Antimicrobial, Pharmacokinetic and Semi-empirical Study of Some Schiff Base Metal Complexes
1Department of Chemistry-Baba Mastnath University -Rohtak
2Department of Chemistry-RPS Degree College, Mahendergarh
Corresponding Author E-mail:Arpnakalonia.bimpat@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/410234
ABSTRACT:The present piece of research work comprises of synthesis and characterisation of Schiff base ligand and its metal complexes with CoII, NiII, CuII and ZnII in their chlorides form. The ligand is derived from phthaldehyde and 2-amino-4-chlorobenzenethiol, a thiol based amine. IR, UV-visible, elemental analysis, magnetic moment, and EPR are used to characterize the compounds. The Agar Well diffusion method was used to screen each complex for antimicrobial qualities. Additionally, ADME studies were conducted to assess their pharmacokinetic behaviour. All metal complexes have been projected to have an octahedral geometry, and the ligand behaves like a tetradentate ligand. The newly synthesized complex has been found to have good antimicrobial properties. Pharmaco-kinetic behaviour reveal about their potential biological application. Semi-empirical studies were performed so obtained the energy minimised geometry of the ligand and the complex.
KEYWORDS:Antibacterial; Metal Complex; Semi-empirical; Schiff Base
Introduction
It is urgent to find new active molecules against new targets in order to combat the worrying issue of microbial resistance to antibiotics. Wild-growing material continues to be the source of many crude medicines used in medical formulations. However, the lifespan of the material supply has been decreased by plant-based medications. The business is always looking for cheaper and more powerful raw resources.1 The development of several features, including steric, electronic, and biological potential, is dependent on the choice of appropriate amines and substituted aromatic carbonyls for the synthesis of Schiff bases.2-5 A vast collection of Schiff bases is widely used to produce transition metal complexes with pharmacological, electrical, magnetic, and structural characteristics.6-8 Due to their versatility, chemists frequently use Schiff bases in processes like cycloaddition and nucleophilic addition with organometallic reagents.9 Schiff bases can be utilized for enzyme immobilization and also employed to overcome drug resistance in cancer.10 Due to conjugation, Schiff bases made from primary aromatic amine and aromatic aldehydes are more stable and are less likely to polymerize than these bases made from primary aromatic amine and aliphatic aldehydes, which are less stable and more likely to do so 11. Although, the Schiff base ligand and its metal complexes have a variety of applications, many more still need to be explored.12
Experimental section
Materials and methods
In the resent research work, chemicals such as 2-amino-4chlorobenzenethiol and phthaldehyde were procured from Sigma -Aldrich. Using a Perkin Elmer BX II spectrophotometer, the FT-IR spectra of all compounds were captured between 4000 and 400 cm-1. Using a Bruker A 300-9.5/12/ S/W spectrophotometer, the EPR spectra of the Cu2+ complex was captured. Mass Spectra of all sample were recorded using Mass Spectrometry SLI EX Triple TOF 5600&5600+/ SCIEX, All compounds UV-Visible spectra were recorded between the wavelengths of 200 and 1000 nm. The disc diffusion method was used to evaluate the synthetic substance’s in vitro antimicrobial efficacies.
Synthesis of Schiff base ligand
For the preparation of ligand we have adopted the method of condensation reaction. For this we have taken 10 mmol (1.34g) of phthaldehyde and dissolved it in methanol. Then drop by drop addition of amine species i.e. 2-amino-4-chlorobenzenethiol 20 mmol (3.19g) was carried out. After thar reaction mixture was refluxed for 3 hrs and light orange coloured mixture was obtained. The mixture was kept for overnight cooling at room temperature and orange colored precipitates were obtained as product. The completion of the reaction was analysed by single spot on TLC. Scheme 1.
Synthesis of Metal complexes
As the ligand found good solubility in methanol, so the methanolic solution of ligand(5 mmol) and metal salts( 5mmol) were allowed to react by refluxing for 4 hrs. Various coloured precipitates were obtained as products. Washings were by given by various organic solvents so that unreacted reaction gets removed out.
Antimicrobial studies
The well-known disc diffusion method was used for the interpretation of antibacterial and antifungal activity of the complexes. The bacterial strain used are E. coli and B. subtilis, whereas the fungal strains used are C. albicans and A. niger. The DMSO was taken as negative control. The positive control in case of antibacterial study was Ciprofloxacin and Amphotericin -B. The MIC values in µg/ml were calculated.
