Investigation of Pure and Potassium Iodide (KI) Doped PVDF/PEO Electrolyte Thin Films: An Optical Study
1Department of Physics, SVREC, Nandyal, India
2Department of Chemistry, SVREC, Nandyal, India
Corresponding Author E-mail: ramn231983@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/410226
ABSTRACT:Solution casting was employed to fabricate Polymer blend-based solid electrolyte membranes of polyvinylidene fluoride (PVDF) and polyethylene oxide (PEO) with varying concentrations of potassium iodide (KI). UV-Visible absorbance analysis was utilized to investigate their optical properties. The results indicate that altering the KI concentration modifies the optical characteristics of the films. Specifically, increasing the KI content shifts the electrolyte samples exhibit an absorption edge shifts to a reduced energy region, leading to a reduction in the energy gap. The energy gap values suggesting the formation of charge exchange complexes within the mixture upon the addition of KI to the polymer system, these complexes enhance the electrical conductivity of the system by introducing additional charges into the lattice.
KEYWORDS:Optical Absorption; PVDF; PEO based polymer electrolyte films; UV-Visible absorbance analysis
Introduction
The primary challenges facing society are the generation, delivery, and storage of energy. Ion-conducting solid polymer electrolytes are essential components of several electro-ionic systems, including transducers, solar power units, high-capacity capacitors, and rechargeable batteries [1-6]. These materials are highly valued because of their ability to support efficient ion transport. Salts can form complexes with a variety of polymer hosts—such as PEO, PEG, PPO, PVP, PMMA, and PVA—to produce ion-conducting polymer electrolytes.
Polyvinylidene fluoride (PVDF) is a polymer that merits special attention due to its charge transfer mechanism, excellent electrical properties, and ease of processing [7]. PVDF, an amorphous polymer, contains a rigid pyrrolidone group, which contributes to its high glass transition temperature (Tg) and its ability to form complexes with various inorganic salts [8]. Polyethylene oxide (PEO), on the other hand, possesses remarkable optical properties, surface resistance, non-tracking behavior, high electrical resistance, and excellent compatibility with other polymers. The significant role of PEO in stabilizing the electrode-electrolyte interface was demonstrated by Appetecchi et al. [9]. Furthermore, Iijima et al. conducted pioneering research on plasticized polymer electrolytes, identifying PEO as a suitable host material [10]. Bohnke [11] also investigated the stability and reaction rates of alkali metal electrodes in gel-derived electrolytes consisting of PEO. Considering these findings, the present work focuses on the optical investigation of polymer-based ionic composite systems consisting of polyethylene oxide (PEO) and polyvinylidene fluoride (PVDF) doped with potassium iodide (KI).
Experimental
Preparation of Films
Solid polymer electrolyte films (120 μm thick) were synthesized via a casting solution technique. This involved the preparation of pure PVDF/PEO mixtures as well as various compositions of PVDF/PEO complexed with KI salt. The samples were created using weight composition ratios of (47.5:47.5:5), (45:45:10), and (42.5:42.5:15).
The process involved dissolving PVDF, PEO, and KI salt in double-distilled water and stirring the mixture at ambient temperatures for 8–10 hours to achieve homogeneity. The uniform solution was subsequently poured onto a polypropylene plate, allowing the solvent to allow evaporation at ambient temperature. Once dried, the composite polymer membranes were carefully removed from the propylenepolymer plate and kept in a dry suction system chamber for preservation.
The Ultraviolet-visible absorption readings of the recorded spectra of the prepared samples spanned the range of 250 to 950 nm at ambient thermal state using a JASCO V-700 spectral analysis system from these readings, optical parameters, including the band edges and optical energy gaps, both direct and indirect, transitions were calculated.
Result and Discussions
The negatively charged particle shift from the peak of the outer electron band to the trough of the free electron band is predominantly accountable for the UV absorption. As shown in Fig. 1, the Ultraviolet-visible absorption readings of the PVDF/PEO mixer and PVDF/PEO/KI solid polymer electrolyte films were documented at environmental temperature in the wavelength range of 250 to 950 nm. The carbonyl group in the pure polymer blend is responsible for the shoulder-like peak observed in the spectrum of the pure PVDF/PEO film at approximately 238 nm, as shown in the figure. In the doped films, the absorption coefficient increases with higher doping levels, and the shoulder shifts toward longer wavelengths. This behaviour may be attributed to the intense communication between PVDF/PEO and KI salt, as well as an increase in carbonyl groups [12].
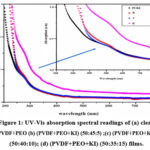 |
Figure 1: UV-Vis absorption spectral readings of (a) clean PVDF+PEO (b) (PVDF+PEO+KI) (50:45:5) ;(c) (PVDF+PEO+KI) (50:40:10); (d) (PVDF+PEO+KI) (50:35:15) films.Click here to View Figure |
Optical constants, parameters like the optical energy gap (Eg) and the location of the primary band edge are determined through Optical absorption analysis of the films. A key characteristic of the absorption edge is the rapid rise in the absorptivity factor (α) as wave-packet energy (hν) increases. The absorptivity factor α, can be ascertained from the spectral readings using the relevant equation. [Fig. 2].
