Chemical Characterization and Docking Insights into Rhein Isolated from Cassia fistula Seeds as a Natural Antioxidant Candidate
Teerthankar Mahaveer College of Pharmacy, Teerthankar Mahaveer University, Moradabad, Uttar Pradesh, India
Corresponding Author E-mail: nainarathore891@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/410530
ABSTRACT:This study explores the phytochemical profiling, isolation, and antioxidant evaluation of bioactive constituents from Cassia fistula seed extracts. Seeds were extracted using ethanol through Soxhlet extraction. The ethanolic extract yielded 32.17% w/w of concentrated phytoconstituents. Preliminary phytochemical screening confirmed the presence of alkaloids, flavonoids, glycosides, carbohydrates, and anthraquinones. Column chromatography led to the isolation of a compound, characterized as Rhein (C₁₅H₈O₆; molecular weight 284.22 g/mol), using FTIR, ¹H-NMR, ¹³C-NMR, and LC-MS. The total flavonoid content (TFC) was quantified at 24.9 mg quercetin equivalents (QE)/g. Antioxidant activity was assessed using DPPH and ABTS assays, with a Trolox Equivalent Antioxidant Capacity (TEAC) of 13.23 μmol/mg & a CTE of 9.78 µg/mL. The DPPH radical scavenging activity demonstrated a Radical Scavenging Activity (RSA) of 5.39%. ABTS assay revealed an RSA of 4.088%, TEAC of 12.022 μmol/mg, and CTE of 3.345 µg/mL. Molecular docking studies using Schrodinger’s Glide XP module showed a Glide Score of −8.267 for Rhein with FXR protein (PDB ID: 30MM), indicating strong binding interactions. The findings suggest that Cassia fistula seeds, especially the Rhein compound, possess notable antioxidant potential, supporting their traditional therapeutic applications and paving the way for further development as natural antioxidants or hepatoprotective agents.
KEYWORDS:Antioxidant activity; Cassia fistula; Farnesoid X Receptor (FXR); Flavonoid content; Molecular docking
Introduction
A member of the Caesalpiniaceae family, Cassia fistula L. is extensively spread in tropical and subtropical countries, including Africa, the Americas, Southeast Asia, and India. It is sometimes called the Indian laburnum and golden shower tree.1 Traditionally, different parts of this plant, especially its pods, seeds, and leaves, have been employed in Ayurvedic and folk medicine systems for managing ailments such as skin disorders, jaundice, and inflammatory diseases.2,3 Previous phytochemical studies have reported the existence of bioactive constituents such as anthraquinones, alkaloids, flavonoids, glycosides, & terpenoids in various Cassia species, indicating their potential for pharmacological exploration.4–6
Anthraquinone derivatives have garnered significant attention for their antioxidant, anti-inflammatory, and hepatoprotective properties. Rhein is notably found in rhizomes of the Rheum genus but is rarely reported in Cassia fistula seeds.7,8 Given the global health burden of oxidative stress-related diseases, including liver disorders, the search for effective natural antioxidants remains critical.9
Despite the well-documented traditional use of Cassia fistula, there is limited scientific data available on the systematic isolation and characterization of its key phytoconstituents from the seeds of this plant. Recognizing this gap, the recent study explored the phytochemical & pharmacological potential of C. fistula seed extracts. The core aims of this investigation were to carry out phytochemical screening of the seed extract, isolate and structurally elucidate bioactive compounds using techniques such as FTIR, NMR, and LC-MS, and estimate the total flavonoid content of the extract. Additionally, using DPPH and ABTS assays, the study assessed the isolated compound’s antioxidant properties. To further support the bioactivity findings, molecular docking analysis was conducted to assess the binding interactions of the isolated compound with the Farnesoid X Receptor (FXR), a protein associated with liver health. Overall, the study aims to establish that seeds of Cassia fistula are a valuable source of naturally derived antioxidants with potential therapeutic applications.
Materials and Methods
Procurement and Authentication of Plant Specimens
Seeds of Cassia fistula were collected from the Botanical Garden, Bareilly and Uttar Pradesh, India. The seeds were purified and dried in the shade. Authentication conducted by Dr. Sunita Garg, Scientist F and Head of the Raw Material Herbarium & Museum (RHMD), NISCPR, New Delhi, India. A voucher specimen (NIScPR/RHMD/Consult/-2023/4519-20) has been deposited with the department for future reference.
