Potential Utilization of Eco Benign Material as Effective Divalent Ions Encapsulant from Aqueous Matrix and Industrial Effluent
Department of Chemistry, PSGR Krishnammal College for Women, Coimbatore, Tamil nadu, India.
Corresponding Author E-mail:muthulakshmiandal@psgrkcw.ac.in
DOI : http://dx.doi.org/10.13005/ojc/410237
ABSTRACT:Release of toxicants from multiple sectors into water streams cause adverse effects in the environment. In recent years, due to their great persistence and toxic nature, heavy metals are considered as a major threat generated from diverse industrial units. Amongst various pollutants, high level of heavy metal (Zn, Pb, Hg, Ni, Cu & Fe) contamination create numerous health hazards like immune system dysfunction, gastrointestinal problems, birth defects, nervous disorders and thereby affecting surface water quality. This study is centred on the preparation of magnetite composite using chemically Treated Anas platyrhyncos Legs Powder (TALPMC) as a precursor to abstract Zn(II) and Pb(II) ions aqueous media. Metal sequestering ability of the treated powder (TALP) and prepared magnetite composite (TALPMC) were studied via pilot method. Batch Equilibration verification was done for TALPMC to optimize the influential parameters viz., particle sizes, sorbent dose, initial concentration of sorbate species, time interval, pH and temperature, so as to regulate the sorptive effectiveness. Microscopic analysis, BET / BJH, FT-IR, SEM / EDAX, TG-DTA and XRD studies were conducted to characterize the physical and chemical nature of the material. Initial and residual experimental run concentrations of metal solutions were interpreted using Atomic Absorption Spectrophotometer. Sorption capacity of TALP (89% Zn(II) and 85% Pb(II)) was observed to multiply by homogenizing it with the prepared magnetite, this resulted in 97% Zn(II) and 95% Pb(II) removal. Desorption and regeneration experiments were performed to examine the reusable nature of exhausted TALPMC. Electrostatic attraction between Zn2+ and negatively charged surface of TALPMC shall be the ascertained reason. Upon derived plots isothermal studies, most linear fit of Langmuir isotherm favoured monolayer sorption. Kinetic studies revealed the applicability of pseudo-second order model. As per the results obtained from Batch Optimization assay, extensive quantity of toxicants was immobilized by TALPMC than TALP. Thus, to ascertain the efficacy of TALPMC against industrial discharges, effluent sample was collected from oil industry and a pilot study was performed for magnetite systems. Trapping of 92% Zn(II) and 88% Pb(II) ions from the raw sample was registered by TALPMC. From the made observations, it is concluded that TALPMC serves as an excellent metal scavenger against its precursor.
KEYWORDS:Batch; Lead; Magnetite; Precursor; Sorption; Zinc
Introduction
Industries use water resource for countless operations such as coolant in power plants, solvent in oil refineries and in other manufacturing units1. Exponential rise in water scarcity is due to expansion of urbanization and industrial operations2. 22% of available water sources is consumed by industries and released directly into water bodies3. Oils, greases, dyes, acids, alkalis and noxious heavy metals are few prominent materials present as toxicants in water4. These are non-biodegradable and progressively accumulate in the food chain5, thereby increasing their concentrations in living organisms over time6. Therefore, water quality maintenance is extremely required to meet the global usability and scarcity of water resources7. Of these, oil producing plants vent out chemicals, which deplete oxygen, stimulate the growth of pathogens and affect the marine ecosystem8. Many methodologies have been endorsed in the process of scavenging these metal ions, of which adsorption is found to be the most operative method because of its selectivity, feasibility and effectiveness, whereas other reformative techniques generate large amounts of sludge which destroy surrounding environs and their handling is very expensive. Pollutants discharged by the oil yielding companies generally contain heavy metals like Cd, Pb, Cu, Fe and Zn. Of these distinct toxicants, abstraction of Zinc and lead ions under laboratory set up employing treated and magnetic sorbent is eventually explained in this work, because of the permissible limit that exceeds slightly in the effluent collected from the concerned industry. Prevalence of heavy metals in oil may be due to contamination that occurs during the collection of seeds, addition of excess fertilizers to soil, plants that affects its metabolic processes or by the procedures employed during oil extraction.
Zinc, the essential element for many physiological reactions that eventuate in human body, thus promoting metabolism and functioning of immune system9. But when surplus amount of zinc (used in food preservation, agriculture, food processing industries etc), consumed via food, cause serious health hazards. Inspite of its widespread applications, excess of zinc ions perturbs human health, causing gastrointestinal disorders, infections in respiratory tract, epigastric pain and other neurological defects10.
