Phytochemical and Antimicrobial Screening of Stem Bark of Thevetia Peruviana
Department of Applied Sciences, Maharaja Surajmal Institute of Technology, New Delhi, India
Corresponding AUthor E-mail:rekhatripathi@msit.in
DOI : http://dx.doi.org/10.13005/ojc/410503
ABSTRACT:This study examines the phytochemical and antimicrobial properties of methanolic extract of stem bark of Thevetia Peruviana. Methanol was used to extract the powdered stem bark that had been shade-dried, and once the solvent had been removed, the methanolic extract was put to column chromatography. α-amyrin, lupeol acetate, ursolic acid, thevefolin, and 3β, 6β, 19α-trihydroxyurs-12-en-28-oic acid (a new substance) were obtained through chemical analysis of the stem bark of this plant. Every single one of these compounds was characterized using spectral studies and physical attributes. The crude stem bark extract was tested for antibacterial and antifungal activity against selected bacteria and fungi by using the disc diffusion method. Significant antibacterial activity was demonstrated by the extract against E. coli (AI 0.87 at 1000 µg/disc and 0.72 at 500 µg/disc). Antifungal efficacy was demonstrated against A. niger (AI 0.63 at 1000 µg/disc and 0.45 at 500 µg/disc).
KEYWORDS:Antibacterial activity; Antifungal activity; Methanolic extract; Phytochemical analysis; Thevetia Peruviana stem bark
Introduction
Since the dawn of humankind, plants have been utilized for food and medicine. Because of the plants’ potential for healing, chemists have been interested in studying their natural compounds. In ancient times, the crude plant extracts were used to cure diseases. Later on, chemists isolated the active principles and established their structures.1-4
Thevetia genus belongs to the family Apocynaceae. Cardiac glycosides are predominant constituents of this genus; other chemical constituents such as terpenoids, flavonoids, steroids and fatty acids were isolated and characterized. Thevetia genus is considered a probable source of biologically active compounds, namely cardiotonic, insecticides, cytotoxics, rodenticides, and neuroprotection against ischemic stroke.5-11
Thevetia Peruviana is a small tree or bush, usually found in gardens or on the roadside as an ornamental plant, and has been referred to with different names as “milk bush”, “yellow oleander”, “lucky nut” and “be still tree”. It is widespread on the American, Asian, and African continents and is now cultivated throughout the tropical region, including India, Srilanka etc., and this plant does not require any maintenance.12 This plant is used in domestic medicine in tropical America and tropical Asia. All parts of this plant are toxic, particularly seeds that are poisonous and contain many cardiac glycosides, including thevetin A, thevetin B, neriifolin, and oleandrin.13,14 Despite its toxicity, different parts of this plant are being used traditionally as a local anesthetic, purgative, emetic, abortive, antineuralgic, and also for hemorrhoids, intermittent fever, constipation, acne, and reducing body weight.15-21 A tincture of stem bark is also useful in malarial fever and snakebite, and the latex is applied to relieve toothache and treat the ulcers also.
An analysis of the literature indicated that this plant contains a variety of alkaloids, steroids, volatile oils, flavonoids, and tannins, including β-sitosterol, epiperuviol acetate, hesperetin-7-glucoside, neolupenyl acetate, oleanolic acid, and ursolic acid. The plant possesses Insecticidal, Antibacterial, Antifungal, Anti-inflammatory, Antidiabetic, Antioxident, Anti-HIV, Molluscicidal, and Pesticidal activity.
Experimental
The plant material (root bark) was collected locally from Jaipur, Rajasthan. The shade-dried and powdered stem bark was exhaustively extracted with methanol on a water bath for 36 hrs, filtered, and concentrated under reduced pressure. The solvent-free gloomy green mass was redissolved in CHCl3 and precipitated by CH2=CHCN to remove fats.
Phytochemical Investigation
The non-fatty extract was column chromatographed over silica gel and eluted with increasing order of polarity of solvents in varying compositions. Isolation, purification, and characterization of 05 compounds were done on the basis of physical and spectral analysis (IR, 1H NMR, 13C NMR).
Isolation and identification
Isolation of α- Amyrin (compound A)
On elution with petroleum ether: benzene (3: 1) compound A was isolated and crystalized as white crystal with m.p. 185-1860C. The spectral data were observed as: IR (KBr) 3350,3200,1755,1735 (C=C), 1384, 1372, and 1060 cm-1(C-O) etc. and MS(m/z) 426[M+], 365,218,203 etc.
