Design, Formulation and Evaluation of Buccal Disintegrating Tablet Containing Anticonvulsant Agent
Oriental College of Pharmacy and Research, Oriental University, Indore, Madhay Pradesh India.
Corresponding Author E-mail: akotkarakshay1@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/410535
ABSTRACT:This study focuses on the formulation and evaluation of buccal disintegrating tablets (BDTs) of Clonazepam, an anticonvulsant drug, to enhance bioavailability, ensure rapid onset of action, and improve patient compliance, especially for non-cooperative or unconscious patients. Various superdisintegrants including Crospovidone, Croscarmellose Sodium (CCS), and Sodium Starch Glycolate (SSG) were utilized in combination to develop and compare formulations. Among the prepared batches, the formulation containing a combination of Crospovidone and CCS (F4) showed the most promising results in terms of disintegration time, drug release and stability.
KEYWORDS:Anticonvulsant; Buccal disintegrating tablet; Clonazepam; Crospovidone; Croscarmellose Sodium; Evaluation; Formulation; Superdisintegrants
Introduction
Buccal drug delivery systems have garnered significant interest as an alternative route to traditional oral administration, particularly for drugs with extensive first-pass metabolism or poor gastrointestinal absorption. Clonazepam, a benzodiazepine derivative, is commonly used in the treatment of epilepsy and other seizure disorders. However, its conventional oral administration may be limited by hepatic metabolism and slower onset of action.1 Buccal disintegrating tablets (BDTs) offer a solution by promoting rapid disintegration and absorption through the buccal mucosa. The inclusion of superdisintegrants enhances the dissolution profile, facilitating quicker therapeutic effect.2
Epilepsy is one of the most prevalent neurological disorders worldwide, affecting over 50 million people according to WHO estimates. It is characterized by recurrent seizures caused by abnormal electrical discharges in the brain. Quick and efficient drug delivery is critical in the management of epilepsy, especially during acute episodes where rapid onset of action is desired. 3, 4
Conventional oral dosage forms such as tablets and capsules often suffer from limitations like delayed onset, hepatic first-pass metabolism and difficulty in swallowing, particularly in pediatric, geriatric, or unconscious patients. Therefore, the development of alternative routes of administration has gained significant attention in pharmaceutical research.5, 6
Buccal Drug Delivery System
Buccal drug delivery involves the administration of drugs through the buccal mucosa (lining of the cheek) to achieve systemic effects. This route offers several advantages including:
Rapid drug absorption through the rich vascular supply of the buccal mucosa,
Avoidance of first-pass metabolism,
Easy administration without the need for water,
Improved patient compliance, especially in populations with swallowing difficulties.
Among various buccal dosage forms, Buccal Disintegrating Tablets (BDTs) have emerged as a promising platform. These tablets are designed to disintegrate quickly in the buccal cavity, allowing the drug to be absorbed directly into the systemic circulation.6, 7
Anticonvulsant Drugs and Their Importance
Anticonvulsant drugs are central to the management of epilepsy and seizure disorders. However, many of them undergo significant first-pass metabolism when administered orally, which reduces their bioavailability. Drugs like Clonazepam, Lamotrigine and Gabapentin have been studied for alternative routes of administration to improve therapeutic efficacy. By formulating anticonvulsants into buccal disintegrating tablets, it is possible to:
Achieve faster onset of action,
Improve bioavailability,
Enhance patient adherence to treatment,
Reduce dose frequency and side effects.8, 9
Need for the Study
There is a clinical need for novel dosage forms that provide rapid onset, especially in seizure emergencies. Buccal disintegrating tablets combine the benefits of quick action and ease of administration. Despite the potential, there is limited commercial availability of buccal formulations for anticonvulsant drugs.
This study aims to bridge this gap by developing and evaluating BDTs of an anticonvulsant drug, ensuring optimal disintegration time, mechanical strength, drug release, and stability.
Objectives
The primary objective of this research work is the design, formulation, and evaluation of buccal disintegrating tablets (BDTs) of a selected anticonvulsant drug to enhance therapeutic efficacy and overcome limitations associated with conventional oral dosage forms.
Specific Objectives
To improve the bioavailability of the selected anticonvulsant drug by bypassing the hepatic first-pass metabolism through buccal delivery.
To achieve a specific site of action by targeting the buccal mucosa for rapid drug absorption into the systemic circulation.
To enhance patient compliance, especially in pediatric, geriatric, and unconscious patients who may face difficulty in swallowing conventional tablets.
To provide a faster onset of therapeutic action in acute seizure conditions through rapid disintegration and absorption via the buccal route.
To increase the disintegration rate of the dosage form by incorporating Superdisintegrants, thereby enabling immediate drug release.
Advantages of Buccal Disintegrating Tablets
Bypass of first-pass metabolism.
Faster onset of action compared to traditional oral tablets.
Improved bioavailability& enhanced patient compliance.