Lead is the highly poisonous metal, affecting almost every organ in the body. Existence of lead ions in pipes, that are employed in the industrial process has a direct or indirect influence in polluting ground water resources11. Thus, long-time exposure to lead can cause anaemia, thereby increasing blood pressure. Severe damage to the brain and kidneys, results in death12. In pregnant women, high exposure to lead may cause miscarriage. Chronic lead exposure results in the reduction of fertility in males13.
Naturally available minerals, biopolymers and nanoparticles have been reported in literature to treat these oil effluent discharges14. The present investigation is focussed on synthesizing magnetite composite using (AL) as precursor, followed by evaluating its adsorbing ability upon zinc and lead ions in aqueous environs. The choice of eco litter material is based on its widespread availability, financial feasibility apart from non-being reported elsewhere in exploring its adsorption properties. Sorption capacity of Anas platyrhyncos Legs was found to be efficient in chelating heavy metals in comparison with other animal bone wastes 15, investigated by N. A. Medellin-Castillo et al.
Materials and Methods
Collection and Modification of Sorbent
Anas platyrhyncos is a variety of duck species, classified under family Anatidae. It is a popular form of poultry and raised throughout temperate parts of Tamil nadu as a part of industrialized farming, making it viable for the edible meat consumption. However, the legs of these ducks are thrown as litter. They were gathered from various localities of Nammakal district, familiar for poultry activities. The collected legs were washed thoroughly and the leg bones were carbonized in an oven (300ºC for 2 hours) and later dried (Fig 1). The parched material was crushed, powdered in an electrical mixer (Fig 2) and categorized into different mesh sizes (85BSS, 72BSS, 52BSS, 36BSS and 22BSS). Further, appropriate doses of these materials were transferred to a beaker containing 100mL of 0.1 N HCl and heated for 3 hours. The same procedure was repeated using 0.1 N NaOH. This chemical modification ensured the enhancement of their surface properties, thereby reflecting upon metal sorbing capacities.
Synthesis of Magnetite
Ferrous and ferric chloride salts were mixed in 1:2 ratio and dissolved in 5 mL distilled water. The yellowish brown mixture was agitated well using magnetic stirrer until it turned to reddish brown hue. Few drops of NaOH solution was added to produce iron oxides and to ensure complete dissolution. The forms black magnetite precipitate was left behind (Fig3) undisturbed for few hours to assure absolute formation of Fe3O4. Colour of the magnetite depends on the intervalence charge transfer between Fe2 + and Fe3 + in its crystal structure16.
Preparation of Anas platyrhyncos Legs Powder Magnetite Composite (TALPMC)
7g of TALP precursor was suspended in a volume of 20mL prepared magnetite solution. This was stirred for 45 minutes to obtain a homogeneous mix of Anas platyrhyncos legs powder magnetite composite, labelled as TALPMC (Fig 4). The derived magnetic composite was decanted, dehydrated (80º C) and stored for further analyses.
 |
Figure 1: Anas platyrhyncos Legs (AL) |
 |
Figure 2: Treated AL Powder (TALP). |
 |
Figure 3: Magnetite. |
 |
Fig 4: TALP Magnetite Composite (TALPMC) |
Adsorbate Solutions Preparation
Specific amounts of zinc sulphate and lead nitrate salts (Sigma Aldrich) were dissolved in 1000 mL double distilled water and labelled as stock solutions. Appropriate dilutions of these stock solutions were done to prepare 100 mg/L standards. Further, aliquots ranging from 5-25 mg/L: 5 mg/L were made up in respective standard flasks.
Pilot Study
Batch Equilibration studies were designed to assess the metal trapping capacity of the sized materials. 50mL of 50mg/L Zn(II) and Pb(II) solutions were agitated for 30 minutes with 1g dose of treated (TALP) and its magnetic counterpart (TALPMC). Better metal confiscation is observed for TALPMC rather than its treated precursor as given in table 1. The Batch reaction conditions were optimized for both the materials, since they unveiled notable percentage removal in the pilot studies.
Table 1: Removal of Divalent ions – Pilot study
| % Removal | ||
| Materials | Zn(II) | Pb(II) |
| TALP | 73.2 | 71.3 |
| TALPMC | 78.4 | 76.2 |
Characterization Studies
Native, treated and magnetite sorbents were subjected to characterization studies using Ultrasound microscope, BET / BJH, FTIR, SEM/EDAX, TG-DTA and XRD, so as to determine their structure, surface area /porous nature, involvement of functional groups, composition of elements both in unloaded and metal loaded materials, thermal behaviour and crystallinity of the magnetic sorbent.
