Evaluation of Phytochemical Composition, Antioxidant Activity, and Bioactive Constituents of Acacia arabica Bark Extract: A Comprehensive Analysis
1College of Pharmacy, Teerthanker Mahaveer University, Moradabad, Uttar Pradesh, India
2 School of Pharmaceutical Sciences, Shri Venkateshwara University, Rajabpur, NH-24, Venkateshwara Nagar, Gajraula, Uttar Pradesh, India
Corresponding Author E-mail:ashwanigupta143@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/410218
ABSTRACT:Acacia arabica is historically known to have medicinal properties in bark. In this work, its phytochemical components, antioxidant efficacy and bioactive substances was assessed for investigating the possibility of using it as natural antioxidant. Soxhlet extraction was used to obtain a hydroalcoholic extract of Acacia arabica bark. Principal ingredients were identified by phytochemical screening and TPC and TFC of a crude extract was determined. DPPH, ABTS, FRAP tests have been used to determine the antioxidant potential of the extract. The extract was analysed with LC-MS to identify bioactive compounds. Phenols, tannins, flavonoids and alkaloids were reported to be present in phytochemical investigation. Antioxidant activity of the extract was very high with IC₅₀ values of 6.44 µg/mL (DPPH), 4.38 µg/mL (ABTS) and 0.86 µg/mL (FRAP). They had a quantified total phenolic content (TPC) of 940 and a total flavonoid content (TFC) of 94 mg quercetin equivalent per gram. Bioactive chemicals as spermine, isocitric acid lactone, and 9-deenoic acid, known for their antioxidant activity, were detected by LC-MS. Considering the past use of Acacia arabica as a natural antioxidant source, the significant antioxidant properties of its bark extract towards its phenolic and flavonoid content are validated. These findings enabled subsequent studies on the applicability of this in functional foods or nutraceutical formulations.
KEYWORDS:Acacia arabica; Antioxidant activity; Flavonoid content; LC-MS; Phenolic content
Introduction
Reactive oxygen species (ROS) and free radicals are exceedingly reactive chemicals which are formed during the metabolism of the body or at exposure to external stressors due to ultraviolet ray, pollution and environmental toxins. ROS are needed for immune defense and cell signaling, but too many of them can upset the balance of cells, which can cause oxidative stress that hurts DNA, proteins, and fats. Oxidative stress is a crucial element in the development of various chronic diseases, such as cancer, diabetes, and cardiovascular ailments. 1
The body’s antioxidant defense system combats ROS through both enzymatic components, such as superoxide dismutase and catalase, and non-enzymatic components, including glutathione, polyunsaturated fatty acids, and vitamins C and E. Under stress situations, this system may prove inadequate, prompting the need for exogenous antioxidants, especially from natural sources, owing to their therapeutic efficacy and safety. Plant-derived compounds, such as phenolics, flavonoids, tannins, and alkaloids, have significant antioxidant properties by eliminating reactive oxygen species, chelating metal ions, and enhancing the body’s intrinsic defenses. 2 3
Acacia arabica, also referred to as Indian gum arabic, is widely recognized in traditional medicine for its anti-inflammatory, antibacterial, and wound-healing attributes 4. It is abundant in phenolic chemicals and flavonoids, which enhance its pharmacological efficacy.5 Despite the prolonged use of Acacia arabica, its antioxidant activity has not been rigorously evaluated, and the bioactive chemicals responsible for these effects remain unidentified. This work aims to investigate the phytochemical composition, antioxidant activity, and bioactive constituents of Acacia arabica bark extract to address this research gap.
Materials and Methods
Chemicals
All the reagents used in this research were supplied by CDH Pvt. Ltd in Delhi, Merk Ltd. in Mumbai, Qualigens in Mumbai, and HiMedia Ltd. in Mumbai, and all were of LR or AR grade.
