fe 3+), its molecular structure as simple ions, complexes or organometallic compounds (e.g. Cd2 +, CdCl +, (CH3)2Cd) as well as its binding in mineral lattices or on surfaces. The compartment in which the species exists is described by physical phases: the dissolved phase of aqueous solution (truly dissolved ions and complexes), the solid phase (as discrete minerals, coprecipitated inclusions or sorbed species), the colloidal phase (nanoparticles and macromolecules which are neither truly dissolved nor filterable), and the gas phase (e.g. dimethylselenide). The interactions between these forms and phases are dynamic, i.e. dissolution, precipitation, adsorption, desorption, complexation, redox transformation etc. control the distribution and behavior of the element. Finally, speciation will establish the contribution that an element makes to biogeochemical cycles, the possible nutrient or contaminant, and the relationship of the element to living organisms.31,32
Master variables governing speciation: pH, Eh, ionic strength, and biological activity
Speciation of an element within any natural or engineered system is not fixed, but is dynamic and is controlled by a combination of master environmental variables. These variables are considered to be primary controls, shifting the equilibrium and driving kinetic processes to determine dominant species pH (The Master of Solubility and Complexation) The concentration of hydrogen ions is perhaps the most powerful single parameter. pH directly influences acid-base reactions, including the protonation of ligands (e.g., CO32-, PO43-, organic acids) and the hydroxylation of metal cations. This, consequently, controls the solubility of minerals (e.g.[ Al and Fe hydroxides are very insoluble at neutral pH), the adsorption processes (most oxide surfaces contain charge dependant on pH) and the aqueous complexes. To illustrate, in the presence of low pH, the cadmium ion is mostly in the form of free Cd 2 +, which is very bioavailable. It hydrolyzes to species such as CdOH+ as pH rises and can harm the solids of carbonates or bind itself more firmly to the particles, which severely diminishes its mobility. Eh (The Master of Electron Flow – Redox Potential): The oxidation state of a multivalent element that is stable is determined by the redox potential which is a measure of how readily the system accepts or donates electrons. Elements such as iron, manganese and sulfur are reduced (e.g. Fe3 + to Fe 2 +, SO4 2 – to H2S) in anoxic environments (low Eh) such as waterlogged soils or sediments. This dissolution may enhance solubility (as in Fe and Mn) but may also cause the formation of very insoluble sulfide mineral (e.g., CuS, HgS, PbS), which has the effect of immobilizing toxic metals. Redox cycling of elements is a major cause of biogeochemical changes, closely associated with the process of microbial respiration. Ionic Strength The overall concentration of ions in solution has an impact on the activity coefficients, which actually alters the effective concentration or the activity of dissolved species. When there is a high ionic strength of water (e.g. in seawater, in brines) shielding of the electrostatic interactions occurs, and may raise the solubility of particular minerals and change the stability of ion pairs and complexes. It also influences the thickness of electrical double layer surrounding colloidal particles, among others, which influences aggregation and adsorption behaviour. Models need to correct ionic strength on the basis of models such as Debye-Huckel equation in order to predict speciation appropriately. Living organisms Biological agents are powerful agents of change of speciation. Microorganisms use redox reactions to generate energy (e.g. dissimilatory iron reduction, sulfate reduction), mediate the methylation/demethylation of elements (e.g. Hg, As), and secrete organic ligands (siderophores, organic acids) which complex and solubilize nutrients. Plants and fungi are able to modify rhizosphere pH and redox to allow the uptake of nutrients. This biotic aspect adds considerable kinetic control and complexity which can often give rise to micro-environment with a strikingly different speciation than that of the bulk system.
These master variables are not independent. They are intensely coupled. The microbial respiration uses oxygen and reduces Eh to generate CO2, which reduces pH. Redox equilibria can be altered by the alteration of pH. Thus, the interactive controls must be considered in understanding the systems based approach to speciation.
Material and Methods
Analytical and Modeling Approaches for Speciation Studies
Determining chemical speciation is analytically challenging, as many techniques risk altering the delicate equilibria during sample handling. The field therefore relies on a complementary triad of approaches: operational wet chemical methods, direct spectroscopic techniques, and computational thermodynamic modeling.
