Chemical Composition and Antioxidant Activity of Fruit Extracts from Different Horticultural Crops
1Department of Fruit Science, SKS CARS, Rajnandgaon, IGKV, Chhattisgarh, India.
2Department of Mathematics and Statistics, JNKVV, Jabalpur (MP), India.
3Department of Plant Protection, Krishi Vigyan Kendra Mamnoor, Warangal, Telangana, India.
4Faculty of Agriculture Sciences, Shri Khushal Das University Hanumangarh, Rajasthan, India.
5Bihar Agricultural University, Sabour, Bhagalpur, India
6Department of Horticulture (Fruit and Fruit Technology), Bihar Agricultural University, Sabour, Bhagalpur, Bhagalpur, India.
7RRS, Agwanpur, Bihar Agricultural University, Sabour, Bhagalpur, India.
8College of Horticulture, Odisha University of Agriculture and Technology, Bhubaneswar, Odisha, India.
9Bhai Gurdas Degree College, BGGI Sangrur, Punjab, India.
10Department of Horticulture, Faculty of Agriculture, Udai Pratap College, Varanasi (U.P. ), India.
Corresponding Author E-mail: shivraj.vns@gmail.com
Download this article as:
ABSTRACT:This review summarizes this complex connection between the phytochemical diversity of horticultural crops and its antioxidant activity as being not a uniform property but the particular action of particular profiles of compounds, say anthocyanins in berries and flavanones in citrus and tannins in pomes. It emphasizes the roles played by genotype, environment, and postharvest processing of this potential. Although in vitro assays are crucial screening tools, to fill the bioactivity gap, more complex models need to be developed, taking into consideration bioavailability and synergistic matrix effects, with evidence of health benefits provided by epidemiological and intervention studies. The future is dependent on standardization of the protocols, the implementation of integrated -omics and biotechnological technology in crop improvement, and the entrenching of sustainability in the form of circular biorefinery. Finally, to achieve the full potential of fruit bioactives will require a multidisciplinary/ holistic approach, which exists between accurate phytochemistry to confirmed human health-effects and long-term market value.
KEYWORDS:ABTS; Ascorbic acid; Bioactive compounds; Carotenoids; Extraction methods; FRAP; Functional foods; Flavonoids; Genotype variation; ORAC; Phenolic acids; Postharvest;
Introduction
The insidious and widespread presence of oxidative stress in human pathophysiology forms a health burden to the rest of the world.1,2 Oxidative damage of lipids, proteins, and DNA is a critical etiological agent in the pathogenesis of non-communicable diseases (NCDs) such as cardiovascular diseases, neurodegenerative diseases, diabetes, and cancer since it is characterized by an imbalance between the formation of reactive oxygen and nitrogen species (ROS/RNS) and the endogenous antioxidant defenses of the body.3,4 Though the body has inbuilt mechanisms of defense against superoxide, such as superoxide dismutase and glutathione peroxidase, these mechanisms are often overwhelmed by15,6 the contemporary lifestyles, environmental contaminants and ageing. This has led to the development of the reinforcement of these defenses by exogenous antioxidants found in the diet as a central approach in both preventive and complementary medicine.17,8 The mechanism of action of dietary antioxidants is complex, which includes direct scavenging of free radicals, the binding of pro-oxidative metal ions, and the activation of endogenous cytoprotective pathways, the most critical being the Keap1-Nrf2-ARE one. This awareness has changed the paradigm of nutrition as a preventative concern towards deficiency diseases to a proactive maximization of health by bioactive food components to the diet becoming one of the primary interfaces between environmental challenge and genomic stability.9,10
In this nutritional environment, the horticultural crops, which comprise a huge phylogenetic pool of fruits, are recognized as unique11,12 sources of these bioactive phytochemicals. In contrast to staple crops that are valued mainly as a source of caloric value, fruits underwent complex secondary metabolomes as adaptive mechanisms to environmental stressors that have coincidentally given them powerful biological activities in humans. Such a chemical arsenal comprises but is not restricted to a gorgeous array of phenolic compounds (flavonoids such as anthocyanins and flavonols,13,14 and non-flavonoids such as phenolic acids and stilbenes), carotenoids, vitamin C derivatives, and vitamin E derivatives as well as alkaloids. These metabolites are not fixed, but they are subject to dynamic control by a three-factor triad genotype (species and cultivar), pre-harvest agronomic and environmental conditions, and post-harvest handling.15,16 This natural variance implies that a blueberry is not just a blueberry it has an antioxidant profile that is a fingerprint of its genetic composition, its terroir, and its path to the consumer. In this way, the horticultural kingdom provides a naturally combinatorial library of antioxidant compounds, both as a challenge to systematic study and an unprecedented source of bioactives of interest.17
It gives rise to the idea of fruits going beyond being mere nutritional commodities to become functional elements in the growing market of functional foods and nutraceuticals. The functional in this context means a provisional.18,19 positive impact on one or more target functions in the body over and above sufficient nutritional impacts, in a manner that is pertinent to enhanced health, health or diminished risk of disease. No longer is fruit extracts and its purified ingredients limited to the produce aisle; they are now20,21 being developed into a continuum of value added products. They are used as natural preservatives in meat and dairy products, as inhibitors of lipid peroxidation; as bioactive fortificants in beverages, baked products and snacks; as essential constituents of dietary supplements and cosmeceuticals. A further complication to this application-based enthusiasm is the necessity to transcend the generic assertion of high antioxidant activity and to have a more specific,22,23 more mechanistic knowledge. The effectiveness of a fruit extract in a food or a human body is not only determined by the overall level of phenols, but also by the compounds contained in it, their bioavailability, their interaction with the food or gut matrix and their metabolism. Thus, clarifying the complex relationship between special chemical structure of a fruit extract and its consequent biological activity is of supreme scientific and business value.24,25
Here, at the intersection between health demand, chemical complexity and application potential, lies the same space upon which the scope of this review cuts, and it is its claims to novelty. Although many of the current studies have enumerated the26,27 antioxidants in a single fruit species or family, or addressed methodological issues of antioxidant assays, there is a strong deficit in literature, namely, a systematic, cross-cropal study that deconstructs the interaction between phytochemical composition and antioxidant effectiveness across the entire horticultural taxa spectrum. This review will attempt to fill that gap. Our new method implies a critical synthesis of data on different categories of crops (berries, stone fruits, pomes, citrus, tropical fruits and vines) to extract patterns, oppositions, and characteristic signatures. We will question why, e.g.,28 the hydrophilic antioxidant activity of berries with anthocyanin as the prevailing protein is expressed differently in assays compared to the lipophilic activity of carotenoid-rich mango or persimmon.29-31 We shall also examine the effect of the stereochemistry of citrus flavanones on the radical-scavenging kinetics of flavanones in relation to the more common flavonols. Moreover, we shall critically assess the effects of using genotypic selection, agricultural practice and processing extraction method to lever the antioxidant yield and strength of these natural products.32,33 With this holistic, comparative approach, this review will not offer just an inventory but will develop a basis of knowledge to guide future research, inform breeding programs of improved nutritional quality, and allow the rational design of functional food ingredients.34,35 Our synthesis is, ultimately, supposed to act as a strategic roadmap to the scientists, horticulturalists, and even the food technologists to enable them to make the most from the multifaceted potential of the horticultural crops in the global battle against oxidative stress and the disease load it entails.