Plant Collection and Authentication
Plant was selected after reviewing the literature, gathering phytoconstituent-based activity data from conventional publications, the internet, and the traditional medical system. The plant’s bark was collected from the roadside of a village in Sambhal, District-Moradabad, Uttar Pradesh, India, in December 2023. Plant for authentication was successfully done, and the authentication letter was received. Authentication was done by Dr. Alok Shrivastava from Plant science department, M.J.P. Rohilkhand University, Bareilly, U.P., India with authentication number – MJPRU/PS/2023/0578.
Extraction technique
The powdered bark was subjected to sequential extraction using a soxhlet apparatus. Initially, defatting was conducted using petroleum ether (60–80°C), succeeded by extraction with 70% ethanol. Whatman No. 1 filter paper was employed for filtration of the extracts. This filtrate was concentrated to a minimal volume using a rotary evaporator at 40°C under reduced pressure (70-100 mBar). Subsequent to each extraction phase, the marc was air-dried at room temperature before to advancing to the next solvent. The yield of hydroalcoholic extract was calculated. The hydroalcoholic extract of Acacia arabica bark was further analyzed for phytochemical constituents and evaluated for antioxidant and to optimize its production 6.
Phytochemical screening
Standard qualitative methods were employed to detect the presence of significant phytocompounds, including flavonoids, terpenoids, tannins, alkaloids, steroids, saponins, and phenolics in the Acacia arabica bark extract 7,8
Estimation of total phenolic content
The evaluation of total phenols in extract was conducted utilizing the Folin-Ciocalteu method. A standard gallic acid solution was prepared by dissolving 25 milligrams of gallic acid in distilled water. The extract was dissolved in various quantities, and flasks were filled with each concentration. The flasks were subsequently calibrated to 25 milliliters, and a UV spectrophotometer (UV-Shimadzu 1900i) was employed to record the absorbance at 750 nm after 90 min of incubation 9
Estimation of total flavonoids content
Using the aluminium chloride colorimetric method, the complete amount of flavonoid was assessed. By dissolving 25 mg of quercetin in methanol, a standard solution with a concentration of 1 mg/mL was prepared. An additional 100 mg/mL solution was produced by diluting the original. It was then mixed with water, potassium acetate, methanol, and aluminium chloride. Various amounts of the flavonoid quercetin were used to plot a standard curve 10
Thin layer chromatography (TLC)
TLC plates (a 0.2 mm thick sheet of pre-coated aluminium silica gel) were taken using the following eluents: mixes of methanol and chloroform (7:3 and 9:1), ethyl acetate and chloroform (6:4), and chloroform with ethyl acetate and formic acid (5:4:1). A pin-drawn line indicates where the spots were inserted, working with a capillary tube, one centimeter from the bottom. After the substance on the plate dried, it was quickly covered, and they placed it into the chromatographic tank. The plate was taken out, labeled, and dried after the solvent had reached the very top. Applying iodine vapor and spraying spotting reagent at UV wavelengths of 254 and 365 nm allowed for the determination of the number of spots. 11
Liquid Chromatography-Mass Spectroscopy (LC-MS)
The sample extract was subjected to LC-MS analysis in a Waters Xevo TQD triple quadrupole mass spectrometric platform coupled with a Waters Acquity H-class UPLC/PDA system (CSIR-CDRI, Lucknow). The two functional tests, ES+ and ES-, were conducted over a mass range of 150 to 2000 nm for approximately 40 minutes. The column temperature was set at 35°C. The pre-injection wash length was 0 seconds, whereas the post-injection wash period was 6 seconds, with an injection volume of 2 μL. The UPLC eLambda operated at 800 nm served as the detector. The device detected and reported the phytoconstituents after comparing them with those in the linked NIST computer library. 11