Spectroscopic Techniques (e.g., XAFS, NMR)
Spectroscopic approaches provide first-order, non-destructive (or destructive to a minimum), and in many cases, element-specific information on the occurrence of molecular-scale speciation in the native environment, with reduced artifacts. The most potent member of the environmental speciation repertoire is arguably X-ray Absorption Fine Structure (XAFS) spectroscopy (including XANES, or X-ray Absorption Near Edge Structure, and EXAFS, or Extended X-ray Absorption Fine Structure). XANES can give accurate data on the oxidation state of the element (e.g. determining the fraction of As(III) to As(V) in a soil), whereas EXAFS provides information on the number, type, and distance of the other atoms surrounding the central element quantitatively. This enables researchers to observe whether metal has been adsorbed as an inner-sphere complex on an mineral oxide, is a part of a mineral lattice or has been attached to sulfur on organic matter. The NMR spectroscopy is incomparable in studying the speciation of some nuclei (e.g., 13C, 31P, 119Sn, 113Cd, 199Hg). It is able to detect individual organic and organometallic compounds in complex matrices. An example is 199Hg NMR that is able to distinguish between methylmercury, ethylmercury and inorganic mercury complexes. Surface-sensitive oxidation state data can be obtained using other methods such as X-ray Photoelectron Spectroscopy (XPS) and Fourier Transform Infrared (FTIR) Spectroscopy can be used to identify functional groups in the process of metal binding. Although the spectroscopic methods offer absolute data on the speciation, they can be expensive to perform due to necessary special equipment, concentrated samples, and complicated analysis and do not operate effectively on frequent monitoring or low-concentration samples.
Thermodynamic Equilibrium Modeling (e.g., PHREEQC, Visual MINTEQ)
The Thermodynamic equilibrium modeling is an effective predictive and interpretative model of speciation. These computer packages (PHREEQC, Visual MINTEQ, MINEQL+, Geochemist Workbench) find solutions to a group of mass-action and mass-balance equations with the assistance of large data bases of thermodynamic constants (formation constants, solubility products, etc.). The model computes the equilibrium distribution of all the possible aqueous complexes and the saturation state of a given water composition (major ions, pH, Eh, temperature) and the distribution of species on adsorbing surfaces (when surface complexation models are added). The power of modeling is in that it allows combining the master variables, performing what-if analysis (e.g., the impact of lime addition on the metal mobility and pH), and determining the important complexes that govern the behavior. As an example, a model may show that lead mobility in a given piece of groundwater is no longer determined by the free Pb2+, but by the neutral complex PbCO3(aq), and that the system is undersaturated with respect to cerussite (PbCO3(s)).32
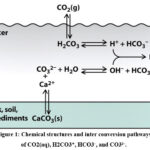 |
Figure 1: Chemical structures and inter conversion pathways of CO2(aq), H2CO3*, HCO3⁻, and CO3²⁻. Click here to View Figure |
Role as pH buffer and master variable for trace element mobility
The major natural buffer system of most soils and aquatic environments is dissolved inorganic carbon (DIC) which occurs as carbonic acid (H2CO3), bicarbonate (HCO3-), and carbonate (CO32) forms. This carbonate equilibrium is the main stabilizer of PH as in this reaction cascade CO2(aq) + H2O ⇌ H2CO3 ⇌ H+ + HCO3– ⇌ 2H+ + CO32- this equilibrium can consume or release protons. With the addition of acid, the equilibrium shifts to H2CO3 and CO2(g), taking up excess H +. Protons are released during the addition of a base to the HCO3- to create CO32-, which opposes an increase in pH. The DIC system indirectly but significantly controls the solubility, speciation, and mobility of essentially all trace elements by controlling pH. Any movement of one unit of pH may alter the amount of a free metal ion by orders of magnitude. Thus, the DIC system becomes the master rheostat of the geochemical and biological reactions by serving as the most ubiquitous pH buffer on Earth, on which all other speciation reactions take place.
Influence on micronutrient (e.g., Fe, Zn) and phytotoxic element (e.g., Cd) Solubility
The pH controlled by the DIC system obtains a predominant influence over the behavior of trace elements in two major ways, which are hydrolysis and adsorption. In the case of micronutrients which are most crucial such as Fe and Zn, the solubility is maximized at low pH. Under acidic environments, these metals are present as soluble free ions (Fe2+, Zn2+).33,34 But they are hydrolyzed out into less soluble hydroxides (e.g., Fe(OH)3, Zn(OH)2 ) as pH increases (usually regulated by the DIC system) and adsorb to the surface of the clay minerals and iron/aluminum oxides, becoming unavailable to the plants. This forms the archetypal micronutrient deficient issue in calcareous alkaline soils. Plants have also developed intricate mechanisms such as the release of root exudates to acidify the rhizosphere, to mobilize these metals. On the other hand, with phytotoxic elements, such as cadmium (Cd), an increase of pH favours immobilization. Cd is very mobile and bioavailable in acidic soils in the form of Cd2+ and this is a major risk to the food-chain. The higher the pH, the more Cd precipitates into hydroxy and carbonate complexes and attaches itself to the surfaces of the particles, the slower its mobility and bioavailability become. Accordingly, pH control through supplements such as lime is a foundational intervention measure that can be applied to reduce Cd pollution in the agricultural industry. The critical lever is therefore the DIC-pH buffer system, which forms a very fine balance between the adequate solubility of nutrients and the reduction of the mobility of toxic elements.35
Precipitation-Dissolution Dynamics of Carbonate Minerals (CaCO₃, MgCO₃)