Methodological Framework for Review
A systematic literature search was completed according to the PRISMA in order to have transparency and reproducibility. They were searched in primary databases (Scopus, Web of Science, PubMed) with organized combinations of keywords: fruit names, extract, phytochemical, phenolic, antioxidant, and the most important acronym of major assays (e.g., DPPH, ORAC, FRAP). Manual screening of reference lists of the relevant reviews was carried out. The inclusion criteria required studies that reported the following: (1) specific phytochemical identification/quantification (through either HPLC, GC-MS, or NMR) and (2) standardized in vitro measurements of antioxidant activity (as percentile). The exclusion criteria were to prevent the studies which lacked adequate chemical characterization, utilized non-standardized assays, or studies which dealt with processed foods which contained added ingredients. Abstracts and articles that were not in English and did not have a complete article were excluded. Such strict approach guaranteed that the data that were examined were obtained on the basis of methodologically reliable, similar studies, creating a solid basis of comparative analysis of cross-crop.
 |
Chart 1: Flow Chart Methodological Framework for Review
|
The phytochemical spectrum: a crop-by-crop analysis
The berry family, both in their true sense (such as blueberries, currants, etc.) and their aggregate sense (raspberries, strawberries), are the canonical high-antioxidant cultivated crops with phytochemical levels of unparalleled density and diversity. Their typical compounds constitute a triadic cooling one: anthocyanin, ellagitannin, and proanthocyanidin (condensed tannins). The pigments that produce the bright red, purple, and blue colors are known as anthocyanins and are majorly expressed by the derivatives of cyanidin, delphinidin, pelargonidin, peonidin, and malvidin. These compounds are strong antioxidants through hydrogen donation and important regulating factors of cellular signaling pathways. The complex hydrolysable tannins present in raspberries, blackberries, and strawberries, which are known as ellagitannins, are hydrolyzed to produce ellagic acid, which has been reported to have the chemopreventive potential. Proanthocyanidins, especially cranberries and blueberries are oligomeric and polymeric flavonoid, which have a high radical-scavenging capacity and anti-adhesive properties, which are unique to them. Nevertheless, it is a gross simplification to say there is a unified pattern of the so-called profile of a berry. There is striking quantitative and qualitative difference between species and cultivars which is determined by the complex genetic programmes. An example is highbush blueberries ( Vaccinium corymbosum ) that has been hailed as having a diverse anthocyanin profile, and bilberries ( Vaccinium myrtillus ) are usually higher in total concentrations. In strawberries, day-neutral varieties might end up storing varying ratios of phenolics as the June-bearing varieties. Environmental factors also modulate this genotypic variation; higher exposure to sunlight generally increases anthocyanin and flavonol synthesis in response to increased environmental protection and nutrient conditions and water stress can play a major role in changing the partitioning of carbon into particular phenolic pathways. The antioxidant potential of a berry extract is therefore not a constant but a dynamic consequence of the interplay between genotype and environment, hence cultivar selection and agronomic management are important instruments in enhancing bioactive production.
Conversely, stone fruits (drupes) of the genus Prunus, cherries, plums, peaches, and apricots, feature a phytochemical pattern based on hydroxycinnamic acids, flavonols, and, in one case only, cyanogenic glycosides. These fruits have flesh and skin that contains abundant neochlorogenic and chlorogenic acids (esters of caffeic and quinic acid) that have played a major role in their non-flavonoid antioxidant capability. The concentration of Flavonols especially quercetin and kaempferol glycosides is found in the skin, which provides them a protective effect against UV radiation. Another characteristic feature of stone fruit chemistry is cyanogenic glycosides, including amygdalin in apricot kernels, which, although a protection mechanism of the plant, should be taken into careful consideration during processing because of the possibility of cyanide release. These classes of compounds are inconsistently balanced. Tart cherries (Prunus cerasus) contain anthocyanins (cyanidin-based) in high amounts and have a high anti-inflammatory activity, whereas sweet cherries (Prunus avium) can have high amounts of hydroxycinnamates. Plums, particularly dark-skinned ones are rich in anthocyanins and have a strong antioxidant effect which is usually attributed to their laxative dihydroxyphenylisatin compound. Peach/Apricots, which are less pigmented, obtain their main antioxidant hydrolysis through chlorogenic acid as well as carotenoids, such as b-carotene and b-cryptoxanthin, and procyanidins. Qualitative difference between cultivars is once again dominant; as such, blood-fleshed peaches do maintain anthocyanins throughout the mesocarp, which white-fleshed cultivars do not, resulting in a difference in the total amount of phenolics and other antioxidant indicators by an order of magnitude.