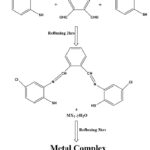 |
Scheme 1: Synthetic Pathway of the ligand and ComplexClick here to View Scheme |
ADME Studies
Swiss ADME prediction software, which is accessible online, was used to conduct in silico ADMET tests of the ligand and metal complexes. Calculating the manufactured complex’s different features, such as adsorption, distribution, metabolism, excretion, and toxicity, can reveal how similar it is to the medications.13-15 Because of its great importance in the field of drug research, the Lipinski rule of five was taken into consideration. Another term for Lipinski’s rule of five is Pfizer’s rule of five, or simply the rule of five (RO5). If a chemical molecule with a certain pharmacological or biological activity possesses both chemical and physical features that would make it a likely oral active medication in humans, then this is a general guideline to assess or clarify the drug-likeness. Christopher A. Lipinski developed the rule in 1997 after seeing that the majority of medications taken orally are tiny, somewhat lipophilic compounds.
Result and Discussion
Schiff base ligand found good solubility in methanol whereas its metal complexes are possessing good solubility in the DMSO. The lower value of molar conductance 20-35 Ω cm2 mol-1 direct towards their non-electrolytic nature.
IR Spectra
Assistance of Infra-red spectroscopy has been taken in order to get an insight into the condensation reaction between an amine and carbonyl species. (Figure 1 & 2) In the IR spectrum of the ligand there is presence of intense peak at near 2400 cm-1 can be attributed to environmental CO2. In the spectrum of the ligand we got an medium intensity peak 1630 cm-1 that corresponds to the condensation product (H-C=N) of amine and carbonyl species. 16 Peak for the thiol (-SH) group is present at 2450 cm-1 merged with the peak of environmental CO2 . In case of metal complexes the frequency of (H-C=N) group is slightly drifted towards lower value that shows coordination of metal ion with the synthesised ligand. In case of metal complex peak due to -SH group is also diminished that points towards formation of bond between sulphur and metal ion.17
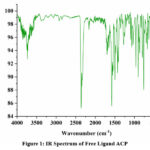 |
Figure 1: IR Spectrum of Free Ligand ACPClick here to View Figure |
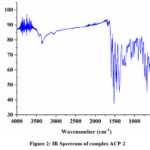 |
Figure 2: IR Spectrum of complex ACP-2Click here to View Figure |
UV Spectra
Electronic spectra of the newly synthesised ligand and its metal complexes were recorded in DMSO in the wavelength range of 250 nm to 1000nm. In the spectrum of free ligand we got an absorption band nearly at 350 nm that may be attributed to π→π* and n→π* transition of the ligand. In case of metal complexes, we got an additional peak for the d-d transition nearly 670 nm in case of ACP-3, 780 nm in case of ACP-2 and 540 nm in case of ACP -1.18 The obtained data is in good consistency with the octahedral geometry of the complex. (Figure 3 &4).
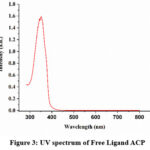 |
Figure 3: UV spectrum of Free Ligand ACPClick here to View Figure |
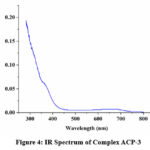 |
Figure 4: IR Spectrum of Complex ACP-3Click here to View Figure |
ESI-MS
For getting the information regarding the monomeric or polymeric behaviour of the complex mass spectrometric data was taken. The molecular ion peak for the ligand is present at m/z value of 417.83 which is in good consistency with the molecular weight of the compound. For rest of the complex molecular ion peak is present at 510.37, 510.13, 514.96 & 516.99.
EPR
Metal complexes having unpaired electrons can be interpreted by taking assistance of EPR spectral studies in order to learn intensive study of complex structure. Room temperature EPR spectra of Cu2+ complex was recorded that consists of an isotropic peak and we did not get any hyperfine splitting. From the spectrum the calculated giso value is 2.019 which indicating the presence of distorted octahedral geometry of the complex.19
Elemental Analysis and physico-analytical data
Ligand (ACP)
Yield: 76 %, Mol. Wt. 417.37; Anal. Found: C, 57.43; H, 3.35; N, 6.70; % Calc.: C, 417.31; H, 3.38; N, 6.71; . Color, Yellowish Orange.