Precipitation and dissolution of carbonate minerals (mainly calcite (CaCO3) and dolomite (CaMg(CO3)2): these are dynamic processes which are inherently connected to the DIC system and pH. Their formation is a significant long-term inorganic carbon trap and controller of water hardness. Dissolution takes place when water that is undersaturated with carbonates, normally because of high levels of CO2 produced by soil respiration (or by the atmosphere), becomes acidified: CaCO3(s) + H2CO3(aq) = Ca2 + + 2HCO3–. In this reaction, the pH is buffered in the mildly alkaline (7.5-8.5) range and Ca2+ and Mg2+ ions are emitted. Precipitation, on the other hand, takes place when either CO2 is released (e.g. in a spring, or when evaporating into a solution), or ions are concentrated within the solution forcing.36
Calcium is a master macronutrient and structural cation, which occurs in soils in a dynamic continuum of chemical pools each having different species, solubility, and reactivity as summarized in Table 1. The immediately soluble pool, which is dominated by the free Ca2+ ion together with small complexes such as CaSO40 and CaHCO3+ is the direct source of uptake by plants where it can also be leached.37 This pool is continually interacting with the exchangeable pool in which Ca2+ is electrostatically fixed to negatively charged sites of clay minerals and organic matter (OM). This reservoir is a short-term buffer that is very important and restores the soil solution through cation exchange reactions and is measured by the cation exchange capacity (CEC) of the soil. Carbonate pool, which is mainly composed of calcite (CaCO3) and dolomite, is a very large bed in neutral- alkaline soils. The key regulator of soil pH in such systems is its solubility, which dissolves in acidic environment (CaCO3 + 2H+ → Ca2+ + H2O + CO2) to release Ca2+ and counter acidification. The dissolution and the precipitation of carbonates which are controlled by the DIC system and the CO2 partial pressure are basic processes that govern the availability of calcium, as well as, as was mentioned above, the co-precipitation and immobilization of trace elements. Conversely, the major mineral pool (e.g. apatite,38 Ca-feldspars), is a long-term, recalcitrant resource, which releases Ca2+ slowly due to the chemical weathering-process, which is further initiated by the acidity and complexation. Lastly, the organically complexed pool is Ca attached to the humic and fulvic acids.39 This pool is variable but may be large with Ca being released by microbial mineralization of OM or by remaining chelated by soluble complexes of organic matters which may increase mobility. Knowledge of the distribution and kinetics of these pools is important to manage the soil fertility (e.g. when to apply lime), to understand the fate of the contaminants and to model the long-term biogeochemical cycling because the mobility and bioavailability of calcium -and the elements that it controls- are determined by its changing speciation.40,41
Table 1: Major Chemical Species and Pools of Calcium in Soils
|
Pool |
Dominant Species/Phase | Solubility / Lability | Key Processes |
| Solution | Ca²⁺ (aq), CaSO₄⁰, CaHCO₃⁺, CaOH⁺ | High, Directly available |
Plant uptake, leaching |
|
Exchangeable |
Ca²⁺ on clay/OM exchange sites | Moderately labile (CEC dependent) | Cation exchange, buffering |
| Carbonate | Calcite (CaCO₃), Dolomite (CaMg(CO₃)₂) | pH-dependent (solubility increases with acidity) |
Precipitation/dissolution, pH buffering |
|
Primary Minerals |
Apatite (Ca₅(PO₄)₃(OH)), Plagioclase (Ca-feldspar) | Very low, slow weathering | Long-term supply via weathering |
| Organically Complexed | Ca bound to humic/fulvic acids | Variable, often labile |
Mineralization, chelation |
Result and Discussion
Conventional Intensive Agriculture: Accelerated Cycles and Degradation
Intensive farming, which is conventional, has far-reaching impacts on soil biogeochemistry because it disrupts natural equilibria, and has severe impacts on calcium pattern and movement of trace elements. Ammonium (NH4+) source and urea-based high-input nitrogen fertilization triggers a deep-rooted cascade of acidification[42]. When applied, urea breaks down to produce ammonium, which is then further converted to nitrate (NO3–), releasing two protons (H+) per mole by soil microbes. This persistent acidification directly mobilizes the base cations particularly the Ca2+ cation in the exchange complex and carbonate pool. The negatively charged nitrate anion, which has now become the dominant ion in the solution, combines with the washed out Ca2+ to help it move down the soil profile (nitrate-induced leaching). This leads to the loss of an important structural ion, soil acidification and the possible release of phytotoxic aluminum (Al3+) in the acidic subsoils.43,44 At the same time, the chemical stress is contrasted by irrigation with high bicarbonate water, typical of arid and semi-arid environments. The high bicarbonate (HCO3–) load elevates the pH and alkalinity of the soil solution. This displacement may cause precipitation of the calcite (CaCO3) because the irrigation water becomes concentrated due to evaporation, which essentially immobilizes the calcium and phosphorus in the insoluble form. In addition, the high PH also greatly decreases the solubility of the micronutrient cations such as iron and zinc, creating deficiency in crops although the overall soil reserves are sufficient- a significant yield constraint in calcareous soils. Last but not least is the process of intensive tillage which fast-tracks the oxidation of soil organic matter (SOM) and releases stored CO2, temporarily reducing soil pH in microsites. It also has the effect of disrupting soil structure in that it breaks apart the very aggregates that were stabilized by Ca2+ which contributes to further erosion and exposes further SOM to decomposition. These practices when combined can form a paradox of simultaneous calcium leaching (through acidification) and calcium immobilization (through calcite precipitation), along with the erosion of the physical and chemical buffering capacity of the soil.45.