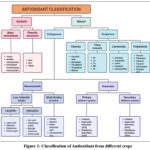 |
Figure 1: Classification of Antioxidant from different crops Click here to View Figure |
This again highlights the fact that even in a single fruit type, the choice of cultivar determines the chemical story and resultant bioactivity. Pome fruits (apples, pears, and quinces) present a phytochemical profile that is unique in terms of focusing on dihydrochalcones, flavan-3-ols and a major hydroxycinnamic acid. The dihydrochalcone phloridzin is nearly an apple fruit (Malus domestica) specific compound, used as a taxonomic marker, and has been reported to have bioactive effects on glucose metabolism. The significant contribution to the antioxidant properties of flesh and, more specifically, peel are flavan-3-ols; especially, monomeric (+)-catechin and (-)-epicatechin and their polymers (procyanidins). Chlorogenic acid is the phenolic acid that dominates among the pomes. The chemical structure of pomes is very compartmentalized: the peel may contain several times the antioxidant compounds in the flesh, so taking the whole fruit or using peel extract becomes important. It is apples that have become legendary in terms of cultivar variation, with old cultivars such as the old cultivar known as Anna or the old cultivar known as Granny Smith often exhibiting other, even more phenolic profiles than some of the newer commercial cultivars, although strong breeding has also yielded new, high-polyphenol cultivars. Pear (Pyrus) chemistry is similar, but often contains a lower total phenolic concentration, and has a different flavor profile including arbutin (a glucoside of hydroquinone) in certain types. Quince ( Cydonia oblonga), commonly used because of astringency, is extraordinarily high in phenolic acids, and has an extremely high antioxidant potential, often higher than those of apple and pear. The direct association of phytochemistry with the sense experience also emphasizes the structural role of procyanidins in pome fruit astringency as it may be necessary to balance breeding and processing to attain palatability. Citrus fruits (oranges, lemons, grapefruits, mandarins) have a distinctive phytochemical niche characterized by a specific group of flavonoids and a combination of limonoids. The flavanones are the most unique ones and they occur in large concentration as glycosides. Sweet oranges and lemons contain hesperidin (hesperetin-7-rutinoside), grapefruit its typical bitter-tasting component naringin (naringenin-7-neohesperidoside), and lemons its neoeriocitrin. These flavanone glycosides have high antioxidant and anti-inflammatory effect, although the bioavailability of these compounds depends on the presence of sugar moiety. Vitamin C (ascorbic acid) can also be found in citrus fruits, and polymethoxylated flavones (e.g., tangeretin, nobiletin) with strong bioactivity, carotenoids, which give color (lycopene in red grapefruit, b-cryptoxanthin in mandarins). Antioxidant and chemopreventive effects are also exhibited by the bitter limonoids (limonin and nomilin) that are present in seeds and tissues. The variation of the profile is highly species- and variety-specific. Anthocyanins which are not common in citrus are found in blood oranges making the fruits more antioxidant. The flavanone profile of Meyer’ lemons differs with that of Eureka. Moreover, tissue specialization is severe: the flavedo (colored peel) and albedo (white peel) do feature reservoirs of polymethoxylated flavones and phenolic acids at levels much higher than those in the juice vesicles, which makes citrus processing waste an excellent source of antioxidant extract. The chemical heterogeneity of the category of tropical and exotic fruits is astonishing, presenting a spectacular range of exceptional bioactive compounds and traditional antioxidants. Examples of this diversity are the xanthones in mangosteen (Garcinia mangostana), especially a- and g-mangostin that are good antioxidants and anti-inflammatories that are not available in temperate fruits. Hydrolysable tannins, known as punica granatum punica granatum is a good source of hydrolysable tannins (punica granatum) known as ellagic acid, which are formed by hydrolyzing punica granatum punic a granatum the unusual content of punica granatum punica granatum is an exceptional scorer on the antioxidant capacity checklist across several assay systems.
Kiwifruits (Actinidia spp.) contain a lot of vitamin C and carotenoids such as lutein. Papaya contains papain, vitamin C and carotenoids. Acai ( Euterpe oleracea ) berries are palm fruits also known as acai berries, which are very high in anthocyanin (cyanidin-3-glucoside) and proanthocyanidin and fatty acids. Mango (Mangifera indica) is a source of gallotannins and a distinct xanthanoid, mangiferin, with strong antioxidant effects. One of the most significant common antioxidant elements in numerous tropical fruits such as guava and camu camu, which is among the richest natural sources as well, is vitamin C content. This change is affected by not only genetics but also deeply by the stage of ripening; the process of ripening causes a dramatic change in the pigment composition (chlorophyll, carotenoids, anthocyanins) and phenolic breakdown of many tropical fruits, which directly and dynamically alter the antioxidant profile of cane fruits as they shift to a fully ripe (more carotenoids and sugars) state. Last, but not the least, vine fruits, mainly grapes (Vitis vinifera), and other significant fruits such as figs and persimmons, fill the palette. Grapes are an interest of two sides, to the fresh fruit, and the product obtained, wine. Their seeds and skin contain gold mines of antioxidants. The skins include stilbenes (the best-known of which is resveratrol and its derivatives, produced in response to fungal stress), anthocyanins (in red/purple varieties) and flavonols (quercetin, myricetin). The grape seeds are extremely enriched with the proanthocyanidins, especially, procyanidin B1 and B2 dimers and complex polymers. The breeding difference is viticulture based on the cultivar; the Cabernet Sauvignon with thick-skinned and small berry is bred towards an abundance of phenolic compounds that is not typical of table grape varieties with thin-skinned. Figs ( Ficus carica) contain phenolic acids, flavonols, and anthocyanins, with much inter- and intra-varietal differences in peel versus pulp and among varieties of different colour. Persimmon (Diospyros kaki), particularly the astringent types prior to ripeness, are also an extraordinarily high source of soluble tannins (proanthocyanidins), which become coagulated in the ripening process, lowering astringency and altering the antioxidant antiretroviral availability. Its composition is further contributed by the presence of carotenoids such as the b-cryptoxanthin and the triterpenoid betulinic acid, which are unique to it. Tannin profiles of the grape and persimmon are very important since the level of polymerization has a significant impact on the antioxidant activity of the fruit as well as its relationship with proteins, which will impact bioavailability and sensory.
This comparative cross-taxa study shows that although the universal language of antioxidant activity is spoken by different mechanisms such as radical scavenging and metal chelation, the dialect is specific to each fruit family, and this is characterized by a characteristic phytochemical lexicon. The horticultural groups, one berry after another, one citrus after another, one pomes and persimmons after another, brings a different chemical solution to the problem of oxidation. The radical qualitative and quantitative diversity within each cohort, which is determined by a complex interaction of genetic origin, environmental conditions and postharvest destiny, render that the antioxidant ability of a fruit is not an abstract term. This knowledge is critical in guiding breeding systems, improved agriculture, and processing, and, finally, making effective decisions in the creation of functional foods and nutraceuticals to utilize the specific phytochemical potential of the varieties of fruit abundant in nature.