Isolation of Lupeol acetate (compound B)
Eluting the column with benzene yielded Lupeol acetate, crystalized with acetone as a colorless crystal, m.p. 204-2060C. Liebermann – Burchard test and TNM test confirmed it as unsaturated triterpene. The spectral data were observed as: IR (KBr) 2946(C-H stretching), 1740 (C=O), 1640 (C=C) cm-1 etc. and absorption at 468[M+], 426, 411, 315, 218 in mass spectra.
Isolation of Ursolic acid (compound C)
It was isolated by eluting the column by chloroform: ethyl acetate (3: 1) as white solid unsaturated triterpene with m.p. 246-2480C. The IR (KBr) spectrum exhibited absorption peaks at 3562 (O-H), 2637(O-H of carboxyl group), 1760(C=O), 1640(C=C) cm-1 and the mass spectra showed absorption at 456[M+], 455[M+H], 440, 419, 411, 390.
Isolation of Thevefolin (compound D)
On elution of the column with ethyl acetate: chloroform (3:1) compound D was isolated and crystallized with methanol as colorless crystals with the melting point 220-2210C. The spectral data observed as: IR (KBr) 3500(-OH), 1710(C=O), 1650, 1590(C=C) and 1060(C-O) cm-1 and (m/z) [M+] at 534.
Isolation of 3β, 6β, 19α-trihydroxyurs-12-en-28-oic acid (compound E)
Further elution of the column with ethyl acetate: methanol (3: 1) afforded compound E. It showed the m.p. 267-2720C. The isolated compound also gave a positive Liebermann – Burchard and TNM test to confirm its identity as unsaturated triterpene. The spectral were data observed as: IR (KBr) 3460-3400(O-H stretching), 2900, 2860(C-H stretching), 1734(C=O), 1650(C=C) and (m/z) [M-H]– at 487.
Antimicrobial Investigation
The crude methanolic extract of stem bark of Thevetia Peruviana was tested for bactericidal efficacy against Staphylococcus aureus, Escherichia coli, Salmonella paratyphi B, and Proteus vulgaris and for fungicidal activity against Aspergillus niger, Aspergillus flavus, Rhizoctonia bataticola, and Fusarium moniliforme using the disc diffusion method.21 Above microbial cultures were collected from SMS hospital Jaipur, Rajasthan, and SMS hospital typically uses microbial cultures sourced from the Microbial Type Culture Collection (MTCC), and the standards used for bactericidal and fungicidal activity were Amikacin and mycostatin respectively.
Results and Discussion
Phytochemical Analysis:
Mass spectra of the compound A gave a molecular ion peak at 426[M]+ corresponding to the molecular formula C30H50O. In the IR spectrum (cm-1, KBr) the broad absorption at 3350 confirmed the presence of the –OH group. The presence of unsaturation (C=C) and C-O stretching was confirmed by absorption at 1755-1735 and, 1060 respectively. 1H NMR showed sharp absorption for eight methyl groups in the region from 0.76 to 1.07. A proton attached at C-3 position was observed as a triplet at 4.45, confirming that the hydroxyl group is attached to C-3 carbon atom. Remaining 24 protons were observed in the region from 1.25 to 2.13.
In 13C NMR spectrum the absorption signal at 79.3 was assigned for C-3 carbon atom and confirms the presence of hydroxyl group. Two signals at 124.8 and 139.9 were due to olefinic carbons, i.e. C-12 and C-13 positions. All this above data confirmed the compound A as α-amyrin (Fig.1).23-24
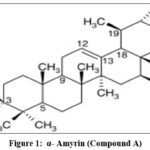 |
Figure 1: α- Amyrin (Compound A). Click here to View Figure |
