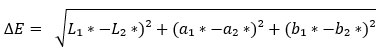Characterization of Color and Application of Natural Dyes from Hagonoy (Chromolaena odorata) Leaves on Cotton and Wool Fabrics
College of Pharmacy and Chemistry, University of the Immaculate Conception, Davao City, Philippines.
Corresponding Author Email:girliemaepastor@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/410513
ABSTRACT:This study examines the characterization of color and application of natural dyes from Hagonoy (Chromolaena odorata) leaves on cotton and wool fabrics. A descriptive-experimental research design was used, employing the maceration method for dye extraction. The fabrics underwent unmordanted, premordanted, metamordanted, and postmordanted processes using potassium aluminum sulfate and ferrous sulfate as mordants. The dyed fabrics were tested for stability under washing, sunlight, and perspiration (acidic and basic) conditions. A colorimeter measured the Lab values, and color differences (ΔE) were analyzed for comparison. Cotton fabrics (ΔE=1.75) showed better dye uptake and stability than wool (ΔE=10.84). Among mordants, ferrous sulfate (ΔE=13.78) produced darker but less stable colors, whereas potassium aluminum sulfate (ΔE= 2.57) yielded brighter and more stable results, especially at lower ratios. Metamordanting on cotton emerged as the most effective process for enhancing dye performance. The study underscores the potential of Chromolaena odorata leaves as a sustainable and eco-friendly dye source for the textile industry, offering an alternative to synthetic dyes that aligns with global environmental objectives.
KEYWORDS:Cotton; Hagonoy Leaves; Mordanting Process; Natural Dyes; Textile Chemistry; Wool
Introduction
Natural dyes are colorants derived from plants and are known for their use in coloring natural fibers1. Nowadays, there is increasing awareness among people towards the use of eco-friendly natural dyes owing to high biodegradability and environmental compatibility. They are non-toxic, non-allergic to skin, non-carcinogenic, abundant, and renewable2. However, synthetic dye dominates the fabric market; 10,000 synthetic dyes are made annually for fabric coloration3. The synthetic dyes are difficult to break down and may damage surrounding surface water and may result in significant environmental wastewater pollution4.
The global fabric industry is a major contributor to environmental wastewater pollution, primarily due to the extensive use of synthetic chemicals in textile production. Synthetic dyes, when discharged into surface waters, leach into groundwater systems and cause severe contamination5. In contrast, natural dyes are environmentally benign, maintaining ecological balance without generating harmful effluents6. Studies have shown that wastewater from natural dyes exhibits a BOD of only 40–85 mg/L, below the permissible limit of 100 mg/L, and significantly lower TSS and TDS levels compared to synthetic dyes7.
Synthetic dyes are also linked to allergic and toxic effects, prompting stricter environmental regulations worldwide and increasing the adoption of natural dyes8. In regions such as South Africa, industrial runoff containing synthetic chemicals has led to ecosystem contamination and fish toxicity. Similarly, the rapid growth of the fabric industry in Manila, Philippines9, has intensified wastewater pollution, with about 33% of industrial effluents10 containing synthetic dye residues, causing discoloration in water bodies11.
Chromolaena odorata (Hagonoy), an invasive species from the Asteraceae family, is prevalent in the Philippines and known for its adverse effects on agriculture and livestock12. Despite its invasive nature13, its potential as a source of natural dye offers economic and environmental benefits14. However, research on C. odorata remains limited, particularly regarding its leaves, as previous studies focused mainly on its stems and flowers. This study aims to explore the dyeing potential of C. odorata leaves to promote sustainable and eco-friendly textile dyeing practices.
Materials and Methods
The methods used in this study are described in recent studies 14,15,16,17,18,19,20,21,22 with minor modifications. The evaluation of dyed fabrics follows the standards set by the International Organization for Standardization (ISO).
Plant collection and identification
The Chromolaena odorata leaves were collected at Brgy. Kaligutan, Laak, Davao de Oro, Philippines. They were cut from the plant stem, including both newly grown and mature leaves. A qualified botanist authenticated the collected plant material.