Table 1: Specific chemical compounds mentioned, categorized by their common compound class and associated fruit group
| Chemical name (specific compound) | Compound class/category | Primary fruit group (example) |
| Cyanidin (derivative) | Anthocyanin | Berries |
| Delphinidin (derivative) | Anthocyanin | Berries |
| Pelargonidin (derivative) | Anthocyanin | Berries |
| Peonidin (derivative) | Anthocyanin | Berries |
| Malvidin (derivative) | Anthocyanin | Berries |
| Ellagic acid | Hydrolysable Tannin (Ellagitannin metabolite) | Berries (e.g., Raspberry, Strawberry) |
| Proanthocyanidins | Condensed Tannins (Flavonoid polymers) | Berries (e.g., Cranberry, Blueberry) |
| Neochlorogenic acid | Hydroxycinnamic Acid | Stone Fruits |
| Chlorogenic acid | Hydroxycinnamic Acid | Stone Fruits, Pome Fruits |
| Quercetin (glycosides) | Flavonol | Stone Fruits, Vine Fruits |
| Kaempferol (glycosides) | Flavonol | Stone Fruits |
| Amygdalin | Cyanogenic Glycoside | Stone Fruits (Apricot kernel) |
| Dihydroxyphenylisatin | Laxative Compound | Stone Fruits (Plum) |
| β-carotene | Carotenoid | Stone Fruits (Peach, Apricot), Tropical Fruits |
| β-cryptoxanthin | Carotenoid | Stone Fruits (Apricot), Citrus, Vine Fruits |
| Procyanidins | Proanthocyanidin subtype | Stone Fruits, Pome Fruits, Vine Fruits |
| Phloridzin | Dihydrochalcone | Pome Fruits (Apple) |
| (+)-Catechin | Flavan-3-ol | Pome Fruits |
| (-)-Epicatechin | Flavan-3-ol | Pome Fruits |
| Arbutin | Hydroquinone Glucoside | Pome Fruits (Pear) |
| Hesperidin (Hesperetin-7-rutinoside) | Flavanone Glycoside | Citrus Fruits |
| Naringin (Naringenin-7-neohesperidoside) | Flavanone Glycoside | Citrus Fruits (Grapefruit) |
| Neoeriocitrin | Flavanone Glycoside | Citrus Fruits (Lemon) |
| Ascorbic Acid (Vitamin C) | Vitamin | Citrus Fruits, Tropical Fruits |
| Tangeretin | Polymethoxylated Flavone | Citrus Fruits |
| Nobiletin | Polymethoxylated Flavone | Citrus Fruits |
| Lycopene | Carotenoid | Citrus Fruits (Red Grapefruit) |
| Limonin | Limonoid | Citrus Fruits |
| Nomilin | Limonoid | Citrus Fruits |
| α-Mangostin | Xanthone | Tropical Fruits (Mangosteen) |
| γ-Mangostin | Xanthone | Tropical Fruits (Mangosteen) |
| Punicalagins | Hydrolysable Tannin (Ellagitannin) | Tropical Fruits (Pomegranate) |
| Lutein | Carotenoid | Tropical Fruits (Kiwifruit) |
| Papain | Proteolytic Enzyme | Tropical Fruits (Papaya) |
| Cyanidin-3-glucoside | Anthocyanin | Tropical Fruits (Açaí) |
| Gallotannins | Hydrolysable Tannins | Tropical Fruits (Mango) |
| Mangiferin | Xanthonoid | Tropical Fruits (Mango) |
| Resveratrol | Stilbene | Vine Fruits (Grape skin) |
| Myricetin | Flavonol | Vine Fruits (Grape skin) |
| Procyanidin B1 | Proanthocyanidin dimer | Vine Fruits (Grape seed) |
| Procyanidin B2 | Proanthocyanidin dimer | Vine Fruits (Grape seed) |
| Betulinic acid | Triterpenoid | Vine Fruits (Persimmon) |
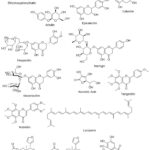 |
Figure 2: Structures of chemical compounds categorized by their common compound class and associated fruit group Click here to View Figure |
The measurement of the antioxidant activity of fruit extracts is an essential but complicated task that is needed to convert the phytochemical composition to elucidable biological action. This analysis is done on a continuum of assays, with different principles, advantages and limitations, and the ultimate antioxidant effectiveness is dictated by a collection of pre- and post-harvest determinants. Strengths and Limitations in In vitro Chemical Assays. First line screening mostly depends on in vitro chemical assays that can be categorized in terms of a reaction mechanism upon which they are based. Single Electron Transfer (SET) Single electron transfer assays are used to test the reducing capacity of an antioxidant. The DPPH (2,2-diphenyl-1-picrylhydrazyl) and ABTS+ (2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) assays are based on decolorization of a stable radical chromogen and the FRAP (Ferric Reducing Antioxidant Power) assay is the decolorization of ferric ions to the coloured ferrous complex. The simplicity, speed, and reproducibility of these assays are the hallmark of these techniques, and they are ideal when it comes to comparative ranking. Yet, they are performed in aqueous or alcoholic solutions, may not be indicative of activity in lipid systems and their pH dependence can bias certain compounds (FRAP is best at low pH and only identifies antioxidants that reduce ferric ions). Hydrogen Atom Transfer (HAT) assays including ORAC (Oxygen Radicals Absorbance Capacity) and TRAP (Total Radical-Trapping Antioxidant Parameter) are in contrast to a peroxyl radical-producing probe to which the assay competes to measure the inhibition of oxidation kinetics. HAT-based techniques are said to be more biologically relevant because they simulate the radical chain-breaking mechanism that is essential in lipid peroxidation. They are however, more technical, time consuming and prone to interference. Importantly, SET and HAT assay results often do not correlate well, because they test different chemical properties; an extract of fruits with lots of fast-reducing species such as vitamin C may score highly in FRAP but poorly in ORAC, whereas a fruit extract with a lot of chain-breaking phenolics may do the reverse. Thus, the use of one assay is not sufficient to give a complete picture and a battery of tests is suggested to reflect the multifaceted nature of antioxidant activity.
In Vivo and Cellular Assays: Significance and Problems. Cellular (e.g. CAA – Cellular Antioxidant Activity) and in vivo assays are used to bridge the gap between the chemical potential and the biological reality. These models take into consideration important factors that have been overlooked by the simple chemical assays: cellular uptake, metabolism, sub-cellular location, and interactions with endogenous antioxidant systems. As an example, the CAA assay measures compound anti-oxidative capacity of compounds to prevent oxidation of a fluorescent probe in living cells, including elements of bioavailability and intracellular activity. Measurement of reduction in oxidative stress biomarkers (e.g. malondialdehyde, 8-hydroxy-2′-deoxyguanosine) in tissues or plasma caused by diet intervention can be measured in animal models. Although these methods have better physiological relevance, they are costly, low-throughput, ethically limited and the outcomes may be confounded by the homeostatic control of the animal and complex interactions involving diet and metabolites. There is a challenge of extrapolating rodent models to the health outcomes of human beings.