The molecular ion peak of Compound B was observed at 468[M]+ and the molecular formula was calculated as C32H52O2. The presence of 52 protons and 32 carbon atoms was indicated by 1H NMR and 13CNMR respectively. The IR spectrum (cm-1, KBr) displayed absorptions at 2946 (C-H stretching), 1740 (C=O stretching), 1640 for C=C stretching. In 1HNMR spectra (δ ppm, CDCl3) a pair of broad singlet at 4.56 and 4.69 in conjugation with a singlet at 1.63 suggested the presence of an isopropenyl side chain. Sharp singlet at 2.04 explained the presence of 3 H+ of acetyl group (-COCH3) locate at C-3. In 13CNMR, the absorption at 81.37 also showed the presence of –OCOCH3 at C-3 position. The absorption at 171.39 and 23.7 confirmed the acetoxyl (-OCOCH3) group at position 3. The signals at 109.78 and 151.30 were observed for C-29 and C-20 carbon atoms, respectively. Hence compound B was identified as lupeol acetate. The compound was further confirmed by comparison of the spectral data with reported values (Fig.2).25
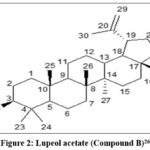 |
Figure 2: Lupeol acetate (Compound B)26 |
The compound C was established as C30H48O3 with the molecular ion peak at (m/z) 456 [M]+. In the IR spectrum (cm-1, KBr) broad absorption at 3562 showed the presence of hydroxyl group. The characteristic peak at 2637(–OH stretching of the carboxyl group and at 1760 (carbonyl group) and 1640 was due to >C=C< stretching. In the 1H NMR (δ ppm, CDCl3), a characteristic triplet was observed at 5.50 for one olefinic proton at C12-C13. The triplet at 3.49 indicated the presence of the –OH group at C-3. The presence of a single proton at C-16 was deduced from the triplet at 2.03. A broad doublet at 2.67 was assigned for the C-18 proton. 13C NMR (δ ppm, CDCl3) also confirmed the presence of (signals at 125.7 and 139.3) double bond in urs-12-en triterpenoid. The most downfield signal resonated at 179.7 and is attributed to the carboxylic acid (C-28). The spectral studies confirmed the identity of compound C as ursolic acid (Fig.3).27
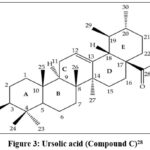 |
Figure 3: Ursolic acid (Compound C)28 |
The molecular formula of compound D was calculated as C30H46O8 with the molecular ion peak at (m/z) at 534 [M]+. The IR spectrum (cm-1, KBr) of the compound assured the presence of a hydroxyl group (absorption at 3500) and >C=C< by showing the absorption at 1650 and 1590. The presence of conjugated carbonyl and C-O-C stretching was confirmed by absorption at 1710 and 1060 respectively.
The proton NMR (δ ppm, CDCl3) showed sharp absorptions at 0.87(s, 3H), 0.96 (s, 3H) indicating the presence of two methyl groups at C-18 and C-19 respectively, which was also supported by 13CNMR. The signal at 5.88 (s, 1H, H-22), 4.96, 4.83 (each 1H, H-21a, H-21b) was due to butenolide of a cardenolide system. The presence of a singlet at 3.97 (m, 1H) was indicative of the attachment of a sugar moiety at C-3 position. In 13C NMR (δ ppm, CDCl3) presence of two signals at 171.34(C-23) and 160.1(C-20) indicated the α, β-unsaturated lactone of cardenolides. The signal at 98.9 was attributed to hydroxyl function at C-14. The anomeric sugar carbon signal was observed at 99.8, along with other sugar carbons at 72.6(C-2’), 83.1(C-3’), 72.4(C-4’), 69.3(C-5’), 17.8(C-6’), and 70.1(C-3’, -OCH3). The signals observed at 12.6 and 7.26 confirmed the presence of methyl groups at C-18 and C-19, respectively. These spectral data were in close proximity to those reported for thevefolin. Thus, the compound D was confirmed as thevefolin (Fig.4).29,30
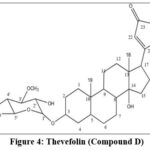 |
Figure 4: Thevefolin (Compound D). |
Compound E (white powder) showed a molecular ion peak at m/z 487 due to its [M-H]– ion, suggesting the molecular formula C30H48O5. In the infrared spectrum (cm-1, KBr), a broad spectrum band at 3460-3400 showed the presence of >O-H stretching. Characteristic absorption at 2900,2860 was due to >C-H stretching, absorption at 1734 indicated the carbonyl group, and stretching at 1650 revealed the presence of >C=C<. In the 1H NMR (δ ppm, CDCl3), a broad triplet at 5.11 was observed for the trisubstituted olifinic bond between C-12/C-13; this was also confirmed by two downfield signals at 128.5 and 139.4 in 13C NMR (δ ppm, CDCl3).