Suitable for emergency situations.8, 10, 11
Preformulation Studies
Organoleptic Characters of Drug12
Solubility Study13
Melting Point Determination12, 14
Micromeritics Properties of Clonazepam8, 14
Calibration Curve of Clonazepam15, 16
FTIR Studies17
Organoleptic Characters of Drug
Table 1: Organoleptic Characters of Clonazepam
| Parameter | Observation | Method of Evaluation |
| Appearance | Crystalline powder | Visual inspection under daylight |
| Color | White to pale yellow | Visual observation |
| Odor | Odorless | Sensory evaluation |
| Taste | Slightly bitter | Gustatory evaluation (<1 mg sample) |
| Texture | Fine, free-flowing powder | Manual touch evaluation |
The above observed organoleptic properties of Clonazepam were consistent with those reported in pharmacopeial monographs, confirming the authenticity and quality of the raw drug before further analytical and formulation studies.
Solubility Study
Clonazepam is poorly water-soluble, and solubility can be enhanced by using surfactants like Sodium Lauryl Sulfate (SLS) in the formulation.
Table 2; Solubility Study of Clonazepam
| Solvent | Solubility |
| Water | Slightly soluble |
| Ethanol | Soluble |
| Methanol | Soluble |
| Acetone | Freely soluble |
| Phosphate buffer (pH 6.8) | Slightly soluble |
Melting Point Determination
Observed Melting Point 240 ± 2°C, which matches the standard reported melting point of Clonazepam. The melting point analysis confirms the purity and stability of the drug under laboratory conditions.
Micromeritics Properties of Clonazepam
The micromeritic properties of the powder blends (F1–F6) were evaluated and the results are shown in Table X. The angle of repose values ranged from 31.0° to 37.6°, indicating that the flow properties of the blends varied from excellent to fair.
Table 3: Micromeritics Properties
| Formulation Code | Angle of Repose | Bulk Density | Tapped Density | Carr’s Index | Hausner’s Ratio |
| F1 | 32.6° | 0.456 | 0.612 | 17.85 | 1.22 |
| F2 | 35.8° | 0.444 | 0.618 | 23.64 | 1.31 |
| F3 | 37.6° | 0.450 | 0.615 | 18.20 | 1.23 |
| F4 | 31.0° | 0.471 | 0.632 | 12.68 | 1.14 |
| F5 | 33.2° | 0.433 | 0.620 | 16.92 | 1.21 |
| F6 | 35.8° | 0.462 | 0.617 | 17.25 | 1.25 |
The bulk density values were found between 0.433–0.471 g/cm³, while the tapped density values ranged from 0.612–0.632 g/cm³. The Carr’s Index values were observed in the range of 12.68–23.64%, and the Hausner’s ratio values ranged between 1.14–1.31. Among all formulations, F4 demonstrated the best micromeritics properties, ensuring better flow, handling, and compressibility of the powder blend. This suggests that F4 may provide more consistent weight variation and tablet quality during compression compared to other formulations.
Calibration Curve of Clonazepam
A calibration curve was constructed using standard solutions of the drug in the concentration range of 2–12 µg/mL. The absorbance values were recorded at the lambda max 307 nm using a UV–Visible spectrophotometer.
Table 4: Absorbance at the lambda max 307 nm
| Sample No. | Concentration (μg/ml) | Absorbance at 307nm |
| 1. | 2 | 0.14 |
| 2. | 4 | 0.28 |
| 3. | 6 | 0.42 |
| 4. | 8 | 0.56 |
| 5. | 10 | 0.70 |
| 6. | 12 | 0.84 |
Equation of Line: y = 0.078x + 0.001
Where y = absorbance and x = concentration (µg/ml)
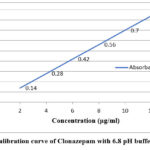 |
Figure 1: Calibration curve of Clonazepam with 6.8 pH buffer at 307 nm.
|
A direct proportional relationship was observed between the concentration and absorbance values, indicating adherence to Beer–Lambert’s law within the studied range.
FTIR Studies
The Confirm the identity of Clonazepam by its characteristic FTIR peaks. Assess possible chemical interactions between Clonazepam and selected excipients (Crospovidone, Croscarmellose sodium, Sodium starch glycolate, MCC, Mannitol, SLS, Aspartame, Mg stearate, Talc) via comparison of FTIR spectra of pure drug, excipients and physical mixtures.
The IR Spectrum preview pictures are as follows:
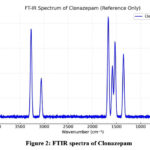 |
Figure 2: FTIR spectra of Clonazepam.
|
Table 5: Identification of Clonazepam with I.R. Spectrum
| Sr.No. | Functionalgroup | Wave number(cm⁻¹) |
| 1. | N–H stretching | 3390 |
| 2. | Aromatic C–H stretching | 3080 |
| 3. | C=O stretching | 1685 |
| 4. | Aromatic C=C stretching | 1605 |
| 5. | C–N stretching | 1320 |
| 6. | C–Cl / ring deformation | 1020 |
No significant shift or disappearance of peaks was observed in the physical mixture compared to the pure drug, indicating no major chemical interactions between Clonazepam and the selected excipients and Conclusion is drug was found to be compatible with the excipients used.