Binocular Ultrasound microscope (OLYMPUS make, Model- CX21I) was used to determine the particle sizes. Bruner-Emmet-Teller (BET) and Barrett-Joyner-Halenda (BJH) plots were used to determine the surface areas and porous nature of the modified precursors, equipped with Adsorption /Desorption data analysis software. Participation of functional groups in the sorption process was studied using Fourier Transform Infrared Spectrophotometer (Shimadzu). Scanning Electron Microscope (SEM) and Energy Dispersive X-ray Spectrometer (EDAX) spectra of ZEISS make were recorded to signify surface morphological changes and framework of the metal laden and unloaded counterparts. TG / DTA curves were derived using NETZSCH JUPITOR STA 449F3 for analysing the thermal stability, mass loss and composition of the material at varying temperatures. Crystalline nature of the magnetite sorbent was estimated by XRD of Bruker D8 Advance make, λ = 0.154 nm.
Batch Equilibration Studies
Sorption efficiencies of the four systems viz., Zn(II)-TALP ; Zn(II)-TALPMC ; Pb(II)-TALP and Pb(II)-TALPMC were optimized with underlying factors viz., particle size (0.18, 0.21, 0.30, 0.42, 0.71 mm); initial sorbate concentrations (5-25 mg/L : 5 mg/L); Dose (10 – 60 mg : 10 mg); agitation time period (3-30 mins: 3mins). Batch experiments were designed in such a way that the specified dose of both the materials being added to a 250 mL Erlenmeyer flasks containing standard volume of 50mL Zn and Pb solutions of specified concentrations. Under the predetermined circumstances, the contents of the flask were shaken in a mechanical shaker (KEMI model) at a speed of 140 rpm. Later, these samples were filtered, analysed for initial and residual metal concentrations using Atomic Absorption Spectrophotometer (Shimadzu (AA 6200) model) at the corresponding wavelengths of 213.9 nm and 324.8nm for Zn(II) and Pb(II). Role of key parameters like pH (ELICO (LI-120) pH metre) 3,5,7,9,11 and temperature (TECHNO thermostat – controlled incubator shaker) 293-333 K: 10K were listed for Zn(II)-TALPMC and Pb(II)- TALPMC systems under fixed conditions, since these factors play a vital role in controlling the concentration and mobility of sorbate ions in the solution.
Desorption / Regeneration Experiments
Adsorption / desorption studies were conducted to evaluate the sustainable nature of TALPMC against aqueous/industrial effluents, thereby promoting economic viability of the process. An effectual desorption procedure includes appropriate eluent selection to desorb the sorbent species and sorption mechanism. The strength of desorbing medium (HCl) was fixed as 0.01N because of its ability to subside the desorption rate, indicating maximal sorbent redemption at various consecutive cycles.
Pilot Study – Oil Effluent Sample
The results obtained from pilot study being in favour of the sequestering ability of TALPMC has led to focus on the up scaling of the material’s applicability to abstract the specified divalent ions from raw effluent sample. However, the choice of heavy metals (Zn & Pb) for the present study was deliberately based on their inclusive occurrence in the oil effluent, transcending the permissible limits. In this context, effluent sample was collected from oil refinery located at Karur, Tamil Nadu, India. Metal concentrations in the collected oil effluent sample analysed using Atomic Absorption Spectrophotometer (AAS) within the corresponding wavelengths and slit width exposed their intensities as to exceed the prescribed tolerance levels (WHO Standards). In order to align with the tested aqueous metal solution concentrations, the sample was processed as follows:
500mL of the sample was poured into a beaker, heated to 80ºC for 30 minutes, followed by the addition of 5N conc. H2SO4, to separate the sludge from the boiled sample. Later, extraction procedure was done to discard the sludge, and the sample was diluted to explicit concentrations. 50mL of the thinned samples were transferred into 250 mL flasks, followed by the addition of TALPMC dose (10- 50mg: 10mg) agitated (120 rpm) in a shaker at preset time intervals (5-30: 5 mins) and filtered. Metal residual concentrations of the sorted and that of the pre-run samples were determined.
Adsorption Isotherms and Kinetics
Adsorption data were graphically determined for Zn(II)-TALPMC & Pb(II)-TALPMC systems, based on the optimized results. Langmuir, Freundlich and Tempkin isotherms models were taken into account to validate and predict the interaction between sorbate ions (Zn(II) & Pb(II)) and sorbent surface (TALPMC). Also, the sorbing efficiency of chosen sorbent against cations was determined using pseudo- first-order and pseudo-second-order kinetic models.
Results and Discussion
Microscopic Studies
Range of porosity in descending order, is represented in figures 5a to 5c. Modified TALP and prepared TALP magnetite composite (Fig 5c) appear to possess granular pore size, with varying fineness than their natant material. This would be due to opening of cavities during acid/base treatment and enabling the formation of magnetite complex respectively.