Conservation and Sustainable Practices: Enhancing Resilience and Regulation
Conversely, conservation agriculture uses methods that aim to coexist with biogeochemical cycles to improve soil resilience and provide more optimal conditions in terms of nutrient availability and metal stabilization. Multi-functional intervention is the application of organic amendments (manure, compost).46 These materials have a direct addition of calcium in forms that are organically complexed and soluble, and their decay elevates Soil Organic Matter (SOM). SOM increases the cation exchange capacity (CEC) of the soil that forms a bigger pool to store Ca2+ and other nutrients against leaching. Complexing functional groups of humic substances buffer pH as well as have the capacity of sequestration of potentially toxic metals (e.g., Cd, Al), which makes them less phytotoxic. The no-till or direct seeding methods reduce disturbances in the soil maintaining the aggregate structure and enhancing continuous biologically active rhizosphere. The increased biological activity causes raising partial pressure of CO2 (pCO2) in soil pores. This increased CO2 makes carbonic acid, which has the ability to a small degree reduce ph, causing Ca bearing minerals to become dissolved and enhancing the solubility of Fe and Zn, therefore, raising their bioavailability in the otherwise alkaline soils.
Lim practice is a high-profile and important way to treat acid soils. It enhances the level of soil pH by simply incorporating a base (e.g., calcite, dolomite, or quicklime), transforming the adverse impact of the acidified pH. Such a move instantaneously inhibits the activity of harmful Al3+ that gets precipitated as hydroxides and reinstates a positive Ca2+/Al3+ equilibrium within the soil solution. The supplemental calcium refills the exchangeable pool, which contributes to the plant nourishment and physical design of the soil. Lastly, speciation change is a biological force caused by cover cropping. Deep-rooted cover crops (e.g., brassicas, legumes) erode mineral substrates, as they exude protons and organic chelators, gradually releasing Ca and other nutrients of primary minerals. Their roots also form dynamic rhizosphere microsites where exudates change local PH and redox conditions and locally change nutrient and contaminant speciation. When cover crop biomass decomposes, the biomass recycles nutrients such as calcium and adds to the active pool of organic matter which supports the CEC.
Table 2: Comparative Effects of Management Practices on Key Speciation Parameters
|
Management Practice |
Typical pH Shift | Effect on Soil pCO₂ | Effect on [HCO₃⁻] | Effect on Ca²⁺ Pools (Solution/Exchangeable) | Implication for Nutrient Availability |
| Conventional (N-fertilizer) | Decrease (Acidification) | Variable | May decrease in bulk soil | Depletion via leaching |
Mn, Al toxicity; P, Mo, Ca deficiency |
|
Liming (CaCO₃) |
Increase | Short-term increase (dissolution) | Significant increase | Large increase | Corrects acidity, supplies Ca, but can induce Fe/Zn deficiency |
| Organic Amendment | Stabilizes (Buffers) | Significant increase (respiration) | Increases in microsites | Increases & stabilizes via OM complexes |
Enhanced micronutrient chelation; improved overall fertility |
|
Saline Irrigation |
Variable | – | Often increases | Complex (competition with Na⁺, Mg²⁺) |
Ionic strength effects; potential Ca deficiency despite high total Ca |
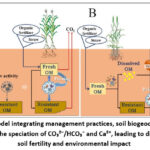 |
Figure 2: Conceptual model integrating management practices, soil biogeochemical processes, and the resulting shifts in the speciation of CO₃²⁻/HCO₃⁻ and Ca²⁺, leading to divergent outcomes for soil fertility and environmental impact |
Implications for Soil Fertility and Environmental Sustainability
A deep understanding of chemical speciation transcends academic interest; it provides the fundamental toolkit for managing soil fertility sustainably and mitigating environmental degradation. By moving beyond total element concentrations to the reactive forms that control bioavailability and mobility, we can design agricultural systems that are both productive and resilient.