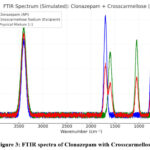 |
Figure 3: FTIR spectra of Clonazepam with Crosscarmellose
|
Table 6: Identification of Clonazepam with Crosscarmellose I.R. Spectrum
| Component | Functionalgroup | Wave number(cm⁻¹) | Observation in Mixture |
| Clonazepam | N–H stretching | 3400 | Present; no significant shift |
| Clonazepam | C=O (amide) | 1685–1710 | Present; position retained |
| Clonazepam | Aromatic C=C bend | 590–1610 | Present; minor broadening only |
| Clonazepam | C–Cl related | 750–780 | Present; intensity comparable |
| Crosscarmellose | O–H stretching | 3300–3500 | Present; overlaps with API N–H |
| Crosscarmellose | COO⁻ asym. stretch | 1600–1625 | Present; no significant shift |
| Crosscarmellose | COO⁻ sym. stretch | 1410–1440 | Present; no significant shift |
| Crosscarmellose | C–O stretch | 1020–1060 | Present; retained |
The absence of new absorption bands, no substantial peak shifting, and no disappearance of diagnostic functional-group peaks of Clonazepam in the presence of Crosscarmellose indicate no chemical interaction under the tested conditions. Therefore, Clonazepam and Crosscarmellose sodium are compatible for formulation development.
 |
Figure 4: FTIR spectra of Clonazepam with Crospovidone
|
Table 7: Identification of Clonazepam with Crospovidone I.R. Spectrum
| Component | Functionalgroup | Wave number(cm⁻¹) | Observation in Mixture |
| Clonazepam | N–H stretching | 3400 | Present, retained |
| Clonazepam | C=O stretching | 1700 | Present, retained |
| Clonazepam | C–Cl stretching | 750 | Present, retained |
| Crospovidone | O–H stretching | 3400 | Present, broad peak retained |
| Crospovidone | C=O stretching | 1660 | Present, retained |
| Crospovidone | C–N vibrations | 1280 | Present, retained |
| Crospovidone | C–H stretching | 2920 | Present, retained |
| Physical Mixture (1:1) | Combination of above | Overlapping but no new peaks | No new peaks; no significant shifts → Compatible |
The retention of major functional group peaks (N–H, C=O, C–Cl for Clonazepam and C=O, O–H, C–N for Crospovidone) in the mixture spectrum indicates that no chemical interaction occurred between the drug and Crospovidone. Minor variations in peak intensity can be attributed to physical mixing and overlapping of bands. Thus, FTIR analysis confirmed the compatibility of Clonazepam with Crospovidone.
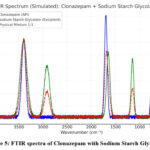 |
Figure 5: FTIR spectra of Clonazepam with Sodium Starch Glycolate.
|
Table 8: Identification of Clonazepam with Sodium Starch Glycolate I.R. Spectrum
| Component | Functional group | Wave number (cm⁻¹) | Observation in Mixture |
| Clonazepam | N–H stretching | 3400 | Present, retained |
| Clonazepam | C=O stretching | 1700 | Present, retained |
| Clonazepam | C–Cl stretching | 750 | Present, retained |
| Sodium Starch Glycolate | O–H stretching | 3400 | Present, retained |
| Sodium Starch Glycolate | C–O stretching | 1020 | Present, retained |
| Sodium Starch Glycolate | CH₂ vibrations | 1420 | Present, retained |
| Physical Mixture (1:1) | Combination of above peaks | All above | No new peaks; no significant shifts → Compatible |
The preservation of major functional group bands of both Clonazepam and Sodium Starch Glycolate in the mixture indicates the absence of chemical interaction between the drug and excipient. Hence, FTIR analysis confirmed that Clonazepam is compatible with Sodium Starch Glycolate.
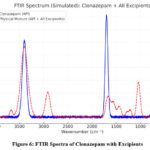 |
Figure 6: FTIR Spectra of Clonazepam with Excipients.