Here, d represents the sample’s thickness, and A denotes the absorbance. The absorption edge is located at 3.87 eV for pure PVDF/PEO films and decreases from 3.70 eV to 3.44 eV for the doped films.
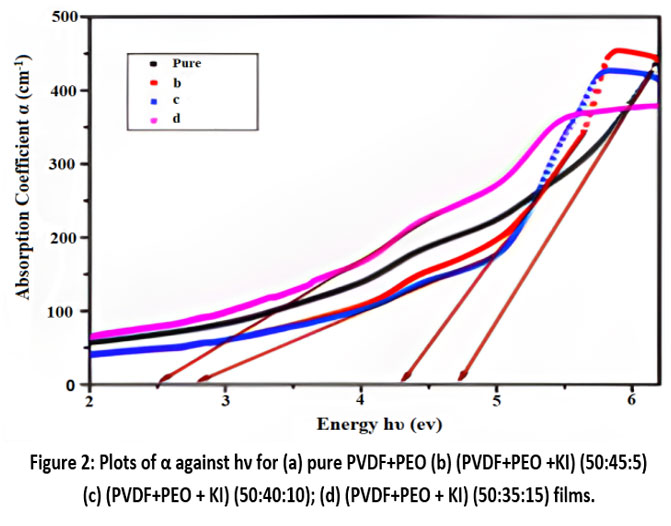 |
Figure 2: Plots of α against hν for (a) pure PVDF+PEO (b) (PVDF+PEO +KI)(50:45:5) (c) (PVDF+PEO + KI) (50:40:10); (d) (PVDF+PEO + KI) (50:35:15) films.Click here to View Figure |
The absorptivity coefficient is influenced by the energy of the incident photons in the following manner when a direct band gap is present [13, 14, and 15].
Where h is the constant of Planck’s, ν is the frequency of light, c is a constant, and Eg is the energy band gap. A linear relationship is observed between (αhν)2 and wave-packet energy (hν), as depicted in Figure 3. The insulating gap can be determined by extending the linear segment of the curve to the point of zero absorption and finding Energy-axis intercept point. The intrinsic band gap for pure PVDF/PEO membrane is 3.99eV, and the values for doped films range from 3.95 to 3.78eV (see table 1.1).
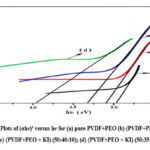 |
Figure 3: Plots of (αhν)² versus hν for (a) pure PVDF+PEO (b) (PVDF+PEO + KI) (50:45:5) (c) (PVDF+PEO + KI) (50:40:10); (d) (PVDF+PEO + KI) (50:35:15) films.Click here to View Figure |
The correlation between the absorption coefficient and photon energy is as follows for indirect transitions that need phonon assistance [15, 13, 14].
Where A and B are parameters determined by the electronic band configuration and Ep is the phonon energy connected to the transition. As illustrated in Fig. 4: Plots of (αhν)¹/² versus hν yielded the indirect energy band gaps. The indirect energy band gap for pure PVDF/PEO film is 3.89eV, whereas the values for doped films spanning from 3.81 eV to 3.59 eV.
 |
Figure 4: Graphs of (αhν)¹/² versus hν for (a) pure PVDF+PEO (b) (PVDF+PEO + KI) (50:45:5) (c) (PVDF+PEO + KI) (50:40:10); (d) (PVDF+PEO + KI) (50:35:15) films.Click here to View Figure |
The table displays the absorption threshold and direct and implicit energy insulating gap energies of the clean and KI-mixed PVDF/PEO macromolecule membranes that were obtained in this work.
Table 1: Optical energy gap values at the absorption threshold for pure and KI-doped PVDF/PEO samples…
| Concentration (Weight %) | Absorptivity edge (eV) | Optics-based band gap energy(eV) | |
| Direct | Indirect | ||
| PVDF+PEO | 5.91 | 5.84 | 5.64 |
| (PVDF+PEO + KI) (50+45+5) | 5.76 | 5.74 | 5.24 |
| (PVDF+PEO + KI) (50+40+10) | 5.61 | 5.70 | 5.20 |
| (PVDF+PEO + KI) (50+35+15) | 4.80 | 5.02 | 4.36 |
Table 1 shows that as the doping level increases, both the direct and indirect energy band gaps reduce. This decline could be imputed to the creation of blemishes within the polymer array.. These imperfections introduce confined states within the optical energy insulating gap, which intersect with the band structure, leading to a reduction in the band gap energy. This suggests that PVDF/PEO films become more semiconducting in nature as the concentration of KI dopant increases [16, 17].