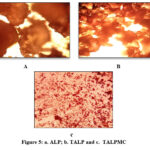 |
Figure 5: a. ALP; b. TALP and c. TALPMC |
BET / BJH
BET/BJH graphs (Fig 6 & 7) clearly show the total specific exterior TALPMC surface area, internal pore volume and pore size distribution data as depicted in table 2. Adsorbents for liquid phase adsorbate should be mesoporous to accommodate bigger liquid molecules. The type IV adsorption-desorption isotherm (Fig 8), which shows its textural feature, demonstrates the mesoporous nature with significant surface area. A narrow porous matrix with a wide capillary width and a non-uniform size is preferred by the pore size distribution pattern, which is centred at 5 nm. Aggregates of plate-like particles with slit pores are represented by a meagre broad H3 type hysteresis loop (clear hysteresis at P/P° > 0.6).
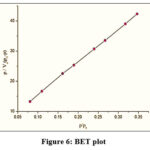 |
Figure 6: BET plot. |
 |
Figure 7: BJH plot. |
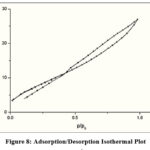 |
Figure 8: Adsorption/Desorption Isothermal Plot |
Table 2: BET / BJH parameters
| Specific surface area (m²/g) | 30.85 |
| Pore size distribution (nm) | 5 |
| Total pore volume (cm3/g) | 0.45 |
FT-IR
Pre and post run spectra of TALP are shown in fig 9a. Notable bands around 700cm-1 and sharp peak at 1410 cm-1 in sorbate ions loaded TALP indicate the participation of Ca ions in sequestering the divalent cations. Broad band shift from 3525cm-1 in TALP to 3484 cm-1, 3437 cm-1 in loaded spectra confirm the stretching of alcoholic –OH group. A sharp peak at 1020cm-1 and 1017cm-1 imply the C-O stretching to adsorb Zn(II) and Pb(II) ions. Involvement of alkane group (C-H stretching) in modified and metal sorbed materials is evident from corresponding peak shift from 2696 cm-1 to 2908 and 2904 cm-1.
FTIR spectra of magnetite complex and its laden counterparts are depicted in Fig 9b. Band at 3514.8cm-1 for OH group, indicate the presence of alcoholic –OH group in the precursor. Appearance of new peaks at 2927cm-1 & 2922cm-1 in the metal laden counterparts denote that the stretching of C-H bonds in alkane group had occurred in the native material. Bands at 1400cm-1and 1080cm-1 refer to the contribution of Ca2+ ions towards sorption of divalent ions, where replacement of cations (Zn2+ and Pb2+) by calcium ions occur on the sorbents’ surface (ion exchange mechanism), thereby extracting the divalent ions from the solution. New peaks at 1706 cm-1 and 1704 cm-1 in the metal loaded sorbents reveals C=C bending in TALPMC. A prominent band around 580cm-1 confirms the binding of Fe3O4 to the treated material. The chelating efficiency of TALPMC against Zn(II) and Pb(II) is further proved by broad and intense bands at 580cm-1in metal laden spectra. Thus, variation and shift in the peaks shows the involvement of functional groups during the sorption process.
 |
Figure 9a: TALP, Zn(II) –TALP , Pb(II)-TALP |
 |
Figure 9b: TALPMC, Zn (II) –TALPMC, Pb(II) -TALPMC. |
Surface Topographical Investigation (SEM/EDAX)
Rough surface pores of activated sorbent, TALP (Fig 10a) is uniformly occupied by Zn(II) (Fig 10b) and Pb(II) ions (Fig 10c) clusters. Rod shape arrangement in TALPMC (fig 11a) substantiates marked changes from its appearance in fig 10a, which may be due to the augmentation of magnetite within the treated matrix (TALP). Post run images of the magnetite fused sorbent (Fig 11b & 11c) depicts the filling up of the open apertures by the respective sorbate ions, confirming their uptake.
 |
Figure 10: a: TALP; b: Zn(II)- TALP c: Pb(II)-TALP |
 |
Figure 11: a: TALPMC; b: Zn(II)-TALPMC; c: Pb(II)-TALPMC |
EDAX image (Fig 12a) depicts a peak at 7KeV corresponding to Cl atom, the presence of which shall be due to acid / base treatment of the raw material. Popping up of new peaks between 4-6 KeV is obvious from fig 12b & 12c, favouring the respective sorption of divalent ions. Prominent peaks in all EDAX images representing Ca ion further support the results envisaged in FT-IR spectra. Peaks at 6-8 KeV in fig 13a refers to Fe2+ ion, thereby ensuring its coherence within TALP to form the magnetite. Additional peaks at 5KeV and 2KeV in fig 13b & 13c confirm the adhesion of divalent ions onto the sorbent layer.
 |
Figure 12: a: TALP; b: Zn(II)-TALP; c: Pb(II)-TALP |
 |
Figure 13: a: TALPMC; b: Zn(II)-TALPMC; c: Pb(II)-TALPMC |
TALPMC was subjected to TG-DTA and XRD studies to determine its thermal stability, magnetic nature and crystallinity.