|
Table 9: Identification of Clonazepam with Excipients I.R. Spectrum
| Component | Functional group | Wave number (cm⁻¹) | Observation in Mixture |
| Clonazepam | N–H Stretch | 3400 | Present, no shift |
| Clonazepam | C=O Stretch (amide) | 1700 | Present, no shift |
| Clonazepam | C–Cl Stretch | 750 | Present, no shift |
| Crospovidone | C=O, C–N | 1660, 1280 | Overlaps, no new peak |
| Croscarmellose | O–H, COO⁻, C–O | 3400, 1600, 1050 | Present, merged |
| SSG | O–H, C–O, COO⁻ | 3400, 1420, 1020 | Present, merged |
| MCC | O–H, C–O–C | 3400, 1050 | Present, merged |
| Mannitol | O–H, C–O, C–H | 3400, 1050, 2920 | Present, merged |
| Aspartame | O–H/N–H, C=O, COO⁻ | 3400, 1650, 1550 | Present, merged |
| SLS | S=O, S–O–C, C–H | 1210, 1050, 2920 | Present, merged |
| Mg Stearate | C–H, COO⁻ | 2900, 1550–1465 | Present, merged |
| Talc | Mg–OH, Si–O | 3675, 1010 | Present, merged |
All the characteristic peaks of the API (Clonazepam) such as N–H, C=O, and C–Cl are found to be intact in the physical mixture. The peaks of the excipients are also observed in the mixture, but no new peaks have appeared. This indicates that the API and excipients are chemically compatible, and only physical mixing has occurred.
Preparation of Buccal Disintegrating Tablet
ethod of Formulation (Direct Compression Method)18,19
To formulate and evaluate buccal disintegrating tablets of Clonazepam using different superdisintegrants by the direct compression method, which is simple, cost-effective, and suitable for moisture-sensitive drugs.
Step 1: Weighing of Ingredients
All ingredients required for each formulation (F1 to F6) were accurately weighed using an electronic balance according to the pre-calculated formula.
Step 2: Sifting
All ingredients, including Clonazepam, superdisintegrants (Crospovidone, Croscarmellose sodium, and Sodium starch glycolate), Microcrystalline Cellulose (MCC), Mannitol, Aspartame, and Sodium Lauryl Sulfate (SLS) were passed through sieve #40 to ensure uniform particle size and remove any lumps.
Step 3: Mixing
The sifted ingredients were geometrically mixed in a mortar and pestle for 10–15 minutes to obtain a uniform blend.
Step 4: Addition of Lubricants
Magnesium stearate and talc (lubricant and glidant) were added last and mixed gently for 2–3 minutes to avoid over-lubrication, which may affect tablet disintegration.
Step 5: Compression
The final blend was directly compressed into tablets using a rotary tablet punching machine equipped with 6 mm flat-faced punches.
Step 6: Storage
prepared tablets were stored in airtight containers at ambient room temperature (25 ± 2°C) for further evaluation studies.
Formulation Table
Formulation of Buccal Disintegrating Tablets of Clonazepam Using Different Superdisintegrants
Table 10: Formulation Composition
| Sr. No. | Ingredients(mg/tablet) | F1 | F2 | F3 | F4 | F5 | F6 | Function |
| 1. | Clonazepam | 1 | 1 | 1 | 1 | 1 | 1 | Active Ingredient |
| 2. | Crospovidone | 6 | – | – | 3 | – | 3 | Superdisintegrant |
| 3. | Croscarmellose Sodium | – | 6 | – | 3 | 3 | – | Superdisintegrant |
| 4. | Sodium Starch Glycolate | – | – | 6 | – | 3 | 3 | Superdisintegrant |
| 5. | Microcrystalline Cellulose | 45 | 45 | 45 | 45 | 45 | 45 | Diluent |
| 6. | Mannitol | 40 | 40 | 40 | 40 | 40 | 40 | Sweetener + Mouth feel |
| 7. | Aspartame | 2 | 2 | 2 | 2 | 2 | 2 | Sweetener |
| 8. | Sodium Lauryl Sulfate (SLS) | 1 | 1 | 1 | 1 | 1 | 1 | Surfactant for solubility |
| 9. | Magnesium Stearate | 2 | 2 | 2 | 2 | 2 | 2 | Lubricant |
| 10. | Talc | 2 | 2 | 2 | 2 | 2 | 2 | Glidant |
Results and Discussion
Weight Variation20,21
Thickness Uniformity 22
Hardness Test23
Friability24
Swelling Studies25
Surface Potential Hydrogen (pH) Study26, 27
Stability Study28
In-Vitro Dissolution Study29, 30
Assay of Clonazepam Buccal Disintegrating Tablet31
Disintegration Test32
Animal Study of Anticonvulsant Activity (MES Model) [23, 34, 35]
Weight Variation
Table 11: Weight Variation
| Formulation | Avg. Wt. (mg) ± SD | % Deviation Range | Pass/Fail |
| F1 | 101.2 ± 1.8 | 98.5 – 103.6 | Pass |
| F2 | 100.5 ± 1.5 | 98.7 – 102.9 | Pass |
| F3 | 99.8 ± 1.6 | 97.4 – 101.9 | Pass |
| F4 | 100.8 ± 1.2 | 99.0 – 102.4 | Pass |
| F5 | 101.5 ± 1.7 | 99.2 – 103.7 | Pass |
| F6 | 100.2 ± 1.4 | 98.1 – 102.6 | Pass |
All formulations (F1–F6) showed uniform tablet weights with deviations well within pharmacopeial limits (±7.5%), The lowest standard deviation was observed in F4, indicating excellent weight uniformity.
