Antibacterial Potential of Rumex Vesicarius: A Phytochemical Perspective
1Department of Clinical Laboratory Sciences, College of Applied Medical Sciences, University of Hai’l, Hai’l, Saudi Arabia.
2Department of Pharmaceutical Chemistry, College of Pharmacy, University of Hai’l, Hai’l, Saudi Arabia.
3Department of Pharmaceutics, College of Pharmacy, Jazan University, Jazan, Saudi Arabia.
4College of Pharmacy, University of Hai’l, Hai’l, Saudi Arabia
5Department of Internal Medicine, King Salman specialist Hospital, Hail, Saudi Arabia
6Department of Pharmaceutics, College of Pharmacy, University of Hai’l, Hai’l, Saudi Arabia.
Corresponding Author E-mail:w.hussein@uoh.edu.sa
DOI : http://dx.doi.org/10.13005/ojc/410534
ABSTRACT:Known as a rich source of physiologically active metabolites, Rumex vesicarius has had multiple uses in Arabian traditional medicine. Present study focused on phytochemical profiling and antimicrobial effects of methanolic extracts of R. vesicarius leaves and flowers (MRL & MRF). GCMS used for detection of bioactive compounds showed the existence of numerous bioactive compounds such as fatty acids, flavonoids, and steroids. Microbial sensitivity assessed by well diffusion method showed significant activity against both Gram-positive and Gram-negative pathogens. To isolate the active ingredients and to further investigate their extensive biological functions, more investigation is required.
KEYWORDS:Antibacterial activity; Bioactive compounds; GC-MS analysis; Rumex vesicarius
Introduction
Rumex vesicarius L (R.V)., commonly known as bladder dock, has long been utilized in Arabian traditional medicine to treat ailments ranging from digestive disorders and fevers to microbial infections and inflammation 1,2. Recent phytochemical investigations have revealed that this desert plant is rich in bioactive constituents, including phenolics, flavonoids, fatty acids, and steroids 3,4. Moreover methanolic extracts of leaves and flowers (MRL &MRF) demonstrated potential antibacterial activity against both groups of gram pathogens 5. Despite its traditional relevance and initial scientific validation, detailed identification of its bioactive compounds—especially via advanced techniques like GC-MS and comprehensive antibacterial profiling remain limited. Aiming that addressing these gaps could support the development of new plant-derived therapeutics, particularly considering rising antibiotic resistance, this study was therefore undertaken.
Materials and Methods
Preparation of Plant Materials
Samples of R. vesicarius leaves and flowers were collected from the Hail city of Saudi Arabia and taxonomically authenticated by Dr. N. Hassan, Hafr Al-Batin University.
Extraction Procedure
Finely powdered RL and RF were used for the extraction. At room temperature, 25 g of the powder was extracted with 250 mL of methanol using ultrasonication in four 25-minute cycles over 24 hours in a water bath to enhance extraction efficiency. The mixture was filtered, air dried and used for further analysis 6.
GC–MS Analysis 7
Gas Chromatography–Mass Spectrometry (GC–MS) was used to analyze the key bioactive compounds in the MRL and MRF.The analysis was performed on a Thermo Scientific GC–MS-AS 3000 system equipped with an external quality detection detector. Separation was achieved using a TR 5MS capillary column, with helium as the carrier gas at 1.2 ml per min flow rate. For each analysis, two milliliters of the extract diluted in methanol were injected to allow partial separation of the chemical constituents. The mass spectra were recorded and processed using Xcalibur software, and the obtained spectra were compared with reference data from the National Institute of Standards and Technology library and the Main Library.
In Vitro Antibacterial Activity
The antibacterial activity of MRL & MRF was tested by well diffusion technique against selected bacterial pathogens on Mueller-Hinton agar plates using ciprofloxacin (CF) (5 micrograms per disc) as standard antibiotic by method followed by Syed. R.U. et al. 6
Results and Discussion
Several bioactive compounds were identified by GC-MS analysis that are likely responsible for the observed antibacterial activity, highlighting the plant’s potential as a natural source of antimicrobial agents and supporting its traditional medicinal applications. Table 2 and Figure 3 provide a summary of the major components of the floral extract while the Table 1 and Figure 2 provide specifics on the important compounds found in the leaf extract.