Optimizing Nutrient Use Efficiency through Speciation-Based Management
The conventional fertilizer prescriptions that are calculated in terms of total or available nutrient pools are not always efficient and result in a tremendous economic waste and environmental effluence. Speciation-based management can have the same effect on Nutrient Use Efficiency (NUE), just at the chemical hurdles that are targeted to be used as a method of management. An example of this is the fact that, since fixation of phosphorus in acid soils is by adsorption on iron/aluminum oxides, and in alkaline soils by precipitation with calcium, completely different amendment plans (acidulation, humic acid additions, respectively) are predictably determined. On the same note, the treatment of micronutrient deficiencies will need the diagnosis of the problem being low total content or low solubility because of adverse pH or redox conditions. Remedies may then be specific, e.g., soil, or foliar treatment of the chelated metals (e.g. Fe-EDDHA) in calcareous soil, or pH correction by sulfur or lime. This will reduce blanket applications and reduce the cost of inputs and will also ensure the nutrients applied are bioavailable in the root zone and not converted to unavailable species.
Mitigating Environmental Risks: Heavy Metal Mobility, Carbon Sequestration, and Leaching Losses
Speciation science is important in evaluating and reducing environmental risks. In the case of heavy metals (e.g. Cd, Pb, As), the total concentration is not a good measure of danger. The mobile and bioavailable fraction determine the risk. Speciation can be directly structured so as to favor immobile forms: acidifying the pH to enable adsorption and precipitation, incorporating organic matter so as to create stable complexes, or controlling redox to favor sulfide precipitation in wetlands. The relationship between soil organic matter (SOM) stability and its interaction with mineral surfaces, as well as multivalent cations, such as Ca2+ and Fe3+/Al3+ is important in regard to its effect on carbon sequestration. Aggregates contain organo-mineral complexes and metal bridges which physically shield SOM against microbial breakdown. Thus, processes which encourage such interactions, like the addition of calcium-rich supplements or the maintenance of the best possible pH to form metal hydroxides, directly increase long-term carbon storage. Lastly, management of leaching losses of nutrients such as nitrate and base cations is undertaken by the speciation of the ions in the vicinity. To reduce the leaching of nitrates, e.g, it is not only necessary to increase the management of N, but also to consider the balance of cations, because the mobility of nitrate is correlated with cations, such as Ca2+. A strong CEC due to the additions of organic matters is a buffer, holding the nutrients in exchangeable forms.
The Role of Speciation in Building Soil Health and Resilience
The ability of soil to be an important living ecosystem is essentially an expression of soil chemical, physical, and biological speciation state. A healthy and strong soil has got a buffered and varied portfolio of speciation. It is chemically stable with a high CEC (saturated with desirable nutrients (high Ca2+, Mg2+) and it can withstand acidification as well as loss of certain nutrients. Physically, the prevailing divalent Ca2+ exchange sites enhances the generation of water-stable aggregates, which provide habitat to microorganisms, enhance water uptake, and erosion.48,49 Biologically This is because both organic and inorganic forms of the bioavailable nutrients should be provided in adequate amounts to support a diverse microbial community which further triggers nutrient cycling and the production of bioactive compounds adjusting the trace element speciation (e.g. via the release of siderophores). Once a soil system is handled in a speciation-oriented manner, it will be able to withstand any form of stressor such as drought, contamination or vigorous production. It is capable of freezing immediate contaminant loads, gradually and steadily unloading nutrients when the plants demand them, and stabilizing gas and water exchange with the help of stable structure. Therefore, the need to enhance the optimal speciation is equivalent to establishing the inherent soil ecosystem sustenance.50
Future Research Directions
The emerging understanding of chemical speciation as the key of the master to soil functioning opens a colorful research perspective. Going forward, the science should progress beyond the descriptive science to predictive, mechanistic and applied science. This demands the scaling of bridges–between molecular and landscape between genomics and geochemistry. The following decade of enquiry will be characterised by four interconnected pathways.
The final hotspot in speciation is the rhizosphere where the biological, chemical, and physical gradients meet. The devastating character of the majority of the sampling and the challenge of observing transient and micro-scale processes cripples the current knowledge. Research will be needed in the future, which will focus on in-situ and operando analytical platforms that can trace speciation in real-time in this living, breathing environment. This includes the development of planar optodes and electrochemical sensors of imaging pH, O2 and ion concentrations in micron resolution. The speciation of elements (e.g., Fe, Mn, Zn, As) around living root systems will be enabled by the incorporation of methods such as micro-XANES and micro-XRF (executed on a synchrotron system) into special rhizotron facilities. Moreover, the spatial distribution and conversion of applied nutrients can be visualized using such techniques as laser ablation ICP-MS coupled with isotope tracing. This is aimed at replacing the snapshots of the disrupted systems with live streams of the chemical communication between the roots, microbes, and soil minerals and recording the kinetic pulses of exudation and nutrient uptake.