Thickness Uniformity
Table 12: Thickness Uniformity
| Formulation | Average Thickness (mm) |
| F1 | 2.58 |
| F2 | 2.61 |
| F3 | 2.59 |
| F4 | 2.62 |
| F5 | 2.60 |
| F6 | 2.61 |
All formulations (F1–F6) showed uniform thickness ranging from 2.58 to 2.62 mm; Results comply with standard pharmacopeial requirements for tablet thickness uniformity.
Hardness Test
Table 13: Hardness Test
| Formulation | Average Hardness (kg/cm²) |
| F1 | 2.7 |
| F2 | 2.8 |
| F3 | 2.9 |
| F4 | 3.0 |
| F5 | 2.8 |
| F6 | 2.9 |
The hardness of formulations ranged between 2.7–3.0 kg/cm², which is within the optimum range for buccal disintegrating tablets, F4 showed the most consistent hardness 3.0 kg/cm² reflecting uniform compression and mechanical strength , all formulations complied with the requirement of providing sufficient strength for handling while maintaining fast disintegration.
Friability
Table 14 Friability (%)
| Formulation | Friability (%) |
| F1 | 0.56 |
| F2 | 0.68 |
| F3 | 0.50 |
| F4 | 0.42 |
| F5 | 0.48 |
| F6 | 0.60 |
All formulations (F1–F6) exhibited friability well below the pharmacopeia limit of 1.0%, indicating good mechanical integrity during handling and packaging and F4 showed the lowest friability (0.42%), consistent with its optimal hardness and compressibility characteristics this suggests the best resistance to abrasion among the tested batches.
Swelling Studies
Table 15: Swelling Studies
| Time (min) | F1 | F2 | F3 | F4 | F5 | F6 |
| 5 | 12.4 | 10.8 | 13.5 | 15.2 | 11.5 | 12.8 |
| 10 | 18.6 | 16.4 | 19.2 | 21.5 | 17.2 | 18.5 |
| 15 | 22.8 | 20.1 | 23.6 | 26.8 | 21.5 | 22.9 |
| 20 | 25.2 | 22.7 | 26.4 | 29.4 | 23.8 | 25.6 |
| 30 | 27.5 | 24.6 | 28.9 | 32.0 | 25.9 | 28.2 |
All formulations showed progressive swelling with time, confirming the hydrophilic nature of the selected Superdisintegrants (Crospovidone, Croscarmellose Sodium, Sodium Starch Glycolate) F4 exhibited the highest swelling index (32% at 30 min), indicating better fluid uptake and rapid disintegration capability compared to other batches, Swelling studies demonstrated that the optimized formulation F4 showed maximum swelling index and fastest hydration, which supports its enhanced disintegration performance and drug release efficiency in the buccal cavity.
Surface Potential Hydrogen (pH) Study
Table 16: Surface Potential Hydrogen pH
| Formulation | Surface pH |
| F1 | 6.7 |
| F2 | 6.6 |
| F3 | 6.5 |
| F4 | 6.8 |
| F5 | 6.7 |
| F6 | 6.6 |
All formulations (F1–F6) exhibited surface pH in the range of 6.5–6.8, which is within the acceptable buccal range, the optimized batch F4 showed a surface pH of 6.8, closest to neutrality, thereby minimizing the risk of irritation to the buccal mucosa.
Stability Study
The optimized buccal disintegrating tablet of Clonazepam under controlled conditions and to detect any early degradation, physical change and Assay and Drug Release as follows:
Table 17: Stability Parameters
| Condition / Time | Appearance | Avg. Wt. (mg) | Hardness (N) | Friability (%) | Disintegration (s) | Surface pH |
| 0 month | White, smooth, no cracks | 99.2 ± 2.1 | 45 ± 3 | 0.35 | 38 ± 4 | 6.7 ± 0.1 |
| 1 M (25°C/60% RH) | No change | 98.9 ± 2.3 | 44 ± 3 | 0.38 | 40 ± 5 | 6.7 ± 0.1 |
| 3 M (25°C/60% RH) | No change | 98.6 ± 2.5 | 44 ± 4 | 0.40 | 42 ± 4 | 6.6 ± 0.1 |
| 1 M (40°C/75% RH) | No change | 98.7 ± 2.4 | 43 ± 4 | 0.42 | 44 ± 5 | 6.6 ± 0.2 |
| 3 M (40°C/75% RH) | Slight increase in friability, acceptable | 98.5 ± 2.6 | 43 ± 4 | 0.48 | 46 ± 5 | 6.6 ± 0.2 |
Table 18: Stability parameters (Assay and Drug Release)
| Condition / Time | Assay (% of label claim) | % Drug Release at 15 min |
| 0 month | 99.2 | 96.8 |
| 1 M (25°C/60% RH) | 98.7 | 96.1 |
| 3 M (25°C/60% RH) | 98.3 | 95.7 |
| 1 M (40°C/75% RH) | 97.9 | 95.4 |
| 3 M (40°C/75% RH) | 95.6 | 94.1 |
The buccal disintegrating tablet formulation of Clonazepam exhibited excellent stability under both long-term and accelerated conditions, Only minor changes were observed in disintegration time and hardness at accelerated storage, but these remained within pharmacopeial limits. Drug content and dissolution profiles confirmed no significant degradation of the active drug or loss of performance.