Antioxidant ASSAY
2,2-diphenyl-1-picrylhydrazyl (DPPH) scavenging
On a 96-well plate, 5 μl of a different test stock was mixed with 0.1 ml of a 0.1 mM DPPH solution. Three copies of the reaction were made, one with five microliters of a chemical at varying concentrations and the other with 0.2 milliliters of DMSO/methanol. The reagent-free wells (DPPH) were designated as blank, and treatment-free wells were designated as control. The dish was incubated in the dark for 30 minutes. At the end of the incubation, the decolorization was measured in a microplate reader (iMark, BioRad) at 517 nm. Serving as the control was a reaction mixture with 20 μl of deionized water 12. The display showed the percentage inhibition of the scavenging activity relative to the control. GraphPad Prism 6 software was used to determine the IC50. A graph was prepared between the X-axis (sample concentration) and the Y-axis (% inhibition with respect to control. Scavenging activity (percentage) was estimated by the below mentioned equation 1-
2,2′-Azinobis-(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) scavenging
Ammonium Persulfate (APS) at 2.45 mM and 7 mM solutions of ABTS were combined to generate ABTS radicals. The ABTS free radical reagent was subsequently diluted by a factor of 100. A 96-well plate with 200 µl of ABTS free radical reagent was incubated at ambient temperature for 10 minutes in the absence of light. Samples and 10 µl of a distinct stock of the reference material (ascorbic acid) were included. The untreated wells were assigned as the control group. Assess the absorbance of the decolorization at 750 nm utilizing an iMark microplate reader (BioRad) following incubation. The results for the negative control were provided 13. The IC50 was determined using GraphPad Prism version 9.5.1 software. A graph was constructed with the X-axis representing sample concentration and the Y-axis indicating % inhibition relative to the control 14–16. Scavenging activity was calculated by equation 1.
Ferric reducing-antioxidant power (FRAP) scavenging assay
A mixture of 0.04 ml of 0.2 M sodium phosphate buffer, pH 6.6 and 1% potassium ferricyanide [K3Fe (CN)6] was mixed. After giving the reaction mixture a thorough vortex, it was incubated for 20 minutes at 50°C. The control wells were those that received no treatment. 0.5 milliliters of trichloroacetic acid (10%) was added to the mixture at the conclusion of the incubation. After that, 50μl of 0.1% ferric chloride and 50μl of deionized water were added. Using a microplate reader (iMark, BioRad), the colored solution was read at 700 nm in comparison to the blank 17. The program Graph Pad Prism 6 was used to determine the IC50. Scavenging activity was calculated by equation 1.
Results
Percentage yield of extract
The yield of hydroalcoholic extract of Acacia arabica bark was found to be 36%.
Phytochemical screening of the extract
Phenols, tannins, flavonoids, and alkaloids were identified in the extracts through preliminary phytochemical screening, as shown in Table 1.
Table 1: Results of phytochemical screening of Acacia arabica bark
| S. No. | Phytoconstituents | A.arabica extract | Name of the test |
| 1 | Phenolic and Tannins | + | Ferric chloride test |
| + | Potassium dichromate test | ||
| – | Lead acetate test | ||
| – | Acetic acid test | ||
| + | Dilute HNO3 test | ||
| 2 | Flavonoids | – | Shinoda test |
| – | Lead acetate test | ||
| – | NaOH test | ||
| 3 | Alkaloids | + | Dragendroff’s test |
| + | Mayer’s test |
Total phenolic and flavonoid estimation
The phenolic amount in the extract of Acacia arabica bark was calculated to be 940 mg/Gallic acid equivalent per gram and the amount of flavonoid was found to be 94 mg/Quercetin equivalent per gram. All the values are stated as Mean ± Standard deviation (SD). Values are mean of three replicates.
Qualitative phytochemistry: Thin layer chromatography
TLC was performed based on the solvent and were presented in Table 2.