The Bioactivity Gap: Concentration and Bioavailability. Such gives rise to the main paradox of the so-called bioactivity gap: a high concentration of an antioxidant in vitro does not necessarily have the same effect in vivo. The critical filter is bioavailability which includes liberation of the food matrix, absorption, distribution, metabolism, and excretion. A large number of powerful polyphenols e.g. ellagitannins and the high-molecular-weight proanthocyanidins are not readily absorbed in the small intestine. They can have local antioxidative effects in the gut, or can be metabolized by colonic microbiota into smaller, absorbable metabolites (e.g. urolithins of ellagitannins), and which have a systemic effect. Moreover, there are compounds, including anthocyanins that cannot be absorbed in the small intestine but can be stabilized by other food substances or contain active metabolites. This highlights the fact that the antioxidant activity reported in a test tube is an opportunity that is regulated by the complicated physiology of the consumer.
The effectors of the antioxidant potential of fruits. This potential, in itself, is neither a predetermined quality nor an unalterable feature but a chain reaction of factors between the table and the field. (1) Genotypic and Cultivar Differences Genetics are mainly responsible because it is the genetic code that determines the biosynthetic pathways to follow. Depending on the cultivar (e.g., blood orange vs. navel orange, highbush vs. lowbush blueberry) a tremendous variation in phytochemical profiles and consequent antioxidant capacity may occur, as explained in Section 3. (2) Agronomic Practices have a great influence to regulate this genetic potential. The production (synthesis) of phenolic compounds in response to controlled water stress (deficit irrigation) is common. Nitrogen fertilization may help in dilution of phenolic concentration through vegetative growth and organic farming method may tend to increase concentration of some antioxidants in some cases than the conventional systems. (3) Effects of Climatic and Environmental Stress are strong inducers. Flavonols and anthocyanins are natural sunscreens that are produced by high levels of solar radiation especially UV-B. There are also moderate thermal stress and reduced growing temperatures which are often observed to be associated with increased phenolic accumulation. (4) Maturity at Harvest and Postharvest Handling are very important time factors. As a rule, the peaks of phenolic antioxidants occur prior to complete physiological ripening, whereas carotenoids and vitamin C tend to increase throughout ripening. Postharvest handling conditions (temperature, atmosphere) may result in degradation of antioxidant activity or in some instances (e.g., chilling stress in sweet potatoes) induction of antioxidant activity. (5) Processing Techniques are the conclusive manipulation. The mechanical operations such as juicing or milling enhance oxidation unless regulated. Heat can inactivate heat-sensitive compounds, such as anthocyanins and vitamin C, but can also enhance extractability of bound phenolics or produce new antioxidants through the Maillard reaction products. Drying will concentrate the compounds but may subject the compound to oxidative losses, whereas fermentation will radically alter the antioxidant profile, such as in wine.
Synergism, Interactions, and Health Implications
The extrapolation of chemical profile of a fruit extract into a biologically relevant phenomenon goes beyond whatever the total of the individual antioxidant activities would entail. This is dictated by complicated interactions that are demonstrated by population research and are explained by molecular biology, which simply make up the basis of the health claims of fruit consumption. The Matrix Effect The Synergetic Interactions of Fruit Phytochemicals. Combined action of a fruit extract can be more powerful than the sum of the effect of its individual constituents, a phenomenon known as synergy or the matrix effect. Such synergy works in a number of ways. First, there are regenerative interactions, i.e. one antioxidant repairs and regenerates another. The recycling of the oxidized vitamin E (tocopheroxyl radical) by vitamin C (ascorbate) is an example of a canonical reaction, which, in turn, can be restored by flavonoids such as quercetin or glutathione. This forms a cascade of antioxidants, and this greatly increases the life and the performance of the system. Second, the pro-oxidant metal chelation caused by substances such as citric acid or some polyphenols inhibits Fenton reactions that get the hydroxyl radicals and hence saving other antioxidants. Thirdly, stability and bioavailability can be enhanced by structural and spatial interactions of the food matrix. An example to illustrate this is that anthocyanins in blueberries are likely to be stabilized by co-existing organic acids, and carotenoids present in mango are more easily absorbed in association with the natural lipids found in the mango fruit. This combination makes a strong case on why whole fruits or full-spectrum extracts are better than single-compound supplements, since the intrinsic matrix is more capable of providing an efficient and resilient biological defense.
Related to Health Outcomes: Epidemiological and Intervention Study Evidence. The inverse relationship between excessive intake of fruits and vegetables and the risk of getting chronic disease associated with oxidative stress such as cardiovascular disease (CVD), some forms of cancer and cognitive impairment has always been shown to be observed in observational epidemiology. Meta-analyses in particular demonstrate the cardiometabolic health benefits of berries demonstrating positive blood pressure, endothelial performance and LDL cholesterol oxidation. Randomized controlled trials (RCTs) are causal: pomegranate juice supplementation has been shown to decrease carotid intima-media thickness, whereas blueberry intervention enhances vascular performance and cognitive ability in vulnerable groups. Nevertheless, the evidence is not unanimous. A number of intervention trials using high-dose, single antioxidant vitamins (such as b-carotene or vitamin E) at such large doses have produced null (and even adverse) effects, thus highlighting the importance of the difference between the intake of antioxidants in natural food conditions and the intake of single, pharmacologic doses. This paradox supports the paradigm of synergy and implies that the synergies are obtained not by a radical scavenging but through the subtle regulation of the redox balance and the cellular signaling by a complex phytochemical blend.
Theoretical Mechanisms: Nrf2 Activation and Enzyme Modulation, and Gene Expression. Health benefits of fruit antioxidants are now seen to be mediated by less direct and stoichiometric radical neutralization in the bloodstream and by the regulation of endogenous defense and signaling pathways. One of the key pathways is the Nrf2 (Nuclear factor erythroid 2-related factor 2) activation pathway. Phytochemicals such as sulfuraphane (crucifers) or phenolic acid may alter Keap1 under oxidative stress to release Nrf2 to translocate to the nucleus. It is bound there on the Antioxidant Response Element (ARE) to stimulate the expression of a set of cytoprotective genes. These are phase II detoxifying enzymes (e.g., glutathione S-transferase, NAD(P)H:quinone oxidoreductase), endogenous antioxidant enzymes (e.g., superoxide dismutase, catalase, heme oxygenase-1), and glutathione synthesis enzyme. Therefore, fruit compounds will be indirect antioxidants, enhancing the bodily defenses system over an extended duration. They also regulate the performances of major enzymes, including blocking pro-inflammatory cyclooxygenase (COX) and lipoxygenase (LOX) or suppressing NADPH oxidase, the major source of cellular superoxide. Moreover, they affect gene expression involving inflammatory (e.g. damping of NF-kB signaling), apoptotic, and metabolic pathways. Simply put, the dietary antioxidants of fruits are not just sacrificial scavengers, that is, they are sophisticated redox-signaling molecules, which cause an adaptive, hormetic response, making the cell more resistant to future oxidative stress.