The buccal disintegrating tablet formulation of Clonazepam was stable up to 6 months under accelerated and long-term conditions without significant changes in physical, chemical, or functional characteristics. Based on assay and dissolution data, the projected shelf-life is at least 24 months when stored in under controlled room temperature.
In-Vitro Dissolution Study
The dissolution profiles of all six formulations (F1–F6) are presented in Table and Figure Among the formulations, F4 exhibited the highest drug release (98% at 10 min), followed by F5 and F6 (97% at 10 min), while F3 showed the lowest release (89% at 10 min).
Table 19: Cumulative % Drug Release
| Time (min) | F1 | F2 | F3 | F4 | F5 | F6 |
| 0.5 | 18 ± 1.2 | 22 ± 1.1 | 15 ± 1.3 | 28 ± 1.5 | 24 ± 1.2 | 23 ± 1.1 |
| 1 | 32 ± 1.5 | 38 ± 1.4 | 26 ± 1.5 | 46 ± 1.6 | 40 ± 1.5 | 39 ± 1.3 |
| 2 | 48 ± 1.6 | 56 ± 1.5 | 42 ± 1.6 | 66 ± 1.8 | 59 ± 1.6 | 57 ± 1.5 |
| 4 | 65 ± 1.8 | 73 ± 1.6 | 58 ± 1.7 | 85 ± 1.9 | 77 ± 1.7 | 75 ± 1.6 |
| 6 | 80 ± 1.7 | 88 ± 1.5 | 73 ± 1.8 | 95 ± 1.5 | 91 ± 1.6 | 89 ± 1.6 |
| 8 | 90 ± 1.6 | 94 ± 1.4 | 84 ± 1.7 | 97 ± 1.2 | 96 ± 1.4 | 95 ± 1.5 |
| 10 | 94 ± 1.4 | 97 ± 1.3 | 89 ± 1.6 | 98 ± 0.9 | 97 ± 1.3 | 97 ± 1.3 |
The superior performance of F4 may be attributed to the optimized combination of crospovidone and croscarmellose sodium, which synergistically enhanced water uptake, swelling, and rapid disintegration of the tablet matrix. The faster dissolution observed with F4 suggests improved drug availability at the buccal site, making it the most promising formulation among the six.
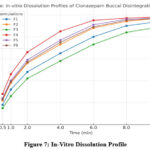 |
Figure 7: In-Vitro Dissolution Profile
|
Assay of Clonazepam Buccal Disintegrating Tablet
Table 20: Assay of Clonazepam Buccal Tablets
| Formulation | % Drug Content (Mean ± SD) |
| F1 | 98.5 ± 0.9 |
| F2 | 98.8 ± 0.8 |
| F3 | 97.2 ± 0.8 |
| F4 | 99.0 ± 0.7 |
| F5 | 98.6 ± 0.6 |
| F6 | 98.9 ± 0.7 |
The assay results confirmed that all formulations contained drug content within the pharmacopeial acceptance criteria of 95–105%.The drug content values ranged from 97.2% to 99.0%, which indicates uniform distribution of clonazepam in the buccal disintegrating tablets. The formulation F4 showed the highest assay value (99.0%) No significant deviations were observed across batches, confirming that the selected excipients and direct compression technique provided consistent drug loading.
Disintegration Test
All formulations disintegrated well within the pharmacopeial limit of 3 minutes, indicating their suitability as buccal disintegrating tablets. Among them, F4 showed the fastest disintegration (42 sec), which correlates with its superior in-vitro dissolution performance.
Table 21: Disintegration Time Tablets
| Formulation | Disintegration Time (sec) |
| F1 | 58 |
| F2 | 50 |
| F3 | 65 |
| F4 | 42 |
| F5 | 48 |
| F6 | 46 |
This enhanced disintegration can be attributed to the synergistic action of Crospovidone and Croscarmellose Sodium as superdisintegrants. Thus, F4 was confirmed as the optimized batch due to its shortest disintegration time, rapid drug release, and consistent assay values.
Animal Study of Anticonvulsant Activity (MES Model)
The anticonvulsant activity of the optimized buccal disintegrating tablet (BDT) of clonazepam was evaluated in Swiss Albino mice using the Maximal Electroshock Seizure (MES) model.