Conclusion
The discussion has shown that the agriculture management activities are effective contributors to the soil chemistry, which significantly transform the speciation of key master variables such as HCO3- and Ca2+. Traditional intensive methods including ammonium-based fertilization and intensive plowing also tend to initiate acidity and promotes leaching of nitrates, draining soluble and exchangeable Ca2+ pools, and releasing toxic Al3+. On the other hand, balancing irrigation with bicarbonate-containing water may cause alkaline conditions, which will precipitate Calcite and fix Ca2+ and phosphorus causing micronutrient deficiencies. Sustainable processes such as liming and organic amendment directly control these speciation pathways; elevated pH to inhibit Al toxicity, increased dissolved inorganic carbon to buffer the system and exploitation of organic complexes to maintain nutrient cations and improve soil structure. Hence, the realization and active control of chemical speciation is not only an academic exercise but is also a basic condition of the development of intelligent agronomic systems. It gives the mechanistic connection between a management intervention (e.g. adding lime) and its indirect ramifications on soil health and plant growth. Selecting both the type and concentration of ions allows us to address some of the most important trade-offs: improving the phosphorus supply without increasing the mobility of heavy metals, or alleviating the acidity of the soil without causing micronutrient deficiencies. This is the key to the triple aim of increased fertility (recycling bioavailable nutrient pools), improved resource management (reducing fertilizer wastage and leaching), and sustainability (developed resilient and buffered soil ecosystems) because this is a speciation-oriented solution. Control of speciation is, ultimately, control of soil activity.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Bolan, N., Sarmah, A. K., Bordoloi, S., Bolan, S., Padhye, L. P., Van Zwieten, L., … & Siddique, K. H. M. (2023). Soil acidification and the liming potential of biochar. Environmental Pollution, *317*, 120632. https://doi.org/10.1016/j.envpol.2022.120632
- Chen, C., Kuppe, C., Roose, T., & Schnepf, A. (2024). Microscale modelling of nutrient uptake and speciation dynamics in the rhizosphere. Geoderma, *441*, 116757. https://doi.org/10.1016/j.geoderma.2023.116757
- de Souza, I. R. P., de Mello Prado, R., & de Cássia Piccolo, M. (2023). Interaction between silicon and calcium in the alleviation of aluminum toxicity in sorghum plants. Journal of Plant Nutrition and Soil Science, *186*(2), 199-209. https://doi.org/10.1002/jpln.202200309
- Ding, Y., Liu, Y., Liu, S., Huang, X., Li, Z., & Tan, X. (2021). Biochar to improve soil fertility. A review. Agronomy for Sustainable Development, *41*(2), 1-18. https://doi.org/10.1007/s13593-021-00649-2
- Fu, H., Wei, C., & Hu, Q. (2022). In-situ speciation analysis of arsenic in paddy soils using diffusive gradients in thin films (DGT) and X-ray absorption spectroscopy. Science of The Total Environment, *806*, 150659. https://doi.org/10.1016/j.scitotenv.2021.150659
- Ghosh, S., & Saha, R. (2023). Chelate-assisted phytoremediation of heavy metals: Mechanisms, efficiency, and future prospects. Environmental Technology & Innovation, *31*, 103196. https://doi.org/10.1016/j.eti.2023.103196
- Giri, A., Heckathorn, S., Mishra, S., & Krause, C. (2021). Interactive effects of elevated CO2, drought, and warming on soil micronutrient availability. Plant and Soil, *467*(1-2), 417-434. https://doi.org/10.1007/s11104-021-05082-w
- Guppy, C., & McLaren, T. (2023). The role of soil organic matter functional groups in trace metal binding: A critical review. European Journal of Soil Science, *74*(1), e13345. https://doi.org/10.1111/ejss.13345
- Haghnia, G. H., & Maftoun, M. (2021). Chemical speciation and bioavailability of zinc in calcareous soils amended with organic materials. Journal of Plant Nutrition, *44*(7), 1023-1038. https://doi.org/10.1080/01904167.2020.1862198
- Jones, D. L., Cross, P., Withers, P. J. A., DeLuca, T. H., Robinson, D. A., Quilliam, R. S., … & Edwards-Jones, G. (2024). Nutrient stripping: The global disparity between food security and soil nutrient stocks. Global Change Biology, *30*(3), e17223. https://doi.org/10.1111/gcb.17223