Plant extraction
The collected Chromolaena odorata leaves were dried and grounded into small pieces using a blender. The plant extraction procedure employed the maceration method18. The maceration process involved the optimization of a mass of Chromolaena odorata leaves with a volume of distilled water in a ratio of 1:10 and 1:15 in a 1L Erlenmeyer flask at room temperature14.
Mordanting Process
The different fabrics (cotton and wool) underwent premordanting, metamordanting, and postmordanting processes individually, using ferrous sulfate and potassium aluminum sulfate as mordants. These processes were carried out to evaluate the effect of different mordanting methods on each fabric type16. Additionally, a fabric sample that was not subjected to any mordanting process was directly dyed without prior treatment.
Premordanting
Before the dyeing process, the fabric was first immersed in 200 ml of distilled water. A mordant was then added at a ratio of 1 tablespoon plus 1 teaspoon per 100 grams of fabric. The mixture was heated at 80°C for 30 minutes. The temperature of the mordant solution was monitored using a thermometer throughout the heating process. Once completed, the fabric was left to dry in the open air for several minutes before moving on to the dyeing process.
Metamordanting
During the dyeing process, a mordant was added at a ratio of 1 tablespoon plus 1 teaspoon per 100 grams of fabric into a 250 ml beaker containing 200 ml of filtered dye solution.
Postmordanting
After the dyeing process, the fabric was immersed in 200 ml of distilled water. A mordant was added at a ratio of 1 tablespoon plus 1 teaspoon per 100 grams of fabric.
The mixtures in mordanting and postmordanting were heated at 80°C for 30 minutes. The temperature of the mixture was carefully monitored using a thermometer during the heating process. After the process was completed, the dyed fabric was allowed to dry in open air for up to 24 hours before proceeding to the colorfastness test.
Dyeing Process
The plant extract, around 200 ml, was transferred into a 250 ml beaker. Different beakers were utilized for varying ratios of plant extract and mordants (ferrous sulfate and potassium aluminum sulfate) applied to each type of fabric material. The fabrics undergoing different mordanting processes were treated with different ratios of plant extract. Each fabric measured 40 ±2 mm by 100 ±2 mm19. The cotton and wool fabrics, each treated with varying concentrations of plant extract, were heated simultaneously on a hotplate for 60 minutes at 80°C. The dye solution was maintained at pH 7. The temperature of the dye solution during heating was monitored using a thermometer. Once the dyeing process was complete, the dyed fabrics were allowed to dry in the open air for 24 hours. The resulting color of the fabrics was evaluated using a colorimeter.
Colorfastness Test
Washing Test
The sample was assessed for colorfastness to washing according to ISO 105 C06 A1S: 2010. The dyed fabrics was washed using a solution composed of 4 grams per liter of European Colorfastness Establishment (ECE) reference phosphate detergent (B) and 1 gram per liter of sodium perborate19. The fabric was immersed in a 1 L beaker and stirred continuously at a speed ranging from 40 to 60 rotation per minute (RPM) on a hotplate set at 60°C for the main washing phase lasting 15 minutes. Subsequently, the fabric underwent a rinsing phase at 40°C for 15 minutes on the same hotplate. The dyed fabric was allowed to dry in the open air after the washing process20. The dyed fabric was monitored for color changes. The resulting color of the fabric was evaluated using the colorimeter.
Sunlight Test
The sample was assessed for colorfastness to sunlight according to ISO 105-B01: 2014. The dyed samples were exposed to sunlight for 6 hours a day for 5 consecutive days19. The fabric samples were suspended on a line outdoors. Throughout the exposure, the average temperature of the sunlight ranged between 40°C and 45°C, with a variation of ±5.0°C17. The samples were monitored each day for color changes. The resulting color of the fabric was evaluated using a colorimeter.