 |
Figure 1: Rumex vesicarius plantClick here to View Figure |
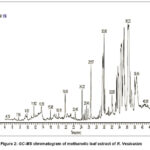 |
Figure 2: GC-MS chromatogram of methanolic leaf extract of R. VesicariusClick here to View Figure |
 |
Table 1: Chemical compositions of Methanolic extract of Rumex leaves via GC-MSClick here to View Table |
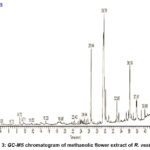 |
Figure 3: GC-MS chromatogram of methanolic flower extract of R. vesicariusClick here to View Figure |
 |
Table 2: Chemical compositions of methanolic extract of Rumex flower via GC-MSClick here to View Table |
Decanoic acid 8, phytol 9, glycerol 1-palmitate10, cis-Vaccenic acid 11, and Stigmasterol12 found in the extracts are considered as key contributors for the antibacterial activity. Decanoic acid, identified in the leaf extract, is a medium-chain fatty acid. 10, 13 Its amphiphilic nature enables integration into bacterial membranes, where it disrupts the integrity of the lipid bilayer, increases ion permeability, and ultimately leads to leakage of intracellular contents and cell death. 13, 14
Phytol, a diterpene present in the leaf extract, has an antibacterial effect by exerting oxidative stress and producing reactive oxygen species (ROS) in the bacterial cells. This mechanism was specifically observed in Pseudomonas aeruginosa, a pathogen that was also inhibited by R. vesicarius in this study. The antioxidant potential of R. vesicarius may also provide synergistic benefits by modulating host defense systems. 9
Monoglycerides such as glycerol 1-palmitate are known antibacterial lipids that insert into bacterial membranes and cause cell leakage. Their amphiphilic nature enhances solubility and interaction with phospholipid bilayers. The detection of glycerol esters in R. vesicarius strengthens the hypothesis that lipid derivatives play a central role in its antibacterial potential. 10, 15 The cis-vaccenic acid, detected in the leaf extract, is a monounsaturated fatty acid that is produced in microbial and plant systems. Its incorporation into bacterial membranes alters fluidity and permeability and impairs cell survival. In E. coli, cis-vaccenoic acid derivatives have been shown to interfere with lipid metabolism, suggesting that their presence in R. vesicarius may explain the strong inhibition against E. coli. 11
The stigmasterol found in the flower extract, was isolated from several medicinal plants and has antibacterial and antioxidant properties. 16 The sterol backbone interferes with microbial membrane enzymes and cell wall synthesis. Its presence may be particularly important for the inhibition of Gram-positive cocci, which is consistent with the strong activity of R. vesicarius against Staphylococcus aureus. 12
Table 3 summarizes the antibacterial activity profile. The results show that the methanolic extracts of R. vesicarius17-19 leaves and flowers showed the strongest inhibitory effects against most bacterial strains; however, their activity was still considerably lower than that of the standard antibiotic, ciprofloxacin.
Table 3: Results of in vitro antibacterial activity
| Test micro-organisms | Methanolic leaf extract |
Methanolic Flower Extract |
Standard Antibiotic Ciprofloxacin (5 mg/Disc) |
| Staphylococcus aureus ATCC 512477 | 22.67 ± 1.15 | 27.67±1.52 | 33.66 ± 1.24 |
| Staphylococcus epidermidis ATCC 12228 | 20.34 ± 1.53 | 25.34±1.53 | 33.3 ± 1.24 |
| E. coli ATCC 25922 | 27.67 ± 1.53 | 30.33±1.154 | 35.66 ± 0.9 |
| P. mirabilis ATCC 299 | 16 ±1 | 26± 1 | 23.66 ± 1.24 |
| Salmonella choleraesuis ATCC 10708 | 23.67 ± 1.52 | 28 ±4 | 33.3 ± 1.24 |
| K. pneumoniae ATCC 700603 | 23.67 ±4.72 | 26.33± 1.52 | 26 ± 1.4 |
| E. faecalis ATCC 29212 | 25.67 ± 3.2 | 28.33 ± 1.5 | 25.33 ± 0.4 |
| Pseudomonas aeruginosa ATCC 27853 | 25 ± 3.6 | 25.66 ±1.53 | 33.3 ± 0.8 |
#Each value is the mean of 3 batches with standard deviation. All values were compared with standard ciprofloxacin disc by performing Tukey Kramer test (post hoc). All the test values are significantly lesser than the standard ciprofloxacin disc at p < 0.05.
Conclusions
Methanolic extracts of R. vesicarius leaves and flowers grown in Hail, KSA, contain diverse bioactive components with antibacterial activity against most bacterial strains, though their potency remains notably lower than that of the standard antibiotic, ciprofloxacin. These findings, consistent with earlier research, reinforce the potential of R. vesicarius as an antibacterial agent in the context of rising antibiotic resistance. R. vesicarius could be a promising source for the development of new antibacterial drugs; however, further investigations are needed to confirm its effectiveness in topical and therapeutic applications and to assess the in vivo activity of its active constituents.
Acknowledgment
None.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
References
- Sarı, F.; Koçyiğit, M. Turk. J. Biosci. Collect. 2021, 5 (2), 123–140. https://doi.org/10.26650/ tjbc.2021930272.