Thermal Analysis – TG /DTA
Thermogram (Fig 14) represents the thermal degradation behaviour at two regions of varied temperature conditions ranging from 100-600 ºC. Endothermic curve (red curve) at 100ºC depicts moisture evaporation. The broader exothermic curve (300-400ºC) imputes the decomposition of organic materials viz., collagen and minerals found in Anas platyrhyncos bones. A steady declined curve observed beyond 400ºC, indicates the carbonization of magnetite under controlled air atmosphere.
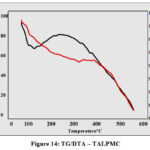 |
Figure 14: TG/DTA – TALPMC |
X-ray Diffraction Analysis
Figure 15represents the XRD pattern with broad peaks of less sharpness, predicting TALPMC’s amorphous nature at room temperature. This powdered characteristic shall be recognized as the presence of organic matter in the magnetite. Characteristic peaks of Fe3O4 at 2Ɵ as 30.1, 35.6, 43.2, 53.4, 57 and 62.7 relate to the planes (220), (311), (400), (422), (511) & (440), as per JCPDS card number: 85-1436. Infusion of Anas platyrhyncos Leg bones as precursor in the magnetite composite is further confirmed by the appearance of peaks at 2Ɵ angle (25.11, 38.8 and 49.5), referring to Hydroxy APatite (HAP), thereby proving the existence of Ca ions.
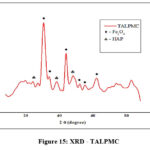 |
Figure 15: XRD – TALPMC |
Batch Optimization Studies
Influence of Particle Size
Particle size has a major impact on its sorption effectiveness towards any noxious substance. Figures 16a and 16b portray the chelating efficacies of derived materials at varying sizes. 0.18 mm of treated and magnetite based sorbent possess greater potential to confiscate divalent ions, evident from the varied bar height as 89% zinc, 85% lead ions (Fig 16a) and 97% Zn(II), 95% Pb(II) ions (Fig 16b). This shows that the sorbent with reduced size has expanded surface area, thence 0.18mm particle size was fixed for further experimental studies.
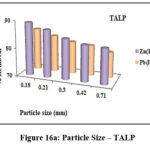 |
Figure 16a: Particle Size – TALP |
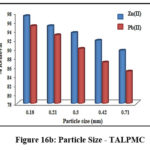 |
Figure 16b: Particle Size – TALPMC. |
Influence of Initial Concentration on Time
Sorption ability of TALP is demonstrated in figures 17a & 17b. Acquisition of active sites on TALP by Zn(II) and Pb(II) ions had occurred at 21 mins for 10 mg/L corresponding solutions. Rapid adsorption of Zn (89%) and Pb (85%) appeared initially and as the contact time boosts up, the ability of TALP grasping the sorbate species diminished, leading to agglomeration, sufficed by the trend of the curves.
Figures 18a & 18b pictures the effect of sorbate concentrations at periodic time intervals on TALPMC. Orderly distribution of Zn(II) and Pb(II) ions on the surface of the magnetite sorbent had occurred at 15 mins for concentrations of 10 mg/L and 15 mg/L. Utmost removal of zinc and lead ions had appeared at 97% and 95.1%, exhibited from the inverted parabolic curves. A decline in the sorption pattern is noticed while increasing the metal concentrations, accounting for a biased desorption of divalent ions on the solid matrix due to saturation of active sites.
 |
Figure 17a: Initial Concentration on Time – Zn(II)-TALP. |
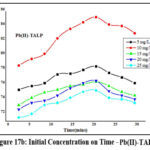 |
Figure 17b: Initial Concentration on Time – Pb(II)-TALP |
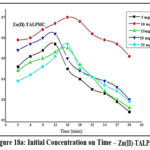 |
Figure 18a: Initial Concentration on Time – Zn(II)-TALPMC |
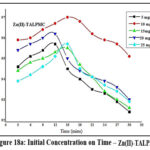 |
Figure 18b: Initial Concentration on Time – Pb(II)-TALPMC |
Influence of TALP and TALPMC Dosage
Variations of doses (10mg to 60 mg: 10 mg) are depicted in figures 19a, 19b & 20a, 20b. Bare orifices on surface pores of TALP had been captured by the divalent ions. The conditions optimized for maximal metal sequestration (Zn(II) – 89.4% & Pb(II)- 85.3%) are 50 mg TALP and 18 mins agitation time frames as manifested from figures 19a & 19b. However, a better inclined pattern is visible from the parabolic curves (fig 20a & 20b) reveals that the optimal TALPMC doses required were 30mg & 40mg for dispensing the capture of 97% Zn(II) and 95% Pb(II) ions respectively. Both the reactions registered uttermost metal abstraction within a time frame of 15 minutes. Decline in the curves, beyond the optimum dosage, imply aggregation of sorbent moieties at higher doses resulting in the decreased metal uptake.