Table 1: Spectral data of 3β, 6β, 19α-trihydroxyurs-12-en-28-oic acid
| H/C | 1H | 13C |
| 1 | – | 41.7 |
| 2 | – | 26.5 |
| 3 | 4.46(t, 1H, oxymethine proton) | 78.6 |
| 4 | – | 39.5 |
| 5 | – | 56.5 |
| 6 | 4.43(t,1H, oxymethine proton) | 67.6 |
| 7 | – | 41.3 |
| 8 | – | 40.5 |
| 9 | – | 48.4 |
| 10 | – | 37.4 |
| 11 | – | 24.4 |
| 12 | 5.11(t, trisubsituted olifinic bond) | 128.5 |
| 13 | 139.4 | |
| 14 | – | 42.6 |
| 15 | – | 29.5 |
| 16 | – | 28.5 |
| 17 | – | 48.7 |
| 18 | 2.17(s, 1H) | 54.7 |
| 19 | 4.49(s, t-OH group) | 72.8 |
| 20 | – | 42.4 |
| 21 | – | 27.2 |
| 22 | – | 38.3 |
| 23 | 0.83(s,3H) | 28.6 |
| 24 | 0.84(s,3H) | 18.2 |
| 25 | 0.91(s,3H) | 17.3 |
| 26 | 1.13(d,3H) | 18.4 |
| 27 | 1.21(s,3H) | 24.7 |
| 28 | – | 180.7 |
| 29 | 0.96(d,3H) | 27.4 |
| 30 | 1.23(d,3H) | 16.6 |
Complete assignment of 1HNMR and 13CNMR is presented in Table 1; on this basis, the compound E was confirmed as 3β, 6β, 19α-trihydroxyurs-12-en-28-oic acid (Fig.5)31.
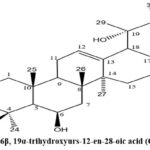 |
Figure 5: 3β, 6β, 19α-trihydroxyurs-12-en-28-oic acid (Compound E). |
Antimicrobial Analysis
The methanolic extract of stem bark of Thevetia Peruviana exhibited maximum antibacterial efficacy against E. coli (activity index 0.87 at 1000 µg/disc and 0.72 at 500 µg/disc). The extract also showed some activity against S. aureus (activity index 0.69 at 1000 µg/disc and 0.43 at 500 µg/disc), but only trace activity was observed against P. vulgaris and S. paratyphi B. In antifungal efficacy, only A. niger exhibited significant activity (activity index 0.63 at 1000 µg/disc and 0.45 at 500 µg/disc); otherwise, R. bataticola showed some activity at a high dose only[Table-2].
Table 2: Antimicrobial efficacy of methanolic extract of stem bark of Thevetia Peruviana
| Tested Bacteria | Tested Fungi | ||||||||
| Dose | S. aureus | E. coli | S.paratyphi B | P.vulgaris | A.niger | A.flavus | R.bataticola | F.moniliforme | |
|
1000µg/disc |
IZ | 13.0 | 18.0 | ± | ± | 13.0 | ± | 9 | ± |
| AI | 0.69 | 0.87 | 0.63 | 0.43 | |||||
|
500 µg/disc |
IZ | 8.0 | 15.0 | ± | ± | 9.0 | ± | ± | – |
| AI | 0.43 | 0.72 | 0.45 | ||||||
Standard: Amikacin = 10 µg/ml (bacteria); Mycostatin = 100 units/disc (fungi)
IZ – inhibition zone including the diameter of the disc (6mm)
AI – activity index (IZ of sample/ IZ of standard)
( ± ) Trace activity: ( -) No activity
Conclusion
α-Amyrin, lupeol acetate, ursolic acid, thevefolin, and 3β, 6β, 19α-trihydroxyurs-12-en-28-oic acid were isolated and characterized from the methanolic extract of the stem bark of Thevetia Peruviana. The extract possesses active principles that exhibited pronounced bactericidal efficacy against E. coli and S. aureus.
Acknowledgement
The authors gratefully acknowledge the Department of Chemistry at the University of Rajasthan for their provision of laboratory facilities.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement-
This statement does not apply to this article.
References
- Jain, R.; Jain, S.C.; Arora, R. Die Pharmazie , 1996,51(4), 253.
CrossRef - Tripathi, R.; Sharma, A. Oriental Journal of Chemistry, 2022, 38(5),1119-23. http://dx.doi.org/10.13005/ojc/380505
CrossRef - Jain, R.; Arora, R.; Jain, S.C. Indian Journal of Chemistry, 1997, 36(B), 61.
- Rakkini, A.M.; Rosaline, L.M.A.; Logapriya M.; Pavithra, A.R.; Joice, A.I. Oriental Journal of Chemistry, 2022,38(6),1396-403.
CrossRef - Alvarado-Cardenas L.O.; Ochoterena H.; Annals of the Missouri Botanical Garden, Missouri Botanical Garden, St. Louis; 2007, MO: 94, 300.