Acknowledgement
The authors wish to thanks to Dr V.Raja lecturer, Physics Division, SVSSC Government Degree College, Sullurpet 524121, India, for helping in the Construction of the manuscript.
Conclusion
The study demonstrates that incorporating varying concentrations of potassium iodide (KI) into PVDF/PEO polymer blend-based solid electrolyte membranes significantly influences their optical properties. UV-Visible absorbance analysis reveals a shift in the absorption edge toward lower energy regions as KI content increases, indicating a reduction in the optical energy gap. This decrease in the energy gap suggests the formation of charge exchange complexes within the polymer matrix. These complexes contribute to enhanced electrical conductivity by introducing additional charge carriers, thereby improving the overall ionic transport within the system. The findings highlight the potential of KI-doped PVDF/PEO membranes for applications in solid-state electrolytes and optoelectronic devices.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Winie, T., Arof, A.K., Thomas, S. (Eds.) Polymer Electrolytes: Materials, Design, and Applications in Energy Storage and Conversion Technologies. Wiley-VCH, (2023).
- Liu, X., Zhang, X., Li, H. Complexation of polyethyleneimine with copper(II), nickel(II), and cobalt(II) ions: Structure and interaction mechanisms. Appl. Polym. Sci. 139 (2022) 46057.
- Yang, X., Wang, Y., Liu, F. Electrical and electrochemical properties of composite polymer electrolytes based on PEO-Sn-LiCF₃SO₃ for lithium-ion batteries. Electrochem. Energy Convers. Storage 18 (2021) 042007.
- Cuiffo, M.A., Yang, J. Metal-organic frameworks and interfacial reactions for advanced energy, environmental, and medical applications. Today Adv. 16 (2022) 100185.
- Bella, F., Lamberti, A., Sacco, A., Chiodoni, A. New electrolyte membranes for flexible dye-sensitized solar cells based on TiO₂ nanotube arrays and light-cured polymer networks. Mater. Chem. A 11 (2023) 1987-1995.
- Surana, K., Singh, P.K., Bhattacharya, B. Enhanced efficiency of dye-sensitized solar cells using reduced graphene oxide as a conductive additive. Mater. Sci.: Mater. Electron. 33 (2022) 11945-11955.
- Ranque, P., Boaretto, N., Perez-Furundarena, H., Gomez Castresana, K. Cellulose-based polysalt polymers for lithium metal polymer batteries: A green and sustainable approach. ACS Appl. Energy Mater. 7 (2024) 2001-2010.
- Nangia, R., Shukla, N.K., Sharma, A. Temperature and frequency dependence of electrical properties in PVA/PEG polymer blend electrolytes. J. 53 (2021) 739-745.
- Liao, C., Sun, X.G., Dai, S. Crosslinked gel polymer electrolytes based on polyethylene glycol methacrylate and ionic liquids for lithium-ion battery applications. Acta 426 (2022) 140876.
- Shao, D., Wang, X., Liu, H. In situ gel polymer electrolytes for high-performance quasi-solid-state lithium-ion batteries. Solid State Electrochem. 26 (2022) 1685-1692.
- Tripathi, S.K., Gupta, A., Jain, A. Electrical and electrochemical studies of magnesium ion-based gel electrolytes for energy storage applications. Solid State Ionics 383 (2023) 116187.
- Abdullah, O.G., Saber, D.R. Optical and electrical characterization of NiCl₂-doped PVA films for optoelectronic applications. Res. Bull. 149 (2023) 111750.
- Ramamohan, K., Sharma, A.K., Xiuyang, L. Structural and electrical properties of PVA/PEG polymer blends doped with NaClO₄ for electrolyte applications. Ionics 27 (2021) 531-539.
- Abdelrazek, E.M., Asnag, G.M., Gumaan, M.S. Structural, optical, and electrical properties of PEMA/PMMA blends filled with CoCl₂ and LiBr for energy storage devices. Electron. Mater. 52 (2023) 2161-2169.
- Mansingh, S., Das, K.K., Parida, K. Hydrogen evolution reactions (HERs) on MoS₂ nanosheets: Synthesis, characterization, and recent progress. Energy Fuels 8 (2024) 89-100.
- Elttayef, A.H.K., Ajeel, H.M., Kudair, A.E. Optical and structural properties of CuSn thin films for solar and photovoltaic applications. Mater. Sci.: Mater. Electron. 34 (2023) 1382-1390.
- Hamdalla, T.A., Hanafy, T.A., Bekheet, A.E. Influence of erbium ions on the optical and structural properties of polyvinyl alcohol films for photonic applications. Spectrosc. 2021 (2021) 8725129.
Accepted on: 03 Apr 2025
Second Review by: Dr. Naresh batham
Final Approval by: Dr. Ioana Stanciu