- Kätterer, T., Börjesson, G., & Kirchmann, H. (2022). Changes in organic carbon in topsoil and subsoil and microbial community composition after four decades of organic and mineral fertilization. Soil Biology and Biochemistry, *165*, 108517. https://doi.org/10.1016/j.soilbio.2021.108517
- Kopittke, P. M., Menzies, N. W., Wang, P., McKenna, B. A., & Lombi, E. (2020). Soil and the intensification of agriculture for global food security. Environment International, *132*, 105078. https://doi.org/10.1016/j.envint.2019.105078
- Li, M., Wang, S., Tian, X., Zhao, C., & Li, H. (2023). Combined use of synchrotron-based techniques to unravel zinc speciation in the rhizosphere of a hyperaccumulator. Journal of Hazardous Materials, *443*, 130234. https://doi.org/10.1016/j.jhazmat.2022.130234
- Li, Y., Wang, L., & Zhang, W. (2020). Effects of conservation tillage on soil organic carbon and nitrogen: A global meta-analysis. Science of The Total Environment, *703*, 134959. https://doi.org/10.1016/j.scitotenv.2019.134959
- Liang, G., Cai, A., Wu, H., Houssou, A. A., & Ren, C. (2022). Soil microbial community structure and activity under different long-term fertilization managements in a cropland. Applied Soil Ecology, *169*, 104211. https://doi.org/10.1016/j.apsoil.2021.104211
- Liu, X., Chen, D., Yang, T., Huang, F., Zhang, S., & Li, L. (2024). Towards a process-based understanding of rhizosphere phosphorus chemistry: Coupling soil geochemistry with root exudation models. Rhizosphere, *29*, 100839. https://doi.org/10.1016/j.rhisph.2023.100839
- Muehe, E. M., & Kappler, A. (2023). Microbial iron cycling in paddy soils: From redox reactions to plant nutrition. Nature Reviews Microbiology, *21*(4), 236-250. https://doi.org/10.1038/s41579-022-00824-8
- Nobile, C., Denoroy, P., & Houben, D. (2020). The dynamics of soil phosphorus fractions and bioavailability under long-term organic and mineral fertilization. Agriculture, Ecosystems & Environment, *301*, 107050. https://doi.org/10.1016/j.agee.2020.107050
- O’Connell, D. W., Birkinshaw, C., & O’Dwyer, T. F. (2021). A review of the use of sequential extraction to assess the biogeochemistry of metals in soils and sediments. Environmental Reviews, *29*(3), 378-402. https://doi.org/10.1139/er-2020-0078
- Pan, W., Lu, Q., Chen, Z., & Li, Y. (2023). The role of dissolved organic matter in the bioavailability and toxicity of metals in soils: A meta-analysis. Environmental Chemistry Letters, *21*(1), 421-437. https://doi.org/10.1007/s10311-022-01515-w
- Raza, S., Miao, N., Wang, P., Ju, X., Chen, Z., Zhou, J., & Kuzyakov, Y. (2020). Dramatic loss of inorganic carbon from paddy soils following agricultural abandonment. Science of The Total Environment, *709*, 135579. https://doi.org/10.1016/j.scitotenv.2019.135579
- Rinklebe, J., Antoniadis, V., Shaheen, S. M., Rosche, O., & Altermann, M. (2021). Health risk assessment of potentially toxic elements in soils along the Elbe River, Germany. Environment International, *146*, 106245. https://doi.org/10.1016/j.envint.2020.106245
- Ros, G. H., Verweij, S. E., & van Rotterdam-Los, A. M. D. (2022). Predicting soil nutrient bioavailability using mid-infrared spectroscopy and chemometrics. Geoderma, *405*, 115399. https://doi.org/10.1016/j.geoderma.2021.115399
- Schulz, H., & Glaser, B. (2023). Biochar effects on the transformation and bioavailability of heavy metals in contaminated soils: A critical review. Journal of Soils and Sediments, *23*(1), 1-18. https://doi.org/10.1007/s11368-022-03342-5
- Shaaban, M., Wu, Y., Khalid, M. S., Peng, Q., Xu, X., Wu, L., … & Hu, R. (2021). Reduction in soil N2O emissions by pH manipulation and enhanced nosZ gene transcription under different amendments. Biology and Fertility of Soils, *57*(1), 31-45. https://doi.org/10.1007/s00374-020-01505-5
- Shi, R., Liu, Z., Li, Y., Jiang, T., Xu, M., Li, J., & Xu, J. (2020). Mechanisms for increasing soil resistance to acidification by long-term manure application. Soil and Tillage Research, *196*, 104433. https://doi.org/10.1016/j.still.2019.104433
- Soil Health Institute. (2023). The North American Project to Evaluate Soil Health Measurements. Soil Health Institute. https://soilhealthinstitute.org/north-american-project/
- Tian, D., & Niu, S. (2022). Soil pH and carbonates jointly shape the global biogeography of arbuscular mycorrhizal fungi. Global Ecology and Biogeography, *31*(6), 1186-1198. https://doi.org/10.1111/geb.13490
- Wang, F., Chen, S., Wang, Y., Zhang, Y., Hu, C., & Liu, B. (2024). Long-term organic fertilization increases the bioavailability of zinc in soil by altering its chemical speciation. Science of The Total Environment, *908*, 168285. https://doi.org/10.1016/j.scitotenv.2023.168285