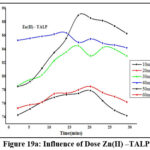 |
Figure 19a: Influence of Dose Zn(II) –TALP |
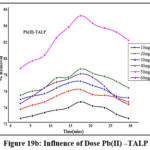 |
Figure 19b: Influence of Dose Pb(II) –TALP |
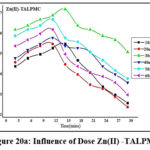 |
Figure 20a: Influence of Dose Zn(II) -TALPMC |
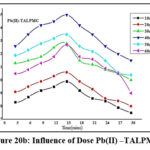 |
Figure 20b: Influence of Dose Pb(II) –TALPMC. |
From the made observations, it is evident that the chelation of divalent ions by TALPMC proved to be better than its precursor TALP. Hence, fixing of other parameters such as pH and temperature was limited to TALPMC systems.
Influence of pH
pH impact on any adsorption system plays a pivotal role, since the abstraction activity of the material can be enhanced in acidic/basic environments. In this context, removal of Zn & Pb ions under varied pH environs was studied and the resulting data is illustrated in fig 21. It is obvious from the figure that, Zn(II) removal had reached its pinnacle at pH 5, due to less competency between acidic H+ and metal cations at higher pH. This facilitates greater occupation of Zn(II) ions on the vacant sites of TALPMC. Thus, effective sorption is adhered, reflecting the deprotonation of carboxylic groups (15). Utmost Pb(II) acquisition had occurred at pH 7. At higher alkaline pH, the divalent ions get precipitated as hydroxides, diminishing TALPMCs’ quenching ability. Also, increasing amount of Na+, in basic medium competes with Zn2+ & Pb2+ ions to get preferentially sorbed on exchangeable active sites available on TALPMC surface.
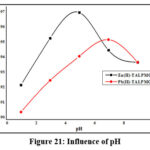 |
Figure 21: Influence of pH |
Influence of Temperature
Temperature studies expose its inverse relation against metal removal (%) as evident from fig 22. Notable confiscation of Zn2+ (97.3%) & Pb2+ (95.1%) ions had occurred at 303K, and 313K respectively. Limited availability of active centres at lower temperature and desorbing nature of metal ions from the magnetite surface at elevated temperatures has reflected in the percentage drop, under respective thermal conditions.
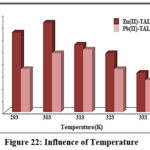 |
Figure 22: Influence of Temperature |
Desorption / Regeneration
Desorption capability and regenerating capacity of metals (Zn2+ & Pb2+) loaded TALPMC were analyzed for three continuous cycles, followed by Batch Optimization studies. Marked variations in adsorption / desorption cycles of spent sorbent are depicted in table 3 and it was found to be more pronounced for Zn(II) -TALPMC than Pb(II)-TALPMC system.
Table 3: Desorption / Regeneration Studies
| Cycle 1 (mg/g) | Cycle 2 (mg/g) | Cycle 3(mg/g) | |||||
| Systems | Adsorption | Desorption | Adsorption | Desorption | Adsorption | Desorption | |
| Aq. Zn(II) -TALPMC | 65.32 | 1.56 | 63.25 | 1.20 | 60.58 | 0.98 | |
| Aq. Pb(II) -TALPMC | 60.15 | 2.76 | 58.68 | 3.59 | 54.28 | 3.72 | |
Sorption Isotherms
Batch experimental data were validated using Langmuir, Freundlich and Tempkin isotherms, wherein their constants / correlation coefficients (R2) were computed from the corresponding plots. Table 4 shows that Langmuir constant qm is considerably higher than the KF, Freundlich constant, as far as both the systems are concerned. Free energy constant [b (L/g)] for the magnetite systems were found to be petite (0.26 and 0.17), inferring appreciable affinity of TALPMC to adsorb divalent ions. This favours the better linear fit of Langmuir model (Fig 23), suggesting monolayer adsorption and homogeneous distribution of active sites on TALPMCs’ surface. Further, it is evident from Freundlich model (Fig 24), that the process is physical in nature, since 1/n values are greater than 1. Fig 25 represents Tempkin isotherm, with greater BT and lower AT derived values. This reflects feeble applicability of this model to explain the interaction between sorbate ions and sorbent. In addition, calculated parametric values from graphical plots of all the three isotherms, reveal that TALPMC exhibited better encapsulation of Zn(II) than Pb(II).