Table 22: Effect of Clonazepam Formulations on MES-Induced Seizures in Mice
| Group | Treatment | Dose (mg/kg) | THLE Duration (sec, Mean ± SD) | Onset Latency (sec, Mean ± SD) | % Protection |
| G1 | Control (Vehicle) | — | 14.2 ± 0.8 | 2.1 ± 0.2 | 0 |
| G2 | Pure Drug Suspension | 1.0 | 6.4 ± 0.5 | 4.3 ± 0.4 | 66.7 |
| G3 | Formulations BDT | 1.0 | 2.8 ± 0.3 | 6.1 ± 0.5 | 83.3 |
Control group showed typical tonic hind limb extension (THLE) of 14 seconds, confirming seizure induction. Pure drug suspension (Clonazepam 1.0 mg/kg) significantly reduced THLE duration and increased latency compared to control, with 67% protection. Formulated tablet (Clonazepam 1.0 mg/kg) showed the best anticonvulsant effect, with shortest THLE (2.8 sec), highest onset latency (6.1 sec), and 83.3% protection. The results demonstrate that buccal disintegrating tablets improve drug performance, likely due to rapid absorption through the buccal mucosa, bypassing first-pass metabolism, and faster onset of action.
Conclusion
The present research work successfully demonstrated the formulation and evaluation of buccal disintegrating tablets (BDTs) of clonazepam using the direct compression technique. A systematic Preformulation, formulation, and evaluation approach led to the development of an formulation (F4), which exhibited superior physicochemical and pharmacodynamics performance compared to other batches. The formulation (F4) of clonazepam buccal disintegrating tablets exhibited rapid disintegration, enhanced dissolution, excellent stability, and superior anticonvulsant activity compared to other batches. These results confirm that buccal delivery of clonazepam offers significant advantages, including bypassing first-pass metabolism, faster onset of action, improved bioavailability, and better patient compliance.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
References
- Sakthivel, M., Halith, S. M., Devi, G. H., Harish, P., Kumar, M. P., & Vinith, B. (2024). Overview of buccal drug delivery system. International Journal of Pharmaceutical Sciences Review and Research, 84(11), 121–124.
CrossRef - Shruthi, B. K., Chandrakala, V., & Srinivasan, S. (2022). Role of superdisintegrants in rapid dissolving oral films. International Journal of Pharmaceutical Sciences Review and Research, 75(2), 110–116.
CrossRef - World Health Organization. (2024, February 7). Epilepsy. https://www.who.int/news-room/fact-sheets/detail/epilepsy
- National Institute of Neurological Disorders and Stroke. (n.d.). Epilepsy and seizures. U.S. Department of Health and Human Services. Retrieved , from https://www.ninds.nih.gov/health-information/disorders/epilepsy-and-seizures
- Gothainayaki, C., & Rajalakshmi, A. N. (2019). Fixed dose combination products as oro-dispersible tablets: A review. Journal of Drug Delivery and Therapeutics, 9(2), 563–573. https://doi.org/10.22270/jddt.v9i2.2452
CrossRef - Akotkar, A. M., Bais, N., & Jain, S. (2025). Formulation and evaluation of buccal disintegrating tablet containing anticonvulsant drug. Journal of Neonatal Surgery, 14(32S), 257-266. https://doi.org/10.47310/jneonatalsurg.2025.7358.
- Pathak, B., & Kumar, K. (2017). Buccal drug delivery system: A tool for the effective delivery of pharmaceuticals. Universal Journal of Pharmaceutical Research, 2(3), 52–59.
CrossRef - Akotkar, A. M., Zanke, A. A., & Ghonge, A. B. (2022) “Formulation and Evaluation of Buccal Disintegrating Tablet of Anticonvulsant Drug” Asian Journal of Research in Pharmaceutical Sciences. 12(2): April – June, 2022.
CrossRef - Anderson, G. D., & Saneto, R. P. (2012). Current oral and non-oral routes of antiepileptic drug delivery. Advanced Drug Delivery Reviews, 64(10), 911–918.
CrossRef - Sabra, R., Kirby, D., Chouk, V., & Mohammed, A. R. (2024). Buccal Absorption of Biopharmaceutics Classification System III Drugs: Formulation Approaches and Mechanistic Insights. Pharmaceutics, 16(12), 1563.
CrossRef - Patel, R. J., Rajabi, M., & Singh, P. (2021). An updated overview on mucoadhesive buccal drug delivery system. Research Journal of Pharmacy and Technology, 14(8), 3382-3390.
- Nagpure, S. R., & Jadhav, P. B. (2018). Formulation development and evaluation of gabapentin buccal tablets. International Journal of Innovative Pharmaceutical Sciences and Research, 6(6), 13–22. ISSN 2347-2154.
- Simonelli, A., Aboutaleb, A., & Abdel-Rahman, A. (1984). Solubilization and stability of clonazepam by different classes of surfactants. Bulletin of Pharmaceutical Sciences, Assiut University, 7(2), 363-379.