Table 2: TLC analysis of Acacia arabica extract
| S. NO. | Solvent used | Ratio | No. of spot | Colour of spot | Rf value |
| 1. | Chloroform : Methanol | 7:3 | 1 | Light brown | 0.91 |
| 2. | Chloroform : Ethyl acetate | 6:4 | 1 | Light brown | 0.44 |
| 3. | Chloroform : Ethyle acetate : Formic acid | 5:4:1 | 2 | Light brown | 0.64 and 0.94 |
| 4. | Chloroform : Methanol | 9:1 | 2 | Light brown | 0.96 and 0.90 |
LC-MS of Acacia arabica extract
The probable identification of compound present in bark extract were presented in Table 3 and Figure 1 for ES (+) and for ES (-) represented in Table 4 and Fig. 2. The highest relative concentration of ions at m/z, 202.22, which were thought to represent spermine. The other substances found in bark extracts were Isocitric acid lactone, Ethylpiperidine-4-carboxylate, 4-t-Butylbenzeneamine, 9-Decenoic acid, 1-Hexanamine, N-hexyl-N-nitroso-, N-Methyldodecanamide, Diethyl decylphosphonate, N-Methyldodecanamide, Barban, 9-Heptadecanol, Nonadecane, 3-methyl-, 1,3,5-Trinitrobenzene and Heptadecane, 3-methyl-.
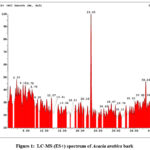 |
Figure 1: LC-MS (ES+) spectrum of Acacia arabica barkClick here to View Figure |
Table 3: LC-MS (ES+) analysis of bark extracts of Acacia arabica
| Sample | MS (m/z) | Tentative identification |
| Bark | 402.19 | Isocitric acid lactone |
| 202.22 | Spermine | |
| 157.11 | Ethylpiperidine-4-carboxylate | |
| 149.12 | 4-t-Butylbenzeneamine | |
| 223.19 | 9-Decenoic acid | |
| 214.2 | 1-Hexanamine, N-hexyl-N-nitroso- | |
| 213.21 | N-Methyldodecanamide | |
| 278.2 | Diethyl decylphosphonate | |
| 213.21 | N-Methyldodecanamide | |
| 257 | Barban |
 |
Figure 2: LC-MS (ES-) spectrum of Acacia arabica barkClick here to View Figure |
Table 4: LC-MS (ES-) analysis of bark extracts of Acacia arabica
| Sample | MS (m/z) | Tentative identification |
| Bark | 256.28 | 9-Heptadecanol |
| 282.33 | Nonadecane, 3-methyl- | |
| 213 | 1,3,5-Trinitrobenzene | |
| 254.3 | Heptadecane, 3-methyl- |
DPPH radical scavenging activity
The IC₅₀ value was determined for each substance. The significantly lower IC₅₀ value of 6.442 ± 0.17 µg/mL for the Acacia arabica extract indicates its robust antioxidant activity in comparison to ascorbic acid’s IC₅₀ value of 22.05 ± 0.026 µg/mL. These results suggest that Acacia arabica may serve as a more potent natural antioxidant than ascorbic acid, demonstrating a superior capacity to scavenge free radicals. The comparative percentage scavenging activity of both ascorbic acid and Acacia arabica bark extract was illustrated in Fig. 3.
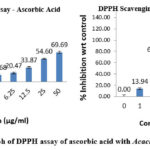 |
Figure 3: Comparison graph of DPPH assay of ascorbic acid with Acacia arabica bark extractClick here to View Figure |
ABTS radical scavenging activity
The IC₅₀ value of extract, representing the concentration required to block 50% of ABTS radicals, was determined to be 4.38 ± 0.089 µg/mL and for ascorbic acid it was found to be 8.414 ± 0.029. The bark extract exhibits significant antioxidant activity, evidenced by its relatively low IC₅₀ value, indicating its efficacy in neutralizing free radicals. The comparative percentage scavenging activity of both ascorbic acid and Acacia arabica bark extract was presented in Fig. 4.
 |
Figure 4: Comparison graph of ABTS assay of Ascorbic acid with Acacia arabica bark extractClick here to View Figure |
FRAP radical scavenging activity
The IC₅₀ for ferric ion reduction in extract was established at 0.8602 ± 1.35 µg/mL while ascorbic acid was observed to be 2.39 ± 0.011 µg/mL. This extract demonstrates remarkable ferric ion-reducing action, evidenced by its low IC₅₀ value, which underscores its significant antioxidant potential. The comparative percentage scavenging activity of both ascorbic acid and Acacia arabica bark extract was presented in Fig. 5.