Perspiration Test in Acidic and Basic solutions
The sample was assessed for colorfastness to perspiration according to ISO 105-E04: 2013. The acidic sweat was prepared using the following formula: 0.5 g of L-Histidine hydrochloride monohydrate, 5.0 g of Sodium chloride, 2.2 g of Sodium dihydrogen phosphate dihydrate, and distilled water (1,000 mL). The pH was adjusted to 5.5 using 0.1 M Sodium hydroxide. The alkaline sweat was prepared using the following formula: 0.5 g of L-Histidine hydrochloride monohydrate, 5.0 g of Sodium chloride, 2.5 g of Di-sodium hydrogen phosphate dihydrate, and distilled water (1,000 mL). The pH was adjusted to 8.0 using 0.1 g of Sodium hydroxide. The test specimen was completely wetted in the acidic solution at pH 5.5 ±0.2 and alkaline solution at pH 8 ±0.2 in a solution ratio of 50:1, and the sample was kept at room temperature for 30 minutes. To remove any excess perspiration fluid from the fabric, it was placed between two perforated acrylic plates (30 × 30 × 5 cm) and subjected to 5 kg of pressure for 30 minutes22. The fabric was then heated in an oven for 4 hours at 37 ±2°C14. The resulting color of the fabric was evaluated using a colorimeter.
Measurement of Color Difference using Colorimeter
The dyed sample that has undergone a colorfastness test is measured for its color difference (ΔE) in comparison with the original sample. The color difference (ΔE) of the dyed sample is calculated using the following equation:
where L1∗, a1∗, b1∗ are the L*a*b* values of the reference color (first or initial sample). On the other hand, L2∗, a2∗, b2∗ are the L*a*b* values of the test color (second or final sample). L*, a*, and b* are the three components of the CIE 1976 (L*, a*, b*) color space, which is a color model developed by the International Commission on Illumination (CIE) to describe color in a perceptually uniform way. The L* represents the lightness of the color, ranging from 0 to 100, where 0 is black and 100 is white. The a* represents the position of the color along the red-green axis. Positive values of a* indicate red hues. Negative values of a* indicate green hues. The b* represents the position of the color along the blue-yellow axis. Positive values of b* indicate yellow hues. Negative values of b* indicate blue hues. From the obtained ΔE values, the classification of color change and the criteria is evaluated23. Mean was used to calculate an estimate of the central tendency of the data set. It represents the average L*a*b* values of the analyzed samples to estimate the color.
Results
In order to gather a comprehensive data on applying natural dyes from Chromolaena odorata to cotton and wool fabric, the schematic diagram as shown in Figure 1 summarizes the processes involved. This includes an analysis of the dyeing and mordanting method, color fastness test, and the overall effectiveness and stability of Chromolaena odorata as a natural dye source for fabrics. The process started with the collection of Chromolaena odorata leaves, drying of leaves, extraction then filtration. The sample preparation shown below are actual documentation of the study conducted. After the natural dye was prepared, mordanting process (premordanting, metamordanting, postmordanting) on cotton and wool fabrics followed then color measurement on cotton and wool fabrics. The stability of natural dyes on fabrics were tested by conducting washing test, sunlight test and perspiration test.