Drugs and Chemicals
Chemicals used in the study were purchased from Central Drug House Ltd., New Delhi, India10
Preparation of extracts
The seeds of Cassia fistula were dried out at 37⁰C in the shade and ground in a mortar. Coarse powder (500 g) of seed was defatted with (500 mL) of petroleum ether (40-60ºc) with the aid of a Soxhlet apparatus for 24 hrs. Then extracted separately with 500 mL each of ethanol & water for 48 hours.11–13 The extracts were concentrated in vacuum using the rotary evaporator, and the yield value was found to be 32.17%, & kept in an airtight container for subsequent use.14,15
Phytochemical Analysis
Cassia fistula extract seeds were subjected to screening tests for different plant constituents by standard procedures.16 Cassia fistula extract was subjected to tests such as Alkaloids, Glycosides, Saponins, Carbohydrates, and Steroidal compounds.
Thin layer chromatography
Cassia fistula extract was found to have been spotted on a silica-coated aluminum plate to saturation. Compared to the solvent system (Hexane: ethyl acetate 90:10). On the completion of the solvent running entirely, the plate was then dried & developed with a suitable reagent and visualized under an iodine chamber.17,18 The Rf values of the individual components were determined by them.19
Calculating the total amount of flavonoids
By dissolving 1 mg of Cassia fistula preparations in 1.5 ml of CH3OH, 0.1 ml of 10% AlCl3, 0.1 ml of 1 M C2H3O2K, and 2.8 ml of distilled water, the extracts’ total flavonoid concentration (TFC) was determined. For half an hour, the mixture was allowed to remain at room temperature.20,21 An Ultrasonic UV-Vis Double Beam Spectrophotometer was employed to measure the absorbance at 415 nm. Comparable therapy was administered at various quercetin concentrations (10-50 μg/ml). The TFC was determined using quercetin as the reference, and the results were presented as mg QE/g of the sample.
Isolation and Characterization
To isolate the component from the Cassia fistula extract, column chromatography was done. After crystallization, the mother liquor was concentrated under vacuum and chromatographed over silica gel (60 g) of mesh size (16-120 mesh). The column was packed into a hydrodynamic column and eluted using a solvent system of hexane: ethyl acetate was used as the mobile phase with increasing polarity.22,23 The fractions were collected and monitored by TLC. The isolated compound was characterized by different spectroscopic techniques (that is, IR, NMR, and LC-MS).24
Dataset ligands and Ligand Optimization
To generate 3D structures of the Rhein compound, 2D structures were passed through potential algorithms and used superior force fields. Rhein’s initial geometrical optimisation and energy minimisation, carried out using the Schrodinger suite’s Ligprep tool, served as the foundation for all computations. A range of ionization states, a special program Epic, plus various possible conformers and tautomers, was generated with the Ligprep module.25
Molecular Docking Studies
Molecular docking studies were performed to assess the binding affinity and interaction profile of Rhein, an isolated compound from Cassia fistula seeds, with a relevant biological target. The target protein was chosen to be the Farnesoid X Receptor (FXR), Bile acid regulation via a nuclear receptor balance & hepatoprotective reactions. The Protein Data Bank provided the three-dimensional crystal structure of FXR (PDB ID: 3O9M). The Protein Preparation Wizard in the Schrodinger Suite was used to construct the protein structure before docking. This involved the removal of water molecules beyond 1 Å, the addition of missing hydrogen atoms, the assignment of proper bond orders, and the optimization of hydrogen bonding networks. Energy minimization was carried out using the OPLS-2005 force field to stabilize the protein structure. The chemical structure of Rhein was drawn and converted into its three-dimensional format using the LigPrep module. Various ionization and tautomeric states were generated using Epik at a physiological pH of 7.0 ± 2.0, and the ligand was optimized with energy minimization. The docking grid was generated around the active site of the FXR receptor, ensuring proper coverage of the binding pocket. The Glide XP (Extra Precision) mode of molecular docking was used to determine the optimal binding pose and energy. The docking score, also known as the GlideScore, along with additional parameters such as Emodel energy and hydrogen bonding interactions, was recorded to evaluate binding efficiency.
Antioxidant activity
DPPH assay
The DPPH test was carried out according to the protocol. In short, 30 minutes were spent measuring the absorbance at 518 nm after 0.1 mL of solution of sample and 3.9 mL of the DPPH radical solution were combined. A plot of a calibration curve using six Trolox standards was made. Effect of Scavenging of DPPH (%). By applying the subsequent formula, inhibition was determined:
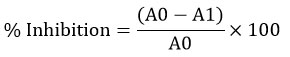
Where A0= Control absorbance, A1= Standard absorbance.