CrossRef - Abou Elfotoh, M. A.; Shams, K. A.; Anthony, K. P.; Shahat, A. A.; Ibrahim, M. T.; Abdelhady, N. M.; Abdel Azim, N. S.; Hammouda, F. M.; El-Missiry, M. M.; Saleh, M. A. Antioxidants 2013, 2 (3), 167–180. https://doi.org/10.3390/antiox2030167.
CrossRef - Salama, S. A.; Al-Faifi, Z. E.; Masood, M. F.; El-Amier, Y. A. Molecules 2022, 27 (10), 3177. https://doi.org/10.3390/molecules27103177.
CrossRef - Tukappa, A. R. L. L. A.; Londonkar, R. L. Am. J. Drug Discov. Dev. 2013, 3, 72–83.
CrossRef - Sweilam, S. H.; Abd El Hafeez, M. S.; Mansour, M. A.; Mekky, R. H. Plants 2024, 13 (13), 1815. https://doi.org/10.3390/plants13131815.
CrossRef - Syed, R. U.; Moni, S. S.; Alfaisal, R. H.; Alrashidi, R. H.; Alrashidi, N. F.; Wadeed, K. M.; Alshammary, F. N.; Habib, A. M.; Alharbi, F. M.; ur Rehman, Z.; Alam, M. S. Arab. J. Chem. 2022, 15 (8), 104006. https://doi.org/10.1016/j.arabjc.2022.104006.
CrossRef - Syed, R. U.; Moni, S. S.; Lila, A. S. A.; Abdallah, M. H.; Abouzied, A. S.; Banu, H.; Alreshidi, K. S. M.; Alrashidi, B. M. W.; Hadi, M. A.; El-Horany, H.; Abdelwahab, S. I.; Taha, M. M. E. Life 2022, 12 (10), 1641. https://doi.org/10.3390/life12101641.
CrossRef - Shen, T.; Chen, L.; Liu, Y.; Shi, S.; Liu, Z.; Cai, K.; Liao, C.; Wang, C. Eur. J. Pharm. Sci. 2021, 157, 105609. https://doi.org/10.1016/j.ejps.2020.105609.
CrossRef - Lee, W.; Woo, E. R.; Lee, D. G. Free Radic. Res. 2016, 50 (12), 1309–1318. https://doi.org/10.1080/ 10715762.2016.1241395.
CrossRef - Yoon, B. K.; Jackman, J. A.; Valle-González, E. R.; Cho, N. J. Int. J. Mol. Sci. 2018, 19 (4), 1114. https://doi.org/10.3390/ijms19041114.
CrossRef - Do, K. H.; Park, H. M.; Kim, S. K.; Yun, H. S. Biotechnol. Bioprocess Eng. 2018, 23 (1), 100–107. https://doi.org/10.1007/s12257-017-0473-9.
CrossRef - Yohanna, C.; Kwaji, A.; Atiko, R. Int. J. Biochem. Res. Rev. 2021, 30 (9), 288–301. https://doi.org/10.9734/IJBCRR/2021/v30i930288.
CrossRef - Black, R. A.; Blosser, M. C. Life 2016, 6 (3), 33. https://doi.org/10.3390/life6030033.
CrossRef - Mett, J.; Müller, U. Sci. Rep. 2021, 11 (1), 6135. https://doi.org/10.1038/s41598-021-85542-3.
CrossRef - Churchward, C. P.; Alany, R. G.; Snyder, L. A. S. Crit. Rev. Microbiol. 2018, 44 (5), 561–570. https://doi.org/10.1080/1040841X.2018.1475319.
CrossRef - Bakrim, S.; Benkhaira, N.; Bourais, I.; Benali, T.; Lee, L.-H.; El Omari, N.; Sheikh, R. A.; Goh, K. W.; Ming, L. C.; Bouyahya, A. Antioxidants 2022, 11 (10), 1912. https://doi.org/10.3390/antiox11101912.
CrossRef - Mostafa, H. A. M.; Elbakry, A. A.; Eman, A. A. Int. J. Pharm. Pharm. Sci. 2011, 3 (2), 109–118.
- Laouini, S. E.; Ouahrani, M. R. Sci. Study Res. Chem. Chem. Eng. Biotechnol. Food Ind. 2017, 18 (4), 367–376.
- Sulieman, A. M. E.; Abdallah, E. M.; Alanazi, N. A.; Idriss, H.; Adnan, M.; Jamal, A.; Shommo, S. A.; Snoussi, M. Front. Microbiol. 2024, 15, 1421143. https://doi.org/10.3389/fmicb.2024.1421143.
CrossRef
Accepted on: 10 Oct 2025
Second Review by: Dr. Mohan Tewari
Final Approval by: Dr. Charanjeet Kaur

