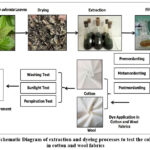 |
Figure 1: Schematic Diagram of extraction and dyeing processes to test the color change in cotton and wool fabrics. |
Color of Natural Dyes
 |
Table 1: Color of Natural Dyes on Cotton and Wool Fabrics |
Table 2: Colorimeter Values
| Mordanting Process | Mordant | Ratio | Cotton | Wool | ||||
| L* | a* | b* | L* | a* | b* | |||
| Unomordanted | N/A | 1:10 | 69.37 | 4.80 | 13.25 | 69.63 | 6.39 | 12.98 |
| 1:15 | 76.13 | 5.44 | 10.69 | 74.85 | 5.46 | 12.23 | ||
| Premordanting | Ferrous Sulfate | 1:10 | 44.69 | 4.95 | 11.35 | 49.75 | 2.37 | 7.97 |
| 1:15 | 45.26 | 4.91 | 12.30 | 56.28 | 4.15 | 7.65 | ||
| Potassium Aluminum Sulfate | 1:10 | 67.23 | 7.80 | 13.89 | 63.47 | 8.17 | 20.97 | |
| 1:15 | 72.31 | 7.40 | 14.94 | 71.50 | 5.59 | 14.41 | ||
| Metamordanting | Ferrous Sulfate | 1:10 | 42.15 | 4.36 | 11.27 | 42.79 | 3.51 | 11.23 |
| 1:15 | 55.65 | 3.52 | 11.47 | 60.34 | 3.16 | 9.14 | ||
| Potassium Aluminum Sulfate | 1:10 | 79.56 | 4.67 | 12.91 | 64.07 | 8.92 | 23.23 | |
| 1:15 | 82.49 | 2.94 | 13.65 | 74.74 | 5.40 | 14.38 | ||
| Postmordanting | Ferrous Sulfate | 1:10 | 64.81 | 4.61 | 9.54 | 58.85 | 2.80 | 10.88 |
| 1:15 | 72.35 | 4.91 | 11.44 | 61.13 | 2.39 | 9.55 | ||
| Potassium Aluminum Sulfate | 1:10 | 74.24 | 3.91 | 9.84 | 76.97 | 6.40 | 13.22 | |
| 1:15 | 79.47 | 3.11 | 7.57 | 72.60 | 4.25 | 11.67 | ||
*N/A – Not Applicable
Tables 1 and 2 present the color characteristics of natural dyes applied to cotton and wool fabrics at dye ratios of 1:10 and 1:15. The 1:10 ratio produced darker and more intense brown shades, as indicated by lower L* values, while the 1:15 ratio yielded lighter, less saturated beige tones with higher L* values. Positive a* and b* values across samples reflected red and yellow hues typical of brown shades. Wool fabrics, dyed without mordant, exhibited lighter brown to beige tones with slightly higher L* values than cotton, consistent with the natural lightness of unmordanted fibers. Overall, a higher dye concentration enhanced color depth and saturation, particularly in cotton samples.
Washing Test
For washing test as shown in Table 3, the resulting color for unmordanted cotton and wool samples exhibits notable color changes based on the dye ratio. For the 1:10 ratio, the ΔE value is 10.24 (distinct change) and 22.23 (different color) respectively, while the 1:15 ratio sample shows a smaller ΔE of 3.21, indicating a “medium” change and 15.49 (different color). This trend suggests that a higher dye concentration (1:10) results in more noticeable color fading or alteration during washing, while a lower concentration (1:15) offers slight improvement. This result aligns with the findings of a study that reported poor washing fastness in unmordanted fabrics dyed with Smith leaves24.
 |
Table 3: Stability of Characterized Colors Postexposure to Washing. |
Sunlight Test
 |
Table 4: Stability of Characterized Colors in Post-exposure to Sunlight |
For sunlight test, Table 4 showed that unmordanted cotton and wool samples reveal moderate color changes. The 1:10 ratio has an ΔE of 4.35, labeled a “medium” change, while the 1:15 ratio shows a minor change with an ΔE of 1.54, categorized as “very light.” This result indicates that lower dye concentrations may provide better lightfastness in unmordanted cotton, possibly because lighter shades fade less visibly than darker ones under sunlight.
The unmordanted wool samples undergo moderate color changes, with ΔE values of 3.49 for the 1:10 dye ratio and 4.01 for the 1:15 dye ratio, both categorized as “medium” changes. These results suggest that unmordanted wool is moderately resistant to fading in sunlight, with similar degrees of color change across both dye concentrations. The higher ΔE value in the 1:15 sample indicates that lighter dye applications may still be somewhat susceptible to light-induced fading.
Potassium aluminum sulfate postmordanted cotton showed even better results, with the 1:10 ratio sample displaying a “medium” change (ΔE = 4.97) and the 1:15 ratio slightly higher at ΔE = 5.81, also “medium.” Though not entirely fade-resistant, potassium aluminum sulfate provided relatively better lightfastness than other mordants.