To assess the antioxidant potential of DPPH radical scavenging, an isolated phytoconstituent was utilized.26.
ABTS
The strategy is predicated on the antioxidant molecules’ capacity to counteract the ABTS radical, which is persistent. To create a stable stock solution of the ABTS radical, 2.45 mmol/L KPS (final concentration) was mixed with 7 mmol/L ABTS aqueous solution. The mixture was allowed to stand at 37 °C in the dark for between twelve and sixteen hours before use. To make an ABTS radical working solution at the beginning of the investigation, the stock solution was diluted in ethanol until it attained a result of 0.70±0.02 at 734 nm. One milligram of sample was then dissolved in one milliliter of methanol and mixed with 4.85 milliliters of diluted ABTS+ radical content. Six minutes after the initial mixing, the absorbance at 734 nm was noted. Different quantities of trolox (10-50 µmol) were prepared and then exposed to an equivalent amount of radical solution. The reading was taken six minutes after a control was made using 4.85 ml of ABTS solution and 0.15 ml of 45% ethanol. The following formula was used to determine the optical density of Trolox and the percentage of ABTS radical scavenging.:

The % scavenging curve generated from Trolox standard was used to estimate the Trolox equivalent(mM/gm) for the isolated compound27.
Results
Extraction
The preliminary analysis of the Cassia fistula ethanolic extract demonstrated significant extraction efficiency, with a percentage yield of 32.17%.
Preliminary phytochemical screening
Cassia fistula extracts were subjected to preliminary phytochemical screening with different solvents. Results of phytochemical screening Table 1 (-) negative, (+) Positive.
Table 1: Preliminary Phytoconstituents Compounds found in Cassia fistula extract
| Phytoconstituents | Test | Extract of seeds of Cassia fistula |
| Alkaloids | Mayer’s testHager’s test | -+ |
| Glycosides | Keller Killiani test | + |
| Flavonoids | Alkaline reagent testLead acetate | ++ |
| Carbohydrates | Molish test | + |
| Proteins | Biuret test | – |
| Saponin | Frothing test | – |
| Steroids | Salkowski test | – |
| Anthraquinone | Test of Carbon tetrachloride | + |
| Phenolics | Test of Ferric chloride | – |
| Tannins | Test of Ferric chloride | – |
Positive (+), Negative (-), Cassia fistula extract
Total flavonoid content
The aluminium chloride (AlCl3) technique was used to calculate the isolated compound’s total flavonoid content (TFC). Quercetin was utilised as a standard to assess the reaction mixture’s absorbance at 415 nm (Error! Reference source not found.Error! Reference source not found.). Quercetin’s calibration curve was linear between 10 and 40 µg/mL. Using the quercetin standard curve’s y=0.001x; R2 = 1, the TFC of the isolated compound was computed and represented as mg of quercetin equivalent QE/gram of the substance. The TFC of the isolated compound was found to be 24.9 mg QE/gram of the compound. This result indicates the presence of a significant quantity of flavonoids, suggesting their potential biological and pharmacological relevance.
Column Chromatography
Approximately five fractions were obtained by TLC analysis after the Cassia fistula extract was eluted using various hexane: ethyl acetate ratios after being exposed to column chromatography on silica gel (Table 2). There were several fractions with no spots at all. But some had blurry areas. We merged fractions with the same Rf value that displayed clear spots (4,5). Utilising FTIR, NMR, and LC-MS spectroscopy, the isolated substance was characterised.
Table 2: Fractions obtained from Cassia fistula extract using column chromatography
| Solvent System | Ratio | Fractions no. | TLC | Code OF Fraction |
| H: E | 100:0 | 1 | Not Clear | EE- A |
| H: E | 90:10 | 2 | No spot | EE-B |
| H: E | 80:20 | 3 | Not Clear | EE-C |
| H: E | 70:30 | 4, 5 | Clear | EE-D |
| H: E | 60:40 | 6 | Not Clear | EE-E |
Hexane: Ethyl acetate
Characterization of isolated compound
The TLC of Cassia fistula extract (mother liquor) was performed using Silica Gel plates and a solvent system consisting of C6H14 and CH3COOCH2CH3 in the ratio of 90:10. The analysis was done according to standard procedures. The Rf value was calculated. In this analysis, one distinct spot was observed. The spot appeared dark green and had an Rf value of 0.53.