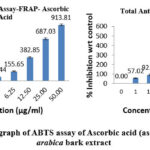 |
Figure 5: Comparison graph of ABTS assay of Ascorbic acid (as standard) with Acacia arabica bark extract.Click here to View Figure |
Discussion
Soxhlet extraction is a process that involves boiling a solid substance to extract soluble components, with polar solvents being better for obtaining the desired compounds. Acacia arabica bark extract, an essential component of natural health products, has high antioxidant qualities due to its phenolic components, which may have antibacterial and anti-inflammatory. Phytochemical screening is conducted to identify and describe the bioactive substances contained in the extract, such as tannins, flavonoids, saponin, phenols, and alkaloids which possess anti-inflammatory, antibacterial, antioxidant and anti-diabetic activity 18
The total phenolic content (TPC) of plant extracts is a significant parameter, because phenolic chemicals are important for maintaining health and preventing illness. The TPC value indicates the presence of phenolic chemicals, which are essential for protecting against inflammation, lowering oxidative stress, and neutralizing free radicals. The high TPC value in the bark extract supports its potential for creating dietary supplements or medications targeting inflammation and disorders linked to oxidative stress 19. Using a colorimetric technique, the quantity of flavonoids in plant extracts is measured by total flavonoid content, or TFC. As shown, the corresponding quantity of rutin or quercetin of material, it is essential for evaluating the antioxidant and medicinal qualities of the extract
TLC, or thin layer chromatography, is an essential analytical method for separating and identifying bioactive compounds in the extract. It can create an extract’s fingerprint profile, which is essential for control and standardization of herbal medication quality. Key components were found by the use of LC-MS such as phenolics, flavonoids, and alkaloids, validating the extract’s potential therapeutic applications and upholding traditional medical usage 20
The DPPH assay evaluates plant extracts’ antioxidant potential by assessing their ability to neutralize free radicals. The bark extract of Acacia arabica has a low IC50 value and significant antioxidant activity, perhaps as a result of the flavonoids, phenolic chemicals, and other bioactive components it contains. The ABTS test measures the extract’s ability to combat free radicals ABTS•+, demonstrating its potential for medicinal and prophylactic applications. Ferric-to-ferrous ion conversion of the extract is measured using the FRAP test, indicating its strong antioxidant properties. The extract’s potential as a preventive measure against oxidative damage is demonstrated by its capacity to convert Fe3+ to Fe2+.12
Conclusion
The findings of this study attributed to robust antioxidant properties of Acacia arabica bark extract and the existence of bioactive phytochemicals such as phenols, flavonoids, and tannins. The extract’s primary bioactive components from LC-MS were found to be -spermine, isotric acid lactone, ethyl piperidine-4-carboxylate, and 9-decenoic acid etc. These compounds demonstrated significant scavenging free radicals through DPPH, ABTS, and FRAP assays. The study’s identified mechanisms of apoptotic inhibitory, inflammatory reducing, and antioxidant effects may be significantly influenced by these substances.
Acknowledgement
We express our profound gratitude to the Department of Pharmacy, Teerthanker Mahaveer University, Moradabad, Uttar Pradesh, India, for furnishing laboratory facilities and endorsing this work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Rudrapal, M.; Khairnar, S. J.; Khan, J.; Dukhyil, A. Bin; Ansari, M. A.; Alomary, M. N.; Alshabrmi, F. M.; Palai, S.; Deb, P. K.; Devi, R. Dietary Polyphenols and Their Role in Oxidative Stress-Induced Human Diseases: Insights Into Protective Effects, Antioxidant Potentials and Mechanism(s) of Action. Front Pharmacol 2022, 13. https://doi.org/10.3389/FPHAR.2022.806470.
CrossRef - Hasibuan, P. A. Z.; Simanjuntak, Y.; Hey-Hawkins, E.; Lubis, M. F.; Rohani, A. S.; Park, M. N.; Kim, B.; Syahputra, R. A. Unlocking the Potential of Flavonoids: Natural Solutions in the Fight against Colon Cancer. Biomed Pharmacother 2024, 176. https://doi.org/10.1016/J.BIOPHA.2024.116827.