Acidic Perspiration
 |
Table 5: Stability of Characterized Colors in Simulated Acidic Perspiration |
Table 5 presents the acidic perspiration test varied color stability based on mordant type and dye ratio. Unmordanted samples exhibit moderate shifts, with ΔE values of 5.92 (1:10) and 2.63 (1:15), labeled as “medium” and “light” changes, respectively. This result indicates that acidic sweat can cause minor fading or alteration in unmordanted cotton, with lower dye concentrations showing slightly better resilience.
The unmordanted wool samples display substantial color shifts, with a ΔE of 12.33 for the 1:10 dye ratio, categorized as a “different color,” and 6.89 for the 1:15 dye ratio, classified as
a “distinct” change. These values indicate that acidic perspiration can cause significant color alteration in unmordanted wool, particularly at higher dye concentrations. The “different color” classification for the 1:10 sample reflects how acidic conditions interact with the dye without mordant, leading to visible color shifts. The results suggest that acidic perspiration is a notable challenge for unmordanted wool, as it causes visible fading.
The potassium aluminum sulfate postmordanted cotton samples perform similarly, with ΔE values of 4.74 (1:10 ratio) and 8.97 (1:15 ratio). The 1:10 sample is rated as a “medium” change, while the 1:15 is categorized as “distinct” change indicating that potassium sulfate postmordanting offers only limited colorfastness in acidic perspiration environments, though it performs slightly better than ferrous sulfate in some cases..
For the potassium aluminum sulfate postmordanted wool, the 1:10 ratio has an ΔE of 7.09, categorized as “distinct,” while the 1:15 ratio shows an ΔE of 9.86, also “distinct.” These results indicate that the potassium aluminum sulfate postmordanting provides limited protection under acidic perspiration conditions, though it performs slightly better than ferrous sulfate. Acidic perspiration is a challenging factor for maintaining wool’s color stability.
Basic Perspiration
In Table 6, the unmordanted samples show moderate color shifts. The 1:10 dye ratio has an ΔE of 5.48, rated as “medium,” while the 1:15 ratio has an ΔE of 4.11, also categorized as “medium.” This suggests that basic perspiration can cause moderate fading in cotton dyed without mordants, with color stability slightly improving at lower dye concentrations. The findings align with one study that observed significant changes in color strength (K/S values) and color appearance for cotton fabrics subjected to perspiration tests30. Their results, which showed increased redness (+a), yellowness (+b), and lightness (L*), indicate that perspiration leads to visible color alterations in unmordanted fabrics. This supports the moderate fading observed in this study under basic perspiration conditions.
The unmordanted wool samples show significant color changes in basic perspiration. The 1:10 dye ratio sample has a ΔE of 12.49, rated as a “different color” change, while the 1:15 dye ratio has an ΔE of 6.87, classified as a “distinct” change. These results indicate that basic perspiration conditions lead to substantial fading or color alteration in unmordanted wool, with higher dye concentrations experiencing even more drastic changes. The potassium aluminum sulfate postmordanted cotton samples show a “distinct” change for both dye ratios, with ΔE values of 6.84 (1:10) and 9.52 (1:15). This implies that potassium aluminum sulfate postmordanting may not offer strong protection in basic perspiration, as significant fading still occurs in both concentrations. The potassium aluminum sulfate postmordanted wool also exhibits significant changes in basic conditions, with ΔE values of 9.67 (1:10, “distinct”) and 6.58 (1:15, “distinct”). These results suggest that potassium aluminum sulfate, while slightly more stable than ferrous sulfate, still does not offer sufficient color protection in basic perspiration. Wool dyes appear particularly susceptible to fading or alteration in alkaline environments.
 |
Table 6: Stability of Characterized Colors in Simulated Basic Perspiration |
Figure 2 shows the visual summary of the colorfastness test in cotton and wool fabrics. Results show the stability of natural dye from Chromolaena odorata in fabrics based on the color change (ΔE) values.