From Lab to Market: Applications and Innovations
The story of bioactive compounds extracted in fruit by-products under laboratory discovery to commercial use can be seen as an interesting example of how modern science can be used to create sustainability and health solutions. This extraction, application and market integration pipeline is converting the waste streams into highly productive resources in various industries. The extraction technology is the first phase in this value chain, and the change towards new technologies is transforming efficiency and environmental effects. Traditional methods like Soxhlet in organic solvents are usually time-consuming, energy-consuming and produce toxic wastes. Contrastingly, new green technologies are yielding more and purer compounds. Ultrasound-assisted extraction, in which the extraction takes place through the use of disruption of plant cell walls by means of the generation of cavitation bubbles, microwave extraction in which extract is made through the use of dielectric heating to achieve rapid internal warming and supercritical fluid extraction, especially in the use of CO2, in which extract is made through the use of a solvent-free method in which the sample is tuned to achieve the desired results. These high-tech methods are not just lab novelties; they are going to an industrial scale, to offer the constant, high quality extracts required in commercial uses, thus closing the critical divide between scholarly study and industry practicability.
Upon extraction, these strong phytochemicals, particularly phenolics, flavonoids and carotenoids are immediately used as natural antioxidants in food preservation in response to the increasing consumer preferences towards clean-label foods. The artificial preservatives such as BHT and BHA are not getting the market reception, and this has provided a profitable niche to the fruit-based antioxidants. Grape seed extracts, pomegranate peel extracts, or citrus by-products are being added to edible coatings on meats and fruits, impregnated into packaging films or added directly to oils and baked goods to prevent lipid peroxidation and microorganism growth. This also has a dual-functional advantage since this technology does not only increase the shelf life, but it also improves the nutritional composition of the foodstuff preserved, which is itself a highly marketable benefit. It is an example of a direct route to lab-scale testing of antioxidant capacity (e.g., through ORAC assays) to viable, preservative solutions in food production.
In addition to preservation, the essence of the ambition is coming up with value added products in the human health and wellness sector mainly through the nutraceuticals, functional foods and cosmeceuticals. Inflammation, cardiovascular and oxidative stress- point nutraceutical market is an enthusiastic user of fruit bioactives in dietary supplements. In the meantime, the functional food sector is evolving by enriching ordinary products yogurts with berry seed flour, beverages with encapsulated citrus flavonoids, snack bars with mango peel powder, with health-related benefits that provide more than just nutrition. The cosmeceutical industry, perhaps most dynamically, uses these compounds due to their anti-aging, brightening, and protective properties. Citrus vitamin C, pomegranate ellagic acid and grape resveratrol are compounded in serums, creams and sunscreens and sold on the effective platform of natural potency. All of these product types would demand a lot of R&D, whether it be stability testing or bioavailability improvement (through nanoemulsions or liposomes) or the more demanding process of clinical evaluation to claim efficacy, negotiating the challenging regulatory environments of food, supplement, and cosmetic regulatory agencies.
The basic principle of this whole ecosystem is the concept of valorization, which makes the use of by-products as complete and sustainable as possible. The latest concept of biorefinery resembles the petroleum refining, in an effort to achieve zero waste. Following first removal of high-value phenolics, the remaining matrix, the one containing fibers, pectins and proteins is further processed. The peels and Pomace can be used as bakery production to provide dietary fiber or pectin to be used in gelling agents; seeds can be used in the production of oils or gluten-free flours; and even used biomass can be used in compost or bio-production. This cascading model of use gives the highest returns in economy and lowest environmental footprint, transforming a cost-center (waste disposal) into a revenue stream. The cyclical bioeconomy is a market that prevails at the end, as each element has its use.
Current Challenges, Research Gaps, and Future Perspectives
To be able to harness the vast potential of fruit-derived bioactive compounds and move past promising research findings to solid, market-ready application, the discipline needs to be able to address several of its critical and inter-related frontiers. The future of this industry does not lie in small steps of making advances in the field of isolation, but in a holistic approach that integrates strict science with sustainable design. The major difficulty is Standardization of Extraction and Analytical Protocols. The existing non-uniformity is that various laboratories adopt different solvents, temperatures, times, and analysis procedures, making the reproducibility of results and comparison of studies quite impossible. Its quality and potency should be uniform to be adopted in the industry. It is necessary to come up with standard reference methods of important classes of compounds to make sure that the term antioxidant-rich pomegranate extract implies the same thing across all the laboratories and products, and achieves credibility both with the regulators and consumers.
The Requirement of Integrated -Omics Approaches is essential in order to comprehend and ultimately optimize the biosynthetic pathways of these useful compounds. Going beyond listing final phytochemicals (metabolomics), scientists need to incorporate the information about the gene expression (transcriptomics) and protein activities (proteomics) under various stresses. This systems biology approach has the potential to disclose the environmental factors that cause bioactive production, the major genetic regulators, and the most appropriate harvesting time resulting in maximum compound production. It is this profound mechanistic knowledge that is the plan of precise improvement.
The information is the motivating force of Breeding and Biotechnology of Enhanced Bioactive Content. There are always ways to breed naturally to end up with a higher concentration of antioxidants, though biotechnology today provides precision instruments. Genetic markers can speed up natural breeding, and gene editing methods such as CRISPR-Cas9 can be applied to activate the expression of a pathway that synthesizes a particular phenolics or vitamin without a foreign gene. This is aimed at designing functional fruits and their by-products as optimized raw materials streams at their inception.
At the same time, the discipline should invest in Moving Beyond In Vitro Assays. Although chemical tests (such as DPPH) can be helpful as a preliminary screen, they do not have a good correlation with real human health benefits. The future is in proven advanced models: complex 3D cell cultures, gut microbiome simulators, and organ-on-a-chip systems. These give important information on bioavailability, absorption, metabolism and tissue-specific effects. This is essential to prove health claims of nutraceuticals and cosmeceuticals, beyond correlation, to causation in biological relevant systems.