Table 4: Isothermal Constants
| Isotherms | Parameters | Zn(II)-TALPMC | Pb(II)-TALPMC |
| Langmuir | qm (mg/g)b (L/g)R2 | 32.150.26350.9960 | 29.360.17890.9526 |
| Freundlich | KF (mg/g)1/nR2 | 5.451.640.8677 | 3.261.420.8329 |
| Tempkin | AT (L/g)bTR2 | 0.15370.500.7389 | 0.11265.3010.6425 |
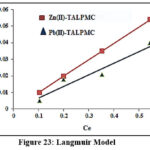 |
Figure 23: Langmuir Model |
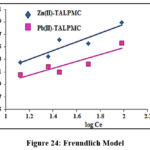 |
Figure 24: Freundlich Model |
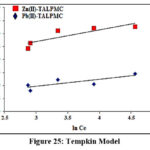 |
Figure 25: Tempkin Model |
Pseudo Kinetics
Data for pseudo-first and second order kinetic models at optimum concentrations within time frames (3 to 30 mins: 3mins) enlisted in table 5 were derived from the slopes and intercepts of the plots. Corresponding graphs (Fig 26 & 27) were drawn using the kinetic equations [log (qe–qt) vs t, t/qt vs t]. The qcal value is greater for 10mg/L Zn (II) & 15mg/L Pb(II) concentrations, ensuring the effective metal ablating nature of magnetite composite. The better fit of second order model than its first order counterpart, is sufficed by the K2 value, marginally lower than K1 and R2 value close to unity. Further, the aforesaid statement was supported by lesser SSE values obtained from respective linear plots.
Table 5: Kinetic Parameters
| Systems | Pseudo first order | Pseudo second order | |||||||
| qcal (mg/g) | K1×10-3 (min-1) | R2 | SSE | qcal (mg/g) | K2×10-3 (min-1) | R2 | SSE | ||
| Zn(II)-TALPMC | 34.52 | 13.74 | 0.8730 | 5.42 | 38.53 | 6.32 | 0. 9708 | 1.06 | |
| Pb(II)-TALPMC | 32.42 | 10.31 | 0.7706 | 7.16 | 35.65 | 5.16 | 0.8585 | 2.25 | |
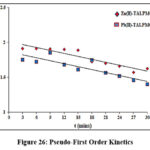 |
Figure 26: Pseudo-First Order KineticsClick here to View Figure |
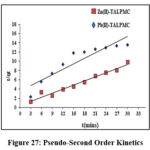 |
Figure 27: Pseudo-Second Order Kinetics |
Influence of TALPMC on Industrial Effluent
Figures 28 & 29 display the potency of TALPMC as (92%) for Zn(II) and (88%) for Pb(II) ions at 30mg and 40mg and 15 minutes contact time. Subsequent drop in the curve pattern at increased dosage is observed in both the systems, which may be due to the saturation of active sites on TALPMC. Three successive cycles were performed to evaluate the desorbing and regenerating ability of TALPMC towards the collected wastewater from oil industry as displayed in table 6. Thus, the adsorption values have minimal difference in the amounts adsorbed, disclosing the excellent reusability of material.
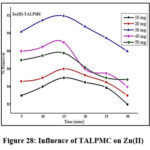 |
Figure 28: Influence of TALPMC on Zn(II) |
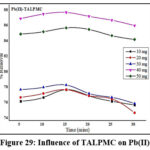 |
Figure 29: Influence of TALPMC on Pb(II) |
Table 6: Desorption / Regeneration Assay – Effluent
| Cycle 1(mg/g) | Cycle 2 (mg/g) | Cycle 3 (mg/g) | ||||
| Systems | Adsorption | Desorption | Adsorption | Desorption | Adsorption | Desorption |
| Zn(II) -TALPMC | 52.36 | 30.12 | 48.17 | 28.14 | 45.69 | 26.33 |
| Pb(II) -TALPMC | 46.32 | 33.56 | 42.54 | 35.32 | 40.96 | 38.55 |
Comparative Analysis
Attraction of Zn(II) & Pb(II) onto the pores of magnetite sorbent surface opposite to TALP is due to the super paramagnetic property exhibited by the unpaired electrons present in Fe atom(16). Moreover, TALPMC flashed up selective sorption of Zn2+ than Pb2+ ions. This can be accounted by the smaller ionic radius of Zn2+ (0.74Ǻ) than Pb2+(1.33 Ǻ), resulting in its effortless confiscation of Zn(II). This is further corroborated by isothermal and kinetic constant values.
Mechanism
In TALP, the existence of functional groups viz., OH–, HPO42- (Lewis bases) will bind with Zn(II) and Pb(II) (Lewis acids), enabling the sorption reaction(17). In addition to the presence of OH–, HPO42-, the strong electrostatic attraction between the cations (Zn2+ &Pb2+) and negatively charged part of the magnetite(18) (O42-) in TALPMC, promotes the divalent ions to get adsorbed effectively on the sorbent’s surface. Also, the formation of complexes due to the prevalence of high ratio of exchangeable Ca2+ ions enhances mechanism 17.