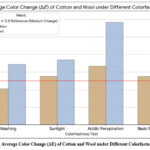 |
Figure 2: Average Color Change (ΔE) of Cotton and Wool under Different Colorfastness Tests. |
These findings not only confirm the potential of natural dyes as viable alternatives to synthetic ones but also underscore their environmental and economic relevance. Utilizing plant-based dyes derived from invasive or locally abundant species offers a sustainable pathway for waste valorization, turning ecological challenges into valuable resources. Moreover, adopting such eco-friendly dyeing practices could benefit local textile and cottage industries, promoting greener production processes, reducing chemical pollution, and enhancing the market appeal of naturally dyed fabrics. Overall, the study highlights the promise of natural dyes as both a sustainable innovation and an opportunity for circular economy integration in the textile sector.
Discussion
This study investigated the characterization of color and application of Chromolaena odorata leaves as a natural dye for cotton and wool fabrics. It evaluated the effects of various factors, including dye solution ratios, types of mordants, mordanting processes, and fabric types, on the color properties and stability of the natural dye. The dyeing process involved varying the mass of leaves and solvent volumes to create dye solutions, resulting in different color shades on cotton and wool fabrics. Cotton and wool exhibited varying levels of dye absorption and retention, leading to more vibrant and saturated colors depending on the dye solution ratio. Mordanting techniques—premordanting, metamordanting, and postmordanting—significantly influenced color uptake, resulting in diverse saturation levels and tonal variations. Additionally, different types of mordants, such as ferrous sulfate and potassium aluminum sulfate, affected the color outcomes and enhanced the dye’s stability across various tests. The findings of this study support the study of Li et al. which observed that unmordanted fabrics exhibited lighter and less saturated hues with significantly lower color strength. The samples treated with ferrous sulfate exhibit darker shades, as indicated by lower L∗ values, with colors predominantly leaning towards greenish-brown hues due to moderate a∗ and b∗ values28.
Moreover, the stability of the dye under different conditions was quantitatively assessed using ΔE (color difference) measurements. The dyed fabrics were subjected to washing, sunlight exposure, and simulated perspiration in acidic and basic conditions to evaluate colorfastness. Both cotton and wool fabrics showed noticeable changes in their initial colors, highlighting the versatility of Chromolaena odorata as a natural dye. By analyzing the effects of mordanting techniques, mordant types, and dye solution ratios, the study identified key factors influencing color stability and hue changes under diverse conditions.
In addition, results also show that potassium aluminum sulfate postmordanted cotton samples exhibited much lower color changes, particularly at the 1:15 ratio, which showed a ΔE of only 1.75, categorized as a “very light” change, while the 1:10 ratio had a slightly higher ΔE of 2.59, labeled as a “light” change. These results demonstrate that potassium aluminum sulfate postmordanting significantly improves wash fastness by enhancing dye stability and minimizing color loss during washing, particularly at lower dye concentrations. These findings align with the results of the study of Hanafy and El-Hennawi wherein alum-treated viscose samples dyed with natural turmeric achieved a color yield of 85%25. Alum-treated fabrics also earned a wash fastness rating of 4 out of 5, signifying good durability and retention of color during washing.
In contrast, on post-exposure to sunlight, the findings of this study contradict the results of Kumbhar et al. which reported that unmordanted fabrics typically exhibit poor lightfastness due to dye molecules being highly susceptible to degradation under light exposure24. However, the ΔE values observed in this study indicate relatively better stability in unmordanted cotton under sunlight, particularly at lower dye concentrations. These differences suggest that the conditions or dye characteristics used in the present study may have contributed to improved lightfastness despite the absence of mordant.
Similarly, the potassium aluminum sulfate postmordanted wool shows similar results, with ΔE values of 5.33 (1:10, “medium”) and 3.64 (1:15, “medium”). While this mordant provides a slightly lower degree of fading at lower dye ratios, both mordants indicate that sunlight remains a moderate change for wool. This results aligns to the study of Kundal and Singh which reported that wool samples exposed to light for over 15 hours showed varying degrees of fastness depending on the mordanting process26.
In acidic perspiration, the findings of this study align with the results of Mongkholrattanasit et al. that unmordanted cotton fabrics dyed with natural indigo exhibited minimal fading27. The study demonstrated color and staining ratings of 4 and 4-5, respectively, under acidic perspiration conditions, highlighting the moderate stability of unmordanted cotton fabrics.