In Figure 1, distinct FTIR peaks of the isolated chemical CFRP were displayed. Vmax IR (KBr): 3339 cm-1 (O-H stretching); 1634 cm-1 (C=O stretching); 1508 cm-1 (C=O stretching), 1336 cm-1 (C-O stretching); 1018 cm-1 (Ar, C-H stretching).
At 500 MHz, DMSO, 1H NMR δ 11.88 (1H s, OH), 8.10 (2H d, J = 4.1 Hz, OH), 7.82 (1H s, CH), 7.74 (2H d, J = 4.4 Hz, CH), and 7.40 (2H d, J = 3.8 Hz (Figure 2).
CH.13C NMR (126 MHz, DMSO) δ 191.20 (s), 180.86 (s), 165.31 (s), 161.33 (s), 160.99 (s), 138.00 (s), 137.51 (s), 133.69 (s), 133.09 (s), 124.51 (s), 124.04 (s), 119.34 (s), 118.63 (d, J = 13.1 Hz), 116.05 (s) (Figure 3).
Based on ES-MS results at m/z 283.03 [M+], the mass spectroscopy of the isolated chemical CFE revealed the molecular formula C15H8O6, and the molecular weight was determined to be 284.22 (Figure 4). By comparing the reported data with the physical characteristics, IR, mass, 1H-NMR, and 13C-NMR data, compound was determined to be Rhein (Figure 5).
 |
Figure 1: Fourier Transform Infrared Spectroscopy. |
 |
Figure 2: 1H NMR spectroscopy of isolated compounds. |
 |
Figure 3: 13C NMR spectroscopy of isolated compounds. |
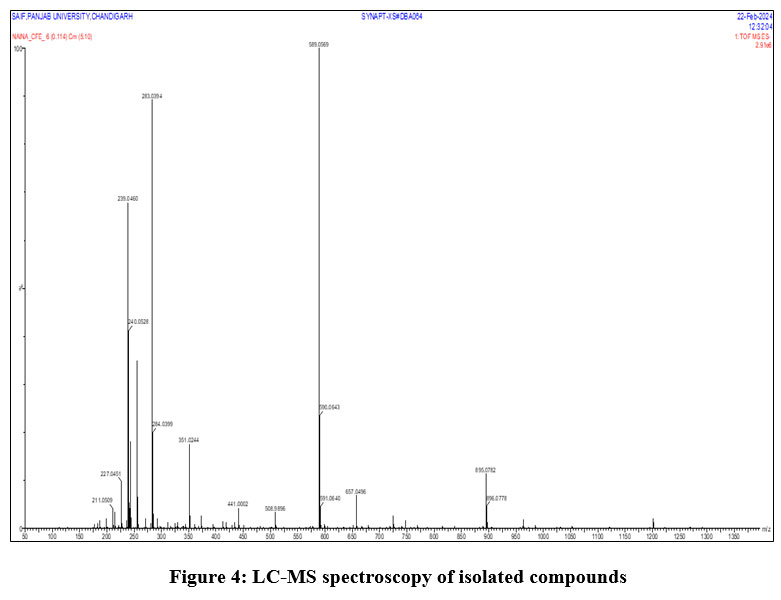 |
Figure 4: LC-MS spectroscopy of isolated compounds. |
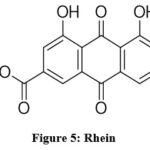 |
Figure 5: Rhein. |
Molecular docking
The molecular docking analysis of Rhein, a key phytoconstituent isolated from Cassia fistula seed extract, was conducted to evaluate its interaction with the Farnesoid X Receptor (FXR), a nuclear receptor linked to the control of bile acid metabolism and liver function. The Schrodinger Suite’s Glide XP module was used for the docking, with the optimized structure of FXR (PDB ID: 3O9M) as the target. The docking results revealed that Rhein binds efficiently within the active site of FXR, demonstrating a Glide XP score of −8.267, indicative of a strong and favorable interaction (Figure 6).
Although the docking did not show specific hydrogen bond formation, the binding was primarily stabilized by van der Waals forces and hydrophobic interactions. These interactions suggest that Rhein fits well into the lipophilic pocket of FXR, maintaining structural complementarity. The Emodel energy and Glide energy, which were recorded as −54.051 and −32.997 kcal/mol, respectively, further support the stability of the ligand-receptor complex.
These findings are significant as they imply that Rhein may act as a potential modulator of FXR activity, which is often targeted in drug development for treating liver diseases, including cholestasis and non-alcoholic fatty liver disease. The docking data also complements the in vitro antioxidant findings, reinforcing the therapeutic relevance of Rhein as a multifunctional bioactive compound derived from Cassia fistula.