Conclusion
Magnetite composite was prepared by co-precipitation technique, employing Anas platyrhyncos Legs. Magnetic characteristics of Fe3O4 encapsulated Treated Anas platyrhyncos legs (TALPMC) solid matrix was validated via various analytical techniques like BET/BJH, FT-IR, SEM/EDAX, TG-DTA and XRD, to evaluate its nature during the sorption reaction. Batch experimentation exposed TALPMC as a promising sorbent for Zn(II) and Pb(II) removal from aqueous environs with maximal chelation of 97% and 95% than TALP (89% Zn2+ & 85 % Pb2+) under pre-fixed parametric conditions. Among the three isothermal models, Langmuir plot fitted well, suggesting monolayer sorption and Pseudo second order kinetics exhibited better linearity. Pilot study was conducted for the oil effluent sample, which produced satisfactory results by seizing 92% Zn(II) and 88% Pb(II) ions using TALPMC. The recorded outcomes and observations reveal that, magnetite biosorbent plays a prominent role in the sequestration of divalent metal ions both from aqueous and industrial effluent.
Acknowledgement
Authors acknowledge the DST FIST, New Delhi, India for the infrastructure provided.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
Figs 1- 4 documented in this article were captured from experimental set up carried out in the chemistry lab in PSGR Krishnammal College for Women
Figs 5a –5c microscopic images, Binocular Ultrasound microscope (OLYMPUS make, Model- CX21I), figs 9a & 9b IR spectra (Shimadzu) and fig 14 Thermogram, TG/DTA (NETZSCH JUPITOR STA 449F3) recorded in PSGR Krishnammal College for Women
Fig 6- 8 BET/BJH plots recorded in Material Analysis & Research Centre, Bangalore
Fig 15, XRD pattern (Bruker D8 Advance make) recorded in VIT, Vellore
Graphs (fig 16a – 29), computation, Origin 8.5 software
Figs 16a – 22, 25 & 26, results of Batch Optimization studies
Figs 23-25, graphs of data derived from isothermal constants
Figs 28 & 29, graphic representation of kinetic constants calculated from corresponding equations.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Behzadian, k; Kapelan, Z , Conserv. Recyc.2015, 99: 84–99.
CrossRef - Yuanan Hu, Hefa Cheng, Environ. Dev. 2013, Volume 8,57-73
CrossRef
- Deblina Dutta , Shashi Arya , Sunil Kumar, Chemosphere, 2021,Volume 285,131245.
CrossRef
- Jebin Ahmed ;Abhijeet Thakur ;Arun Goyal, , RSC, 2021
- Jessica Briffa, Emmanuel Sinagra , Renald Blundell, Heliyon , 2020
- Kosar Hikmat Hama Aziz, Fryad S. Mustafa, Khalid M. Omer et al, RSC Adv. 2023, Issues 6
- Gessesse Kebede,Tekle Tafese,Ebrahim M. Abda, M. Kamaraj, and Fassil Assefa, J. Chem. 2021.
CrossRef
- David Molden, J. Water Resour. Dev. 2020, Vol 36
CrossRef - Amer Charbaji et al, JMMCE.2021, 09(03):257-270
CrossRef - Sasireka Velusamy, Anurag Roy, Senthilarasu Sundaram, Chem. Rec. 2021
- Narendra Singh, Jinhui Li, Adv. Mat. Res. 2013, 878.
- Aliyu Haruna Sani, Amanabo Musa, , Biol. Pharm. Sci. 2021
- Nleya Y, Simate GS, Ndlovu S, Clean. Prod. 2016, 113, 17-27
CrossRef - Shagun Kainth a, Piyush Sharma, Appl. Surf. Sci. Adv. 2024,Volume 19,100562
CrossRef
- Nahum Andres Medellin-Castillo, Miguel Mauricio Aguilera-Flores& Bridinette Thiodjio Sendja , Springer, Porous. Mater: 2021, 272-314
- Attarad Ali, Hira Zafar, Muhammad Zia, Ihsan ul Haq, Abdul Rehman Phull, Joham Sarfraz Ali, and Altaf Hussain, , Sci . Appl, 2016
- Sarah Diallo-Garcia, Manel Ben Osman, Jean-Marc Krafft, Sandra Casale, Cyril Thomas, Jun Kubo, and Guylène Costentin, Phy. Chem, 2014,118 (24) 12744–12757
CrossRef - Fawzia I. El-Dib, Dalia E. Mohamed, Omnia A.A. El-Shamy, Marwa R. Mishrif, J. Pet, 2020, 1-7
Accepted on: 09 Apr 2025
Second Review by: Dr B R Venkatraman
Final Approval by: Dr. Pounraj Thanasekaran

