All these scientific activities should be supported by the firm spirit of Sustainability and Circular Economy in Bioactive Compound Production. The whole procedure, including cultivation and harvest, extraction to the residual waste, should be modeled in terms of environmental and economic circularity. This includes utilization of solvents that are green and extraction that is energy saving (such as microwave or ultrasound) and which are also fed by renewable energy. It requires the cascading valorization strategy where the extraction product biomass is further transformed to biofertilizers, biochar or bioenergy, and ultimately leads to almost zero waste. The sustainability story is not merely an ethical requirement, but a fundamental market benefit and a force behind sustainability.
Conclusion
The presence of antioxidant compounds in the diverse horticultural crops immediately points to a given fact: the intense bioactivity of fruits is based on their complex and diverse phytochemical structures. This review has established the various horticultural categories, including berries, stone fruits, pomes, citrus, tropical fruits, and vines, exhibit different signatures of bioactive compounds, such as anthocyanins and ellagitannins, flavanones, and xanthones. The compounds do not perform independently and their final antioxidant activity is a dynamic phenomenon carefully influenced by a triad of modifying factors: genetic blueprint, environmental and agronomic factors and postharvest processing. This diversity is not an obstacle, but a possibility, and varieties with the best nutrition can be selected and developed. More importantly, the process of chemical composition to proven health benefit is complicated. Although standardized in vitro assays (DPPH, FRAP, ORAC) offer the necessary comparative frames, it is the initial step. The importance of bioavailability, metabolic transformation, and, most importantly, synergistic interactions in the natural food matrix cannot be underestimated due to the so-called bioactivity gap between laboratory potential and in vivo effect. The trend is clearly to demonstrate that the entire extract, its complex interplay of compounds, frequently has more biological significance than an individual constituent and there is a growing need to consume whole fruits and minimally processed extracts. To ensure this potential is completely realized in the future, a convergent and multidisciplinary approach is the way to go. The future of research should focus on standardization of the methods of analysis, application of integrated -omics technologies to elucidate the biosynthetic pathways and the application of more sophisticated in vitro and clinical models to authenticate health claims. At the same time, breeding and biotechnological innovation have the potential of providing so-called designer crops with a high bioactive value. Finally, all these efforts should be properly incorporated into a circle of bioeconomy, in which green-extraction technologies and full exploitation of by-products will make the health gain consistent with the sustainability of the planet. Through converging horticultural science, food technology, nutrition and systems biology we can turn the natural variety of fruits into an effective, evidence-based preventive health and sustainable innovation powerhouse.
Acknowledgement
None
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Halliwell, B., & Gutteridge, J. M. C. (2015). Free radicals in biology and medicine(5th ed.). Oxford University Press.
CrossRef - Lobo, V., Patil, A., Phatak, A., & Chandra, N. (2010). Free radicals, antioxidants and functional foods: Impact on human health. Pharmacognosy Reviews, 4(8), 118–126. https://doi.org/10.4103/0973-7847.70902
CrossRef - Liu, R. H. (2013). Health-promoting components of fruits and vegetables in the diet. Advances in Nutrition, 4(3), 384S–392S. https://doi.org/10.3945/an.112.003517
CrossRef - Shahidi, F., & Ambigaipalan, P. (2015). Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects – A review. Journal of Functional Foods, 18, 820–897. https://doi.org/10.1016/j.jff.2015.06.018
CrossRef - Skrovankova, S., Sumczynski, D., Mlcek, J., Jurikova, T., & Sochor, J. (2015). Bioactive compounds and antioxidant activity in different types of berries. International Journal of Molecular Sciences, 16(10), 24673–24706. https://doi.org/10.3390/ijms161024673
CrossRef - Cásedas, G., Les, F., Gómez-Serranillos, M. P., Smith, C., & López, V. (2017). Anthocyanin profile, antioxidant activity and enzyme inhibiting properties of blueberry and cranberry juices: A comparative study. Food & Function, 8(11), 4187–4193. https://doi.org/10.1039/c7fo01205e
CrossRef - Usenik, V., Fabčič, J., & Štampar, F. (2008). Sugars, organic acids, phenolic composition and antioxidant activity of sweet cherry (Prunus avium). Food Chemistry, 107(1), 185–192. https://doi.org/10.1016/j.foodchem.2007.08.004
CrossRef - Wojdyło, A., Oszmiański, J., & Laskowski, P. (2008). Polyphenolic compounds and antioxidant activity of new and old apple varieties. Journal of Agricultural and Food Chemistry, 56(15), 6520–6530. https://doi.org/10.1021/jf800510j
CrossRef - Goulas, V., & Manganaris, G. A. (2012). Exploring the phytochemical content and the antioxidant potential of Citrus fruits grown in Cyprus. Food Chemistry, 131(1), 39–47. https://doi.org/10.1016/j.foodchem.2011.08.007
CrossRef - Viuda-Martos, M., Fernández-López, J., & Pérez-Álvarez, J. A. (2010). Pomegranate and its many functional components as related to human health: A review. Comprehensive Reviews in Food Science and Food Safety, 9(6), 635–654. https://doi.org/10.1111/j.1541-4337.2010.00131.x
CrossRef - Zaffino, C., Bruni, R., & Buzzi, R. (2015). HPLC-DAD-ESI-MS/MS characterization of unusual highly glycosylated acylated flavonoids from cauliflower (Brassica oleraceabotrytis) agro-industrial byproducts. Food Chemistry, 179, 259–267. https://doi.org/10.1016/j.foodchem.2015.01.136
CrossRef - Nile, S. H., & Park, S. W. (2014). Edible berries: Bioactive components and their effect on human health. Nutrition, 30(2), 134–144. https://doi.org/10.1016/j.nut.2013.04.007