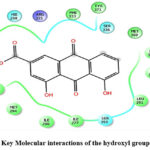 |
Figure 6: Key Molecular interactions of the hydroxyl group of Rhein. |
Evaluation Parameters (In Vitro)
DPPH free radical scavenging assay
The antioxidant capacity of the isolated molecule was evaluated using the DPPH free radical scavenging experiment (Y 0.3668x- 8.7137) (Figure 7). The absorbance value of 0.228 was used to calculate the compound’s RSA and TEAC. With an RSA of 5.39%, it was found to have a moderate capacity to scavenge free radicals. Furthermore, the compound’s antioxidant ability in comparison to the standard antioxidant Trolox was demonstrated by the calculated concentration of Trolox Equivalent (CTE) at 9.78 μg/mL and the TEAC value at 13.23 μmol/mg (Table 3Error! Reference source not found.).
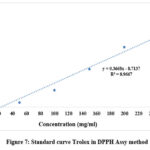 |
Figure 7: Standard curve Trolox in DPPH Assy method. |
Table 3: Calculated the RSA and TEAC of a sample using recorded absorbance and the trendline equation of the standard
| Type | Sample | Absorbance | RSA (%) | CTE (μg/ml) | TEAC (μmol/mg) |
| Sample | CERP | 0.228 | 5.39 | 38.46 | 9.78 |
Trolox equivalent antioxidant capacity (TEAC) and radical scavenging activity (RSA).
ABTS-Trolox equivalent antioxidant capacity (TEAC)
The antioxidant potential of the isolated chemical was assessed using the ABTS, which free radical scavenging test. The scavenging activity was calculated using the trendline equation obtained from the standard (Trolox) and the absorbance values observed at 734 nm (Figure 8). The findings are reported in Table 4, where the isolated compound’s Concentration of Trolox Equivalent (CTE), TEAC, and RSA were measured 28 with an absorbance of 0.657, the isolated chemical showed an RSA of 4.088 percent. In comparison to the Trolox standard, the compound’s antioxidant capacity was demonstrated by the computed CTE of 3.345 μg/mL and the TEAC value of 12.022 μmol/mg.
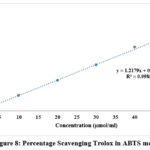 |
Figure 8: Percentage Scavenging Trolox in ABTS method. |
Table 4: Calculated RSA and TEAC of samples using recorded absorbances and trendline equation of Trolox
| S.No. | Type | Sample Code | Absorbance | RSA (%) | CTE (μg/ml) | TEAC (μmol/mg) |
| 1 | Sample | CERP | 0.657 | 4.088 | 3.345 | 12.022 |
Discussion
The current investigation sought to investigate the molecular interactions, phytochemical makeup, and antioxidant capacity of bioactive substances extracted from Cassia fistula seeds. The ethanolic extraction yielded a significant amount (32.17%), suggesting a good solubility of polar and semi-polar phytochemicals in the ethanol solvent. Preliminary phytochemical screening confirmed the presence of flavonoids, glycosides, anthraquinones, and carbohydrates, which are often associated with various pharmacological properties such as antioxidant and anti-inflammatory activities.14,16
Among the isolated compounds, Rhein was identified as the major bioactive constituent through spectroscopic methods including FTIR, ¹H-NMR, ¹³C-NMR, and LC-MS. The structure elucidation showed characteristic useful groups such as hydroxyl (–OH) and carbonyl (C=O) groups, which are well-known contributors to RSA40. The TFC was determined to be 24.9 mg quercetin equivalents per gram, indicating a rich presence of flavonoid-type antioxidants. This value is comparable to previous studies that reported significant TFC in various parts of Cassia fistula and associated it with free radical neutralization.14,16
Antioxidant activity, assessed by DPPH and ABTS assays, demonstrated that the isolated compound exhibited moderate radical scavenging potential with TEAC values of 13.23 µmol/mg and 12.02 µmol/mg, respectively. These results align with earlier findings where C. fistula extracts showed dose-dependent antioxidant properties, attributable to their phenolic and flavonoid constituents15,29. The antioxidant mechanism may involve hydrogen atom donation or electron transfer, neutralizing free radicals and thereby reducing oxidative stress.