In contrast, for basic perspiration, results of the study contradict the result of Li et al. which reported that wool samples subjected to perspiration testing displayed significantly better resistance to color changes28. Their findings indicated minimal overall color shifts, with only a slight but statistically significant increase in yellowness (+b) and no notable decrease in color strength (K/S values). The results of previously cited study emphasize that wool generally demonstrates better perspiration fastness compared to cotton, retaining its color more effectively under similar conditions, which is inconsistent with the considerable fading observed in this study.
Conclusion
Cotton exhibited superior dye absorption and retention compared to wool. Increased leaf concentration resulted in deeper, more saturated colors, while higher solvent volumes produced lighter shades. Among mordanting techniques, metamordanting and postmordanting yielded enhanced color stability, particularly with potassium aluminum sulfate. These results underscore the potential of Chromolaena odorata leaves as an eco-friendly and sustainable natural dye for textile applications.
Acknowledgement
The authors would like to thank the Director of the Research Publication and Innovation Center of the University of the Immaculate Conception for all the support.
Funding Sources
The authors did not receive any sponsorship to carry out the research reported in the present manuscript. However, article processing fee of publication will be requested for funding.
Conflict of Interest
The authors declared that they have no conflicts of interest to disclose.
Data Availability Statement
This statement does not apply to this article.
Ethical Statement
The study does not involve human participants that require ethics approval, instead Certificate of Exemption was acquired.
Informed Consent Statement
This statement does not apply in this article.
Author’s Contribution
Jelome Tagud: Visualization, Methodology, Experimented, Data analysis, Writing- original draft.
Girlie Mae Zabala: Visualization, Draft corrections, Writing- review and editing.
Venchie Badong and Kathleen Olaño: Writing- review and editing.
References
- Purwar, S. J. Home Sci. 2016, 2 (2), 283–287. https://www.homesciencejournal.com/ archives/2016/vol2issue2/PartE/2-2-40.pdf.
- Ado, A.; Yahaya, H.; Kwalli, A. A.; Abdulkadir, R. S. J. Environ. Monit. Prot. 2014, 1 (5), 76–81.
- Fobiri, G. K. Leather Rev. 2022, 5, 180–198. https://doi.org/10.31881/tlr.2022.22.
CrossRef - Yaseen, D. A.; Scholz, M. J. Environ. Sci. Technol. 2018, 16 (2), 1193–1226. https://doi.org/10.1007/s13762-018-2130-z.
CrossRef - DeMark, K. Apparel Merchandising and Product Development Undergraduate Honors Theses 2021, https://scholarworks.uark.edu/ampduht/17.
- Hong, I. K.; Jeon, H.; Lee, S. B. Ind. Eng. Chem. 2015, 24, 326–332. https://doi.org/10.1016/j.jiec.2014.10.004.
CrossRef - Saxena, S.; Raja, A. S. M. N. In Textile Science and Clothing Technology; Springer: Singapore, 2014, 37–80. https://doi.org/10.1007/978-981-287-065-0_2.
CrossRef - Sanda, B.; Indrie, L. Handbook of Natural Dyes; Wiley, 2021, 85–110. https://doi.org/10.1002/9781119710288.ch4.
CrossRef - Montiague, E. N.; Guzman, J. P.; Unciano, N. M.; Panerio, E. G.; Bigol, U. G.; Castro, I. J. L.; Jose, J. P. G.; Mantaring, S. D. A. Res. Environ. Appl. Mycol. 2020, 10 (1), 167–175. https://doi.org/10.5943/cream/10/1/17.
CrossRef - Bergkamp, G; Lim, J.; Vandeweerd, V.; Whisnant R. Current Status and Opportunities; 2018, 15-16. https://seaknowledgebank.net/sites/default/files/wastewater_management_and_resource_recovery_in_Philippines_0.pdf.