CrossRef - Alenshkina, Y.A.; Berezhinskaya, V.V. Farmakol Toksikol, 1962, 25, 720-5.
- Reed, D.K.; Freedman, B.; Ladd, T.L. J Econ Entomol., 1982,75,1093-97.
CrossRef - Cardellina, J.H; Gustofson, K.A.; Beutler, T.C.; Mckee, Y.W.; Hallock, F.; Boyd M. R. Human Medicinal Agents from Plants; American Chemical Society; Washington: 1993. P 218-41.
CrossRef - Oji, O.; Madubuike, F.N.; Ojimelukwe, P.C.; Ibeh, C.M. Herbs Spices Medicinal Plants, 1994, 2 ,3-10.
CrossRef - Oji, O.; Madubuike, F.N.; Nwalozie, M.C. Fitoterapia, 1993, 64, 137-9.
- James, K.T.; Stuart, P.; Mary, B.T.; Shwan, B.; Daniel, J.R.; Dexter, L.M.; David, C.L Nati Acad Sci USA; 2006, 103(27),10461. https://doi.org/10.1073/pnas.0600930103
CrossRef - Rajhans, S.; Pandya, J.; Mankad, A.U.; Pandya, H.A. International Journal of Scientific Research and Review, 2019, 8(3),1391-95.
- Aguilar, A.; Zolla, C. Editada por la subdirección; General Medica; 1982, 196.
- Omolara, O.O.; Ibiyemi, A.S.; Lamidi, A.U. Research Journal of Applied Sciences, 2007, 2, 188-91.
- Enriquez, M.E.M.; Guerrero, G.A.M.; Contreras, A.A.; Sepulveda A.E.C. West Pharmacol. Soc., 2006, 49, 69-71.
- Oji, O, Okafor, Q. E. Res., 2000, 14(2),133-5. https://doi.org/10.1002/(sici)1099-1573(200003)14:2%3C133::aid-ptr598%3E3.0.co;2-k
CrossRef - Langford, S.D.; Boor, P.J. Toxicology, 1996, 109, 1-13.
CrossRef - Parikh, C.K.; Textbook of medical jurisprudence and toxicology, 4,Ed., Medical Publication, Bombay, 1989.
- Schvartsman, S.; Plantas venenosas e animals peconhentos, 2Ed, Savier, Sao Paulo, 1992.
- Monroy, C.; Castillo, P.; Plantas medicinales empleadas en el estado de Morelos. CIB-UAEM, MEXICO, 2000.
- Martinez, M.E.; Moreno, L.A.; Luna, M.; Magos, G.A.; Aguilar, A.; Campos A.E. West. Pharmacol. Soc., 2002, 45,131-3.
- Kanwar, A.S.; Bhutani, K.K. Res., 2010, 24, 1562-66. https://doi.org/10.1002/ptr.3218
CrossRef - Wood, C.A.; Lee, K.; Vaisberg, A.J.; Kingston, D.G.; Neto, C.C.; Hammond, G.B. Pharm. Bull., 2001, 49, 1477-78.
CrossRef - Joshi, K.C.; Singh, P.; Pardasani, R.T. Ind. Chem. Soc. ,1979, 56 ,327-8.
- Gearien, J.E.; Klein, M. Pharm. Sci., 1975, 64,104-8.
CrossRef - Pereira Beserra, F.; Xue, M.; Maia, G.L.d.A.; Leite Rozza, A.; Helena Pellizzon, C.; Jackson, C.J. Molecules2018, 23, 2819. https://doi.org/10.3390/molecules23112819
CrossRef - Guvenalp, Z.; Kilic, N.; Kazaz, C.; Kaya, Y.; Demirezer, O. J. Chem., 2006, 30(4), 515-23. https://journals.tubitak.gov.tr/chem/vol30/iss4/12
- Mlala, S.; Oyedeji, A,; Gondwe, M.; Oyedeji, O. Molecules. 2019, https://doi.org/10.3390/molecules24152751
CrossRef - Miyagawa, T.; Ohtsuki, T.; Koyano, T.; Kowithayakorn, T.; Ishibashi, M. J Nat. Prod., 2009, 72(8), 1507-11. https://doi.org/10.1021/np900202n
CrossRef - Ali, M.; Ravinder, E.; Ramachandram, R. Pharmazie, 2000, 55, 385-9.
- Ma, B.; Liu, S.; Xie, Y.; Kano, Y.; Yuan, D. Asian Journal of Traditional Medicines, 2009, 4(3), 85-91.
Accepted on: 22 Sep 2025

