Furthermore, the molecular docking analysis of Rhein with the Farnesoid X Receptor (FXR) revealed a Glide XP score of −8.267 and favorable Emodel (−54.051) and Glide energies (−32.997 kcal/mol). These results suggest a high binding affinity of Rhein toward the FXR binding pocket, supporting the hypothesis that Rhein could modulate FXR-mediated pathways involved in bile acid homeostasis and liver detoxification.27,30 Although no direct hydrogen bonding was observed, the presence of strong hydrophobic and van der Waals interactions implies good structural compatibility within the active site of FXR.
FXR is a well-established target in managing liver-related disorders, particularly non-alcoholic steatohepatitis (NASH), cholestasis, and metabolic syndrome. Compounds that enhance FXR activation are reported to exert hepatoprotective effects by reducing inflammation and lipid accumulation in hepatic tissues30. The interaction of Rhein with FXR, supported by computational docking, not only reinforces its potential as a hepatoprotective agent but also adds a novel dimension to its pharmacological profile.
Taken together, the findings of this study support the traditional use of Cassia fistula in treating hepatic and inflammatory disorders. The integration of phytochemical screening, bioactivity assays, and molecular docking provides a comprehensive approach for validating the therapeutic potential of natural compounds. While the in vitro antioxidant and in silico docking results are promising, further in vivo pharmacological and toxicological studies are required to establish Rhein’s efficacy and safety as a drug candidate.
Conclusion
The present investigation highlights the successful extraction and identification of Rhein, a key bioactive compound, from the seeds of Cassia fistula. Comprehensive spectroscopic characterization affirmed its chemical identity. The compound demonstrated antioxidant efficacy through DPPH and ABTS assays, supported by a substantial flavonoid content. Moreover, molecular docking analysis revealed strong binding affinity with the Farnesoid X Receptor (FXR), suggesting Rhein’s potential role in maintaining hepatic function and managing oxidative stress. These findings provide scientific support for the traditional medicinal use of Cassia fistula and pave the way for future research into its application as a natural hepatoprotective and antioxidant agent. Further in vivo and clinical investigations are encouraged to confirm these results and explore therapeutic applications.
Acknowledgement
This research was a part of Ph.D. Program of Teerthanker University, Moradabad. Also, authors thanks to Prof Anurag Verma, Prof Mayur Porwal and Prof Piyush Mittal for their help and support for the publication of the manuscript.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Authors Contributions
Naina: Original draft, Methodology, Investigation, Software.
Phool Chandra: Conceptualization, Investigation, Formal analysis, Visualization, Software, Writing – review & editing, Supervision
References
- Mueller, S.; research, L. S.-H. medicine: evidence and; 2010, undefined. Liver Stiffness: A Novel Parameter for the Diagnosis of Liver Disease. Taylor & Francis 2010, 2, 49–67. https://doi.org/10.2147/hmer.s7394.
CrossRef - Asrani, S. K.; Devarbhavi, H.; Eaton, J.; Kamath, P. S. Burden of Liver Diseases in the World. J. Hepatol. 2019, 70 (1), 151–171. https://doi.org/10.1016/j.jhep.2018.09.014.
CrossRef - Arman, M.; Chowdhury, K. A. A.; Bari, M. S.; Khan, M. F.; Huq, M. M. A.; Haque, M. A.; Capasso, R. Hepatoprotective Potential of Selected Medicinally Important Herbs: Evidence from Ethnomedicinal, Toxicological and Pharmacological Evaluations. Phytochem. Rev. 2022, 21 (6), 1863–1886. https://doi.org/10.1007/S11101-022-09812-5.
CrossRef - Lee, W.; Stravitz, R.; Hepatology, A. L.-; 2012, undefined. Introduction to the Revised American Association for the Study of Liver Diseases Position Paper on Acute Liver Failure 2011. Wiley Online Library 2011, 55 (3), 965–967. https://doi.org/10.1002/hep.25551.
CrossRef - Saidurrahman, M.; Mujahid, M.; Siddiqui, M. A.; Alsuwayt, B.; Rahman, M. A. Evaluation of Hepatoprotective Activity of Ethanolic Extract of Pterocarpus Marsupium Roxb. Leaves against Paracetamol-Induced Liver Damage via Reduction of Oxidative Stress. Phytomed. Plus 2022, 2 (3). https://doi.org/10.1016/j.phyplu.2022.100311.
CrossRef - Mwangi, R. W.; Macharia, J. M.; Wagara, I. N.; Bence, R. L. The Medicinal Properties of Cassia Fistula L: A Review. Biomed. Pharmacother. 2021, 144. https://doi.org/10.1016/j.biopha.2021.112240.