CrossRef - Govindasamy, K. (2011). Formulation and evaluation of gabapentin oral controlled release matrix tablets (Master’s thesis, The Tamil Nadu Dr. M.G.R. Medical University, Chennai). Department of Pharmaceutics Reg. No: 26106811, Page No.54.
- Pimlott SJ, Addy M. Site Dependent Absorption Study on Buccal Mucosae. Oral Surg Oral Med Oral Pathol Oral RadiolEndod 1985; 59:145-48.
CrossRef - Miri, H., & Jalali, N. (2013). Dispersive liquid-liquid micro-extraction as a sample preparation method for the determination of clonazepam in pharmaceutical preparations and water samples. Journal of Research in Pharmaceutical Sciences, 2(2), 103-110.
CrossRef - Hearnden V, Sanker V, Hull K, Jural DV, Greenberg M, Kerr RA, Lockhart PA. New Developments and Opportunities in Oral Mucosal Drug Delivery for Local and Systemic Disease. Advance Drug Delivery Reviews. 2011; 1-13.
CrossRef - Govindasamy, K. (2011). Formulation and evaluation of gabapentin oral controlled release matrix tablets (Master’s thesis, The Tamil Nadu Dr. M.G.R. Medical University, Chennai). Department of Pharmaceutics Reg. No: 26106811, Page No.52.
- Timmermans, J., & Moes, A. J. (1994). Factors controlling the buoyancy and gastric retention capabilities of floating matrix capsules: New data for reconsidering the controversy. Journal of Pharmaceutical Sciences, 83(1), 18–24.
CrossRef - Indian Pharmacopoeia Commission (IPC). (2018). Indian Pharmacopoeia 2018 (Vol. II, pp. 212). Ghaziabad, India: Government of India, Ministry of Health and Family Welfare.
- United States Pharmacopeia. (2021). United States Pharmacopeia and National Formulary (USP 43–NF 38). The United States Pharmacopeial Convention.
- Avis, K. E., Lieberman, H. A., & Lachman, L. (1989). Pharmaceutical dosage forms: Tablets (3rd ed., Vol. 1, pp. 42–56). New York, NY: Marcel Dekker.
- Lachman, L., Lieberman, H. A., & Kanig, J. L. (2009). The theory and practice of industrial pharmacy (4th ed., pp. 293–296). New Delhi, India: CBS Publishers & Distributors.
- Indian Pharmacopoeia Commission (IPC). (2018). Indian Pharmacopoeia 2018 (Vol. II, pp. 204–205). Ghaziabad, India: Government of India, Ministry of Health and Family Welfare.
- Robinson, J. R., & Lee, V. H. L. (1987). Controlled drug delivery: Fundamentals and applications (Vol. 29). New York, NY: Dekker.
CrossRef - Shipp, L., Liu, F., Kerai-Varsani, L., & Okwuosa, T. C. (2022). Buccal films: A review of therapeutic opportunities, formulations & relevant evaluation approaches. Journal of Controlled Release, 352, 1–1144.
CrossRef - Koirala, S., & Nepal, P. (2021). Formulation and evaluation of mucoadhesive buccal tablets of aceclofenac. Heliyon, 7, e06439.
CrossRef - European Medicines Agency. (2006). ICH topic Q1A(R2): Stability testing of new drug substances and products (CPMP/ICH/2736/99).
- Indian Pharmacopoeia Commission. (2018). Indian Pharmacopoeia (Vol. II, pp. 1143–1144). Government of India, Ministry of Health and Family Welfare.
- Shirsand, S. B., & Suresh, S. (2008). Design and evaluation of fast dissolving tablets of clonazepam. Indian Journal of Pharmaceutical Sciences, 70(6), 791–795.
CrossRef - Seshadri, V. C., & Manohari, P. J. (2013). Formulation and characterization of mouth dissolving tablets containing benzodiazepine. Indo American Journal of Pharmaceutical Research, 3(12), 15494–15499.
- Mohanty, D., Bakshi, V., & Singh, M. (2017). Preparation and in-vitro evaluation of novel orally disintegrating tablet of clonazepam containing extracted starch as natural disintegrant. International Journal of Pharmaceutical Research and Health Sciences, 5(3), 1707–1712.
CrossRef - Castel-Branco, M. M., & Alves, G. L. (2009). The maximal electroshock seizure (MES) model in the preclinical assessment of potential new antiepileptic drugs. Methods and Findings in Experimental and Clinical Pharmacology, 31(2), 101–106.
CrossRef - Woodbury L.A and V.D Davenport. Archives International pharmacodynamic and therapie. 1952;92:97-107.
- Kumar, H. K., & Kishore, M. S. (2018). Study of anticonvulsant activity of acetazolamide on albino rats and its influence on anticonvulsant activity of sodium valproate. International Journal of Basic & Clinical Pharmacology, 7(5), 976–986.
CrossRef
Accepted on: 03 Oct 2025
Second Review by: Dr. Shreya Shanyal
Final Approval by: Dr. Charanjeet Kaur

