- Cimafranca, A. J.; Cueto, A.; Desengano, K. A.; Janer, C.; Mejia, M. A.; Balatico, G.; Mariano, L. I.; Antorcha, M. E. N. Antorcha 2016, 3 (1). https://research-manila.letran.edu.ph/read/30.
- Zahara, M. IOP Conf. Ser.: Mater. Sci. Eng. 2019, 506, 012022. https://doi.org/10.1088/1757-899x/506/1/012022.
CrossRef - Buchori, D.; Rizali, A.; Lukvitasari, L.; Triwidodo, H. Diversity 2020, 12 (9), 344. https://doi.org/10.3390/d12090344.
CrossRef - Pham, C. T. N.; Phan, H. N.; Hoang, T. T.; Dao, T. T. T.; Bui, H. M. J. Text. Apparel 2022, 26 (4), 499–510. https://doi.org/10.1108/rjta-07-2022-0089.
CrossRef - Bai, R.; Yu, Y.; Wang, Q.; Yuan, J.; Fan, X. Fibers Polym. 2016, 17 (10), 1613–1620. https://doi.org/10.1007/s12221-016-5598-5.
CrossRef - Baaka, N.; Ticha, M. B.; Guesmi, A. Fibers Polym. 2019, 20 (12), 2522–2528. https://doi.org/10.1007/s12221-019-9115-5.
CrossRef - Adetuyi, A.; Jabar, J.; Oyetade, J.; Ugwu, J.; Abe, T.; Fagbenro, M. ResearchGate 2020, 3. https://www.researchgate.net/publication/341443308.
- Dulmalik, D.; Rosliani, S.; Rosliana, S. Eng. Appl. Technol. 2021, 2 (2). https://doi.org/10.21831/jeatech.v2i2.42030.
CrossRef - Kiron, M. I. C. Textile Learner 2021, 1. https://textilelearner.net/colour-fastness-to-washing-procedure/.
- Rasel, Z. U.; Khatun, H.; Ahmed, F.; Kadri, J.; Maitra, B.; Saha, B. P.; Rabbi, M. A. Square 2022, 4-5. https://doi.org/10.21203/rs.3.rs-1470064/v1.
CrossRef - Musdalifah; Maulina, R. D.; Nurmasitah, S.; Damayanti. IOP Conf. Ser.: Earth Environ. Sci. 2022, 969, 012042. https://doi.org/10.1088/1755-1315/969/1/012042.
CrossRef - Park, S.; Kim, H.; Lee, S. RSC Adv. 2023, 13 (41), 28444–28461. https://doi.org/10.1039/d3ra04276f.
CrossRef - Saefudin; Basri, E. IOP Conf. Ser.: Earth Environ. Sci. 2022, 976, 012049. https://doi.org/10.1088/1755-1315/976/1/012049.
CrossRef - Kumbhar, S.; Hankare, P.; Sabale, S.; Kumbhar, R. Chem. Lett. 2019, 17 (2), 1161–1166. https://doi.org/10.1007/s10311-018-00854-w.
CrossRef - Hanafy, N.; El-Hennawi, H. M. J. Text. Polym. Sci. Technol. 2022, 19 (2), 349–355. https://doi.org/10.21608/jtcps.2023.213870.1185.
CrossRef - Kundal, J.; Singh, S. V. Prod. Chem. Res. 2016, 4 (3). https://doi.org/10.4172/2329-6836.1000214.
CrossRef - Mongkholrattanasit, R.; Klaichoi, C.; Mudchiew, O.; Punrattanasin, N.; Sasivatchutikool, N.; Rungruangkitkrai, N. Mater. Res. 2014, 1030–1032, 418–421. https://doi.org/10.4028/www.scientific.net/amr.1030-1032.418.
CrossRef - Li, Y. V.; Malensek, N.; Sarkar, A. K.; Xiang, C. Text. Res. J. 2016, 34 (3), 223–234. https://doi.org/10.1177/0887302×16647124.
CrossRef
Accepted on: 21 Oct 2025
Second Review by: Dr. Ramesh Bhargaw
Final Approval by: Dr. Tanay Pramanik