CrossRef - Sun, W.; Shahrajabian, M. H. Therapeutic Potential of Phenolic Compounds in Medicinal Plants—Natural Health Products for Human Health. Molecules 2023, Vol. 28, Page 1845 2023, 28 (4), 1845. https://doi.org/10.3390/MOLECULES28041845.
CrossRef - Patwekar, M.; Patwekar, F.; Mezni, A.; Sanaullah, S.; Fatema, S. R.; Almas, U.; Ahmad, I.; Tirth, V.; Mallick, J. Assessment of Antioxidative and Alpha-Amylase Potential of Polyherbal Extract. Evid Based Complement Alternat Med 2022, 2022. https://doi.org/10.1155/2022/7153526.
CrossRef - Ansari, P.; Islam, S. S.; Akther, S.; Khan, J. T.; Shihab, J. A.; Abdel-Wahab, Y. H. A. Insulin Secretory Actions of Ethanolic Extract of Acacia Arabica Bark in High Fat-Fed Diet-Induced Obese Type 2 Diabetic Rats. Biosci Rep 2023, 43 (5). https://doi.org/10.1042/BSR20230329.
CrossRef - Ansari, P.; Flatt, P. R.; Harriott, P.; Hannan, J. M. A.; Abdel-Wahab, Y. H. A. Identification of Multiple Pancreatic and Extra-Pancreatic Pathways Underlying the Glucose-Lowering Actions of Acacia Arabica Bark in Type-2 Diabetes and Isolation of Active Phytoconstituents. Plants (Basel) 2021, 10 (6). https://doi.org/10.3390/PLANTS10061190.
CrossRef - Faujdar, S.; Sati, B.; Sharma, S.; Pathak, A. K.; Paliwal, S. K. Phytochemical Evaluation and Anti-Hemorrhoidal Activity of Bark of Acacia Ferruginea DC. J Tradit Complement Med 2018, 9 (2), 85–89. https://doi.org/10.1016/J.JTCME.2018.02.003.
CrossRef - Dubale, S.; Kebebe, D.; Zeynudin, A.; Abdissa, N.; Suleman, S. Phytochemical Screening and Antimicrobial Activity Evaluation of Selected Medicinal Plants in Ethiopia. J Exp Pharmacol 2023, 15, 51–62. https://doi.org/10.2147/JEP.S379805.
CrossRef - Pérez, M.; Dominguez-López, I.; Lamuela-Raventós, R. M. The Chemistry Behind the Folin-Ciocalteu Method for the Estimation of (Poly)Phenol Content in Food: Total Phenolic Intake in a Mediterranean Dietary Pattern. J Agric Food Chem 2023, 71 (46), 17543–17553. https://doi.org/10.1021/ACS.JAFC.3C04022.
CrossRef - Dibacto, R. E. K.; Tchuente, B. R. T.; Nguedjo, M. W.; Tientcheu, Y. M. T.; Nyobe, E. C.; Edoun, F. L. E.; Kamini, M. F. G.; Dibanda, R. F.; Medoua, G. N. Total Polyphenol and Flavonoid Content and Antioxidant Capacity of Some Varieties of Persea Americana Peels Consumed in Cameroon. ScientificWorldJournal 2021, 2021. https://doi.org/10.1155/2021/8882594.
CrossRef - Abdel‐hamed, A. R.; Mehanna, E. T.; Hazem, R. M.; Badr, J. M.; Abo‐elmatty, D. M.; Abdel‐kader, M. S.; Goda, M. S. Plicosepalus Acacia Extract and Its Major Constituents, Methyl Gallate and Quercetin, Potentiate Therapeutic Angiogenesis in Diabetic Hind Limb Ischemia: HPTLC Quantification and LC-MS/MS Metabolic Profiling. Antioxidants (Basel) 2021, 10 (11). https://doi.org/10.3390/ANTIOX10111701.