CrossRef - Vineeta; Shukla, G.; Bhat, J. A.; Chakravarty, S. Species Richness and Folk Therapeutic Uses of Ethnomedicinal Plants in West Bengal, India – A Meta-Analysis. Phytomed. Plus 2022, 2 (1). https://doi.org/10.1016/j.phyplu.2021.100158.
CrossRef - Tariq, M.; Ahmad, N.; Nisa, M. U.; Rahim, M. A.; Zongo, E. Phytochemicals Profiling of Cassia Fistula Fruit Extract and Its Effect on Serum Lipids and Hematological Parameters in High-Fat Diet-Induced Hyperlipidemic Female Rats. Food Sci. Nutr. 2024, 12 (8), 5776–5784. https://doi.org/10.1002/FSN3.4229.
CrossRef - Wasu, S.; Muley, B. P. Hepatoprotective Effect of Cassia fistula Linn. Ethnobot. Leaflets 2009, 13, 910–926.
- Chaerunisaa, A. Y.; Susilawati, Y.; Muhaimin, M.; Milanda, T.; Hendriani, R.; Subarnas, A. Antibacterial Activity and Subchronic Toxicity of Cassia Fistula L. Barks in Rats. Toxicol. Rep. 2020, 7, 649–657. https://doi.org/10.1016/j.toxrep.2020.04.013.
CrossRef - Irshad, Md.; Zafaryab, Md.; Singh, M.; Rizvi, M. M. A. Comparative Analysis of the Antioxidant Activity of Cassia Fistula Extracts . Int. J. Med. Chem. 2012, 2012, 1–6. https://doi.org/10.1155/2012/157125.
CrossRef - Killedar, S.; more, H.; Nadaf, S.; Nale, A.; Pawar, A.; Tamboli, U. Isolation, characterization, and evaluation of Cassia fistula Linn. seed and pulp polymer for pharmaceutical application. Int. J. Pharm. Invest. 2014, 4(4), 215-225. https://doi.org/10.4103/2230-973X.143128.
CrossRef - Bhalodia, N. R.; Acharya, R. N.; Shukla, V. J. Evaluation of in Vitro Antioxidant Activity of Hydroalcoholic Seed Extratcs of Cassia Fistula Linn. Free Rad. Antioxid. 2011, 1 (1), 68–76. https://doi.org/10.5530/AX.2011.1.11.
CrossRef - Tabrez, S.; Rahman, F.; Ali, R.; Alouffi, A. S.; Alshehri, B. M.; Alshammari, F. A.; Alaidarous, M. A.; Banawas, S.; Bin Dukhyil, A. A.; Rub, A. Assessment of the Antileishmanial Potential of Cassia Fistula Leaf Extract. ACS Omega 2021, 6 (3), 2318–2327. https://doi.org/10.1021/ACSOMEGA.0C05629.
CrossRef - Abdellatif, N. A.; Eltamany, E. E.; El-Shenawy, N. S.; Nafie, M. S.; Hassan, Y. M.; Al-Eisa, R. A.; Badr, J. M.; Abdelhameed, R. F. A. Cassia Fistula Leaves Extract Profiling and Its Emphasis on Induced Ulcerative Colitis in Male Rats through Inhibition of Caspase 3 and Cyclooxygenase-2. Arab. J. Chem. 2024, 17 (4). https://doi.org/10.1016/j.arabjc.2024.105672.
CrossRef - Abdullah, R.; Younas, Q.; Kaleem, A.; Iqtedar, M.; Aftab, M.; Saleem, F. Phytochemical and Antimicrobial Properties of Different Plants and in Silico Investigation of Their Bioactive Compounds in Wound Healing and Rheumatism. Saudi J. Biol. Sci. 2024, 31 (3). https://doi.org/10.1016/j.sjbs.2024.103942.
CrossRef - Sumi, S.: Oommen P. S. Antibacterial, anthelmentic and phytochemical investigations on the pod extracts of Cassia fistula Linn. Int. J. Med. Phar. Sci. 2012 2(1), 6-14.
- Arulpandi, I.; Sangeetha, R. Antibacterial Activity of Fistulin: A Protease Inhibitor Purified from the Leaves of Cassia Fistula. Int. Sch. Res. Notices. 2012, 2012 (1), 584073. https://doi.org/10.5402/2012/584073.
CrossRef - Silver, J. Let Us Teach Proper Thin Layer Chromatography Technique! J. Chem. Educ. 2020, 97 (12), 4217–4219. https://doi.org/10.1021/ACS.JCHEMED.0C00437.