CrossRef - Apak, R., Özyürek, M., Güçlü, K., & Çapanoğlu, E. (2016). Antioxidant activity/capacity measurement. 1. Classification, physicochemical principles, mechanisms, and electron transfer (ET)-based assays. Journal of Agricultural and Food Chemistry, 64(5), 997–1027. https://doi.org/10.1021/acs.jafc.5b04739
CrossRef - Prior, R. L., Wu, X., & Schaich, K. (2005). Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. Journal of Agricultural and Food Chemistry, 53(10), 4290–4302. https://doi.org/10.1021/jf0502698
CrossRef - Wolfe, K. L., & Liu, R. H. (2007). Cellular antioxidant activity (CAA) assay for assessing antioxidants, foods, and dietary supplements. Journal of Agricultural and Food Chemistry, 55(22), 8896–8907. https://doi.org/10.1021/jf0715166
CrossRef - Tomás-Barberán, F. A., & Espín, J. C. (2001). Phenolic compounds and related enzymes as determinants of quality in fruits and vegetables. Journal of the Science of Food and Agriculture, 81(9), 853–876. https://doi.org/10.1002/jsfa.885
CrossRef - Borges, G., Degeneve, A., Mullen, W., & Crozier, A. (2010). Identification of flavonoid and phenolic antioxidants in black currants, blueberries, raspberries, red currants, and cranberries. Journal of Agricultural and Food Chemistry, 58(7), 3901–3909. https://doi.org/10.1021/jf902263n
CrossRef - Kevers, C., Falkowski, M., Tabart, J., Defraigne, J. O., Dommes, J., & Pincemail, J. (2007). Evolution of antioxidant capacity during storage of selected fruits and vegetables. Journal of Agricultural and Food Chemistry, 55(21), 8596–8603. https://doi.org/10.1021/jf071736j
CrossRef - Aaby, K., Mazur, S., Nes, A., & Skrede, G. (2012). Phenolic compounds in strawberry (Fragaria x ananassa) fruits: Composition in 27 cultivars and changes during ripening. Food Chemistry, 132(1), 86–97. https://doi.org/10.1016/j.foodchem.2011.10.037
CrossRef - Rodríguez-Carpena, J. G., Morcuende, D., Andrade, M. J., Kylli, P., & Estévez, M. (2011). Avocado (Persea americana) phenolics, in vitro antioxidant and antimicrobial activities, and inhibition of lipid and protein oxidation in porcine patties. Journal of Agricultural and Food Chemistry, 59(10), 5625–5635. https://doi.org/10.1021/jf1048832
CrossRef - Basu, A., Rhone, M., & Lyons, T. J. (2010). Berries: Emerging impact on cardiovascular health. Nutrition Reviews, 68(3), 168–177. https://doi.org/10.1111/j.1753-4887.2010.00273.x
CrossRef - Williamson, G., & Clifford, M. N. (2017). Role of the small intestine, colon and microbiota in determining the metabolic fate of polyphenols. Biochemical Pharmacology, 139, 24–39. https://doi.org/10.1016/j.bcp.2017.03.012
CrossRef - He, X., & Liu, R. H. (2007). Triterpenoids isolated from apple peels have potent antiproliferative activity and may be partially responsible for apple’s anticancer activity. Journal of Agricultural and Food Chemistry, 55(11), 4366–4370. https://doi.org/10.1021/jf063563o
CrossRef - Vauzour, D., Rodriguez-Mateos, A., Corona, G., Oruna-Concha, M. J., & Spencer, J. P. E. (2010). Polyphenols and human health: Prevention of disease and mechanisms of action. Nutrients, 2(11), 1106–1131. https://doi.org/10.3390/nu2111106
CrossRef - Azmir, J., Zaidul, I. S. M., Rahman, M. M., Sharif, K. M., Mohamed, A., Sahena, F., Jahurul, M. H. A., Ghafoor, K., Norulaini, N. A. N., & Omar, A. K. M. (2013). Techniques for extraction of bioactive compounds from plant materials: A review. Journal of Food Engineering, 117(4), 426–436. https://doi.org/10.1016/j.jfoodeng.2013.01.014
CrossRef - Galanakis, C. M. (2012). Recovery of high added-value components from food wastes: Conventional, emerging technologies and commercialized applications. Trends in Food Science & Technology, 26(2), 68–87. https://doi.org/10.1016/j.tifs.2012.03.003
CrossRef - Ozturk, B., Parkinson, C., & Gonzalez-Miquel, M. (2018). Extraction of polyphenolic antioxidants from orange peel waste using deep eutectic solvents. Separation and Purification Technology, 206, 1–13. https://doi.org/10.1016/j.seppur.2018.05.052
CrossRef - Pandey, K. B., & Rizvi, S. I. (2009). Plant polyphenols as dietary antioxidants in human health and disease. Oxidative Medicine and Cellular Longevity, 2(5), 270–278. https://doi.org/10.4161/oxim.2.5.9498
CrossRef - D’Archivio, M., Filesi, C., Di Benedetto, R., Gargiulo, R., Giovannini, C., & Masella, R. (2007). Polyphenols, dietary sources and bioavailability. Annali dell’Istituto Superiore di Sanità, 43(4), 348–361.
- Manach, C., Scalbert, A., Morand, C., Rémésy, C., & Jiménez, L. (2004). Polyphenols: Food sources and bioavailability. The American Journal of Clinical Nutrition, 79(5), 727–747. https://doi.org/10.1093/ajcn/79.5.727
CrossRef - Giampieri, F., Tulipani, S., Alvarez-Suarez, J. M., Quiles, J. L., Mezzetti, B., & Battino, M. (2012). The strawberry: Composition, nutritional quality, and impact on human health. Nutrition, 28(1), 9–19. https://doi.org/10.1016/j.nut.2011.08.009
CrossRef - Ignat, I., Volf, I., & Popa, V. I. (2011). A critical review of methods for characterisation of polyphenolic compounds in fruits and vegetables. Food Chemistry, 126(4), 1821–1835. https://doi.org/10.1016/j.foodchem.2010.12.026
CrossRef - Capanoglu, E., Beekwilder, J., Boyacioglu, D., Hall, R. D., & de Vos, R. C. (2008). Changes in antioxidant and metabolite profiles during production of tomato paste. Journal of Agricultural and Food Chemistry, 56(3), 964–973. https://doi.org/10.1021/jf072990e
CrossRef - Dillard, C. J., & German, J. B. (2000). Phytochemicals: Nutraceuticals and human health. Journal of the Science of Food and Agriculture, 80(12), 1744–1756. https://doi.org/10.1002/1097-0010(20000915)80:12<1744::AID-JSFA725>0.CO;2-W
CrossRef - Jacob, J. K., & Paliyath, G. (2012). Chapter 12: Functional foods: Antioxidants and aging. In E. C. Y. Li-Chan, P. R. Jeon, & Y. Mine (Eds.), Bioactive foods and extracts: Cancer treatment and prevention(pp. 271–298). CRC Press.
Second Review by: Dr. Jennicav Jove
Final Approval by: Dr. Ayssar Nahle

