- Zhao, F. J., Ma, Y., Zhu, Y. G., Tang, Z., & McGrath, S. P. (2021). Soil contamination in China: Current status and mitigation strategies. Environmental Science & Technology, *55*(13), 8455-8464. https://doi.org/10.1021/acs.est.1c02967
- Alloway, B. J. (2013). Heavy metals in soils: Trace metals and metalloids in soils and their bioavailability(3rd ed.). Springer. https://doi.org/10.1007/978-94-007-4470
- Bravin, M. N., Tentscher, P., Rose, J., & Hinsinger, P. (2009). Rhizosphere pH gradient controls copper availability in a strongly acidic soil. Environmental Science & Technology, 43(15), 5686–5691. https://doi.org/10.1021/es900055
- Brown, G. E., Jr., & Calas, G. (2012). Environmental mineralogy – Understanding element behavior in ecosystems. Comptes Rendus Geoscience, 344(11–12), 577–586. https://doi.org/10.1016/j.crte.2012.10.0
- Fageria, N. K., Baligar, V. C., & Jones, C. A. (2011). Growth and mineral nutrition of field crops (3rd ed.). CRC Press. https://doi.org/10.1201/b1016
- Gu, C., & Margenot, A. J. (2021). A soil science renaissance. Geoderma, 403, 115388. https://doi.org/10.1016/j.geoderma.2021.11538
- Hinsinger, P., Bengough, A. G., Vetterlein, D., & Young, I. M. (2009). Rhizosphere: Biophysics, biogeochemistry and ecological relevance. Plant and Soil, 321(1–2), 117–152. https://doi.org/10.1007/s11104-008-9885-
- Kallenbach, C. M., Frey, S. D., & Grandy, A. S. (2016). Direct evidence for microbial-derived soil organic matter formation and its ecophysiological controls. Nature Communications, 7, 13630. https://doi.org/10.1038/ncomms1363
- Kopittke, P. M., Menzies, N. W., Wang, P., McKenna, B. A., & Lombi, E. (2019). Soil and the intensification of agriculture for global food security. Environment International, 132, 105078. https://doi.org/10.1016/j.envint.2019.10507
- Kruse, J., Abraham, M., Amelung, W., Baum, C., Bol, R., Kühn, O., Lewandowski, H., Niederberger, J., Oelmann, Y., Rüger, C., Santner, J., Siebers, M., Siebers, N., Spohn, M., Vestergren, J., Vogts, A., & Leinweber, P. (2015). Innovative methods in soil phosphorus research: A review. Journal of Plant Nutrition and Soil Science, 178(1), 43–88. https://doi.org/10.1002/jpln.20140032
- Lal, R. (2015). Restoring soil quality to mitigate soil degradation. Sustainability, 7(5), 5875–5895. https://doi.org/10.3390/su705587
- Lindsay, W. L. (1979). Chemical equilibria in soils. John Wiley & Sons.
- Lorenz, S. E., Hamon, R. E., & McGrath, S. P. (1994). Differences between soil solutions obtained from rhizosphere and non-rhizosphere soils by water displacement and soil centrifugation. European Journal of Soil Science, 45(4), 431–438. https://doi.org/10.1111/j.1365-2389.1994.tb00528.
- Marschner, P. (Ed.). (2012). Marschner’s mineral nutrition of higher plants(3rd ed.). Academic Press. https://doi.org/10.1016/C2009-0-63043-
- Mendez, A., & Mihajlovic, M. (2021). Chemical speciation of iron, manganese, zinc, and copper in the rhizosphere and non-rhizosphere of Helianthus annuus in calcareous soil. Journal of Plant Nutrition and Soil Science, 184(2), 248–259. https://doi.org/10.1002/jpln.20200032
- Öborn, I., Andrist-Rangel, Y., Askekaard, M., Grant, C. A., Watson, C. A., & Edwards, A. C. (2005). Critical aspects of potassium management in agricultural systems. Soil Use and Management, 21(s1), 102–112. https://doi.org/10.1079/SUM200530
- Parkhurst, D. L., & Appelo, C. A. J. (2013). *Description of input and examples for PHREEQC version 3: A computer program for speciation, batch-reaction, one-dimensional transport, and inverse geochemical calculations* (U.S. Geological Survey Techniques and Methods, Book 6). U.S. Geological Survey.
- Schwertmann, U. (1991). Solubility and dissolution of iron oxides. Plant and Soil, 130(1–2), 1–25. https://doi.org/10.1007/BF0001185
- Sparks, D. L. (2003). Environmental soil chemistry(2nd ed.). Academic Press.
- Tessier, A., Campbell, P. G. C., & Bisson, M. (1979). Sequential extraction procedure for the speciation of particulate trace metals. Analytical Chemistry, 51(7), 844–851. https://doi.org/10.1021/ac50043a01
- S. Department of Agriculture, Natural Resources Conservation Service. (2021). Soil health. https://www.nrcs.usda.gov/wps/portal/nrcs/main/soils/health
Accepted on: 22 Dec 2025
Second Review by: Dr. Yudit Topo
Final Approval by: Dr. Tanay Pramanik

