CrossRef - Verma, S. C.; Vashishth, E.; Subhani, S.; Singh, R.; Pant, P.; Padhi, M. M.; Kumar, A. Phytochemical Study of Stem Bark versus Small Branches of Cassia Fistula Linn. Using High Performance Thin Layer Chromatographic Ultra Violet Detection Method …. wjpr.s3.ap-south-1.amazonaws.com 2015, 4 (3), 1910–1920.
- Irshad, M.; Shreaz, S.; Manzoor, N.; … L. K.-P.; 2011, undefined. Anticandidal Activity of Cassia Fistula and Its Effect on Ergosterol Biosynthesis. Taylor & Francis 2011, 49 (7), 727–733. https://doi.org/10.3109/13880209.2010.544318.
CrossRef - Pathak, R.; Sachan, N.; Kabra, A.; Alanazi, A. S.; Alanazi, M. M.; Alsaif, N. A.; Chandra, P. Isolation, Characterization, Development and Evaluation of Phytoconstituent Based Formulation for Diabetic Neuropathy. Saudi Pharm. J. 2023, 31 (8), 1-19. https://doi.org/10.1016/J.JSPS.2023.06.020.
CrossRef - Sachan, N.; Saraswat, N.; Chandra, P.; Khalid, M.; Kabra, A. Isolation of Thymol from Trachyspermum Ammi Fruits for Treatment of Diabetes and Diabetic Neuropathy in STZ-Induced Rats. Biomed Res Int 2022, 2022, 1-20. https://doi.org/10.1155/2022/8263999.
CrossRef - Taher, M. A.; Laboni, A. A.; Islam, M. A.; Hasnat, H.; Hasan, M. M.; Ferdous, J.; Shompa, S. A.; Khan, M. Isolation, Characterization and Pharmacological Potentials of Methanol Extract of Cassia Fistula Leaves: Evidenced from Mice Model along with Molecular Docking Analysis. Heliyon 2024, 10 (7). https://doi.org/10.1016/j.heliyon.2024.e28460.
CrossRef - Roggia, M.; Natale, B.; Amendola, G.; Di Maro, S.; Cosconati, S. Streamlining Large Chemical Library Docking with Artificial Intelligence: The PyRMD2Dock Approach. J. Chem. Inf. Model 2024, 64 (7), 2143–2149. https://doi.org/10.1021/ACS.JCIM.3C00647.
CrossRef - Rumpf, J.; Burger, R.; Schulze, M. Statistical Evaluation of DPPH, ABTS, FRAP, and Folin-Ciocalteu Assays to Assess the Antioxidant Capacity of Lignins. Int. J. Biol. Macromol. 2023, 233. https://doi.org/10.1016/j.ijbiomac.2023.123470.
CrossRef - George, J.; Edwards, D.; Pun, S.; Williams, D. Evaluation of Antioxidant Capacity (ABTS and CUPRAC) and Total Phenolic Content (Folin-Ciocalteu) Assays of Selected Fruit, Vegetables, and Spices. Int. J. Food. Sci. 2022, 2022. https://doi.org/10.1155/2022/2581470.
CrossRef - Deyalage, S. T.; Wickramasinghe, I.; Amarasinghe, N.; Thilakarathna, G. Influence of Cooking Methods on Antioxidant Activities of Selected Leafy Vegetables Gymnema Lactiferum, Wattakaka Volubilis, and Argyreia Populifolia in Sri Lanka. Int. J. Food Sci. 2021, 2021. https://doi.org/10.1155/2021/6660308.
CrossRef - Siddhuraju, P.; Mohan, P. S.; Becker, K. Studies on the Antioxidant Activity of Indian Laburnum (Cassia Fistula L.): A Preliminary Assessment of Crude Extracts from Stem Bark, Leaves, Flowers and Fruit Pulp. Food Chem. 2002, 79 (1), 61–67. https://doi.org/10.1016/S0308-8146(02)00179-6.
CrossRef - Xia, Q.; Lei, Y.; Wang, J.; Wang, Q. Probiotic Management and Inflammatory Factors as a Novel Treatment in Cirrhosis: A Systematic Review and Meta-Analysis. Open Life Sci. 2023, 18 (1). https://doi.org/10.1515/BIOL-2022-0741/HTML.
CrossRef
Accepted on: 18 Oct 2025
Second Review by: Dr. Astha Jaiswal
Final Approval by: Dr. Pounraj Thanasekaran

