CrossRef - Rumpf, J.; Burger, R.; Schulze, M. Statistical Evaluation of DPPH, ABTS, FRAP, and Folin-Ciocalteu Assays to Assess the Antioxidant Capacity of Lignins. Int J Biol Macromol 2023, 233. https://doi.org/10.1016/J.IJBIOMAC.2023.123470.
CrossRef - Ilyasov, I. R.; Beloborodov, V. L.; Selivanova, I. A.; Terekhov, R. P. ABTS/PP Decolorization Assay of Antioxidant Capacity Reaction Pathways. Int J Mol Sci 2020, 21 (3). https://doi.org/10.3390/IJMS21031131.
CrossRef - Gupta, R.; Sharma, M.; Lakshmy, R.; Prabhakaran, D.; Reddy, K. S. Improved Method of Total Antioxidant Assay. IJBB Vol.46(1) [February 2009] 2009, 46, 126–129.
- Kambayashi, Y.; Binh, N. T.; Asakura, H. W.; Hibino, Y.; Hitomi, Y.; Nakamura, H.; Ogino, K. Efficient Assay for Total Antioxidant Capacity in Human Plasma Using a 96-Well Microplate. J Clin Biochem Nutr 2009, 44 (1), 46–51. https://doi.org/10.3164/JCBN.08-162.
CrossRef - Cao, G.; Prior, R. L. Comparison of Different Analytical Methods for Assessing Total Antioxidant Capacity of Human Serum. Clin Chem 1998, 44 (6), 1309–1315. https://doi.org/10.1093/CLINCHEM/44.6.1309.
CrossRef - Choromańska, B.; Myśliwiec, P.; Kozłowski, T.; Łukaszewicz, J.; Vasilyevich, H. P.; Dadan, J.; Zalewska, A.; Maciejczyk, M. Antioxidant and Antiradical Activities Depend on Adrenal Tumor Type. Front Endocrinol (Lausanne) 2022, 13. https://doi.org/10.3389/FENDO.2022.1011043.
CrossRef - Nuralın, L.; Gürü, M.; Çete, S. Extraction and Quantification of Some Valuable Flavonoids from Pinecone of Pinus Brutia via Soxhlet and Supercritical CO2 Extraction: A Comparison Study. Chem Zvesti 2021, 75 (10), 5363–5373. https://doi.org/10.1007/S11696-021-01644-5.
CrossRef - Alide, T.; Wangila, P.; Kiprop, A. Effect of Cooking Temperature and Time on Total Phenolic Content, Total Flavonoid Content and Total in Vitro Antioxidant Activity of Garlic. BMC Res Notes 2020, 13 (1). https://doi.org/10.1186/S13104-020-05404-8.
CrossRef - Kašpar, M.; Bajer, T.; Bajerová, P.; Česla, P. Comparison of Phenolic Profile of Balsamic Vinegars Determined Using Liquid and Gas Chromatography Coupled with Mass Spectrometry. Molecules 2022, 27 (4). https://doi.org/10.3390/MOLECULES27041356.
CrossRef
Abbreviations
SOD-Superoxide dismutase; ROS-Reactive oxygenase; GSHPx-Glutathione peroxidase; GSH-Glutathione; DILI-Drug Induced Liver Injury; ALF-Acute liver failure; IDILI-Intrinsic drug induced liver injury; APAP-Acetaminophen; ADH-Alcohol dehydrogenase; NAD+-Nicotinamide adenine dinucleotide; NAPQI-N-acetyl-p-benzoquinone imine; CYP2E1-Cytochrome P450 2E1; KCs Kupfer cells; NF-kB-Nuclear factor kappa B; DPPH-2,2-diphenyl-1picrylhydrazyl; ABTS-2,2-azino-bls (3-ethylbenzothiazoline-6-sulfonic acid); FRAP-Ferric Reducing Antioxidant Power.
Accepted on: 27 Mar 2025
Second Review by: Dr. Shouvik Kumar Nandy
Final Approval by: Dr. Dinesh Chand Sharma