ACP1 complex
Yield: 64 %, Mol. Wt. 510.31; Anal. Found: C, 470.1; H, 3.10; N, 5.44; M, 11.49 % Calc.: C, 47.07; H, 3.16; N, 5.49; M, 11.55. Color, Greyish ; Molar conductivity (Ω-1 mol-1cm2) in DMSO 14. Magnetic moment μ eff (BM): 3.76.
ACP2 complex
Yield: 69 %, Mol. Wt. 510.07; Anal. Found: C, 47.08; H, 3.12; N, 5.45; M, 11.49 % Calc.: C, 47.10; H, 3.16; N, 5.49; M, 11.51. Color, yellowish green; Molar conductivity (Ω-1 mol-1cm2) in DMSO 14. Magnetic moment μ eff (BM): 3.05.
ACP3 Complex
Yield: 68 %, Mol. Wt. 514.93; Anal. Found: C, 46.55; H, 3.10; N, 5.42; M, 12.30 % Calc.: C, 46.65; H, 3.13; N, 5.44; M, 12.34. Color, Yellow; Molar conductivity (Ω-1 mol-1cm2) in DMSO 14. Magnetic moment μ eff (BM): 1.79.
ACP4 Complex
Yield: 70 %, Mol. Wt. 516.76; Anal. Found: C, 46.44; H, 3.10; N, 5.37; M, 12.59 % Calc.: C, 46.49; H, 3.12; N, 5.42; M, 12.65. Color, Yellowish cream; Molar conductivity (Ω-1 mol-1cm2) in DMSO 14. Magnetic moment μ eff (BM): 0.0
Biological studies
Antimicrobial studies
All the synthesised compounds were screened for their antimicrobial efficacies and their MIC values were calculated.20 It is observed from the collected data that complex ACP1 is possessing good activity against B. Subtilis and C. albicans and even good activity in caparison with that of free ligand ACP. In the similar way Complex ACP4 is having good activity against C. albicans with a MIC value of 16 (ug/ml). In nut shell we can conclude that all the complex possessing good activity against C. albicans. The good activity may be explained on the basis of Tweedey’s chelation theory. (Table 1& Figure 5).
Table 1: MIC (ug/ml) of the synthesised compounds
| Compounds | E. coli | B. Subtilis | C. albicans | A. niger |
| Ligand-ACP | 64 | 64 | 32 | 32 |
| ACP-1 | 32 | 16 | 16 | 64 |
| ACP-2 | 128 | 32 | 16 | 64 |
| ACP-3 | 128 | 32 | 32 | 32 |
| ACP-4 | 64 | 64 | 16 | 32 |
| Ciprofloxacin | 6.25 | 6.25 | —– | —— |
| Amphotericin | —— | —– | 12.5 | 12.5 |
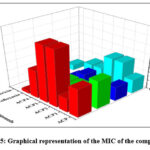 |
Figure 5: Graphical representation of the MIC of the compounds.Click here to View Figure |
ADME Studies
Tables 2 and 3 list the different parameters that were computed for the ligand and complexes. Each of the several factors describes a specific attribute of the chemical and has a unique meaning. Topological Polar Surface Area is referred to as TPSA. As the name suggests, this characteristic is related to the compound’s polar atom surface. It aids in figuring out the medications’ or compound’s transport characteristics. The TPSA value falls between 102.32 and 108.1 Å. A certain number of rotational hydrogen atoms contribute flexibility to the compounds. (See Tables 2 & 3) Nevertheless, in the current investigation, the ligand contains 4 rotational hydrogen atoms while the remaining compounds have none. This indicates that the ligand possesses greater flexibility compared to the other complexes. The bioavailability score for all the complexes and the ligand was determined to be 0.55, suggesting a higher likelihood of bioactivity for these compounds. The number of hydrogen donors is 4 in all instances, but there are no donors in the ligand. All compounds exhibit drug-like properties, and pharmacokinetic evaluations indicate that none of the complexes are capable of crossing the blood-brain barrier, nor do they have synthetic accessibility scores comparable to the free ligand. The bioavailability radar is illustrated in Figure 7. The pink region in the image represents the ideal range for each parameter, while the red line indicates the values obtained for the corresponding parameters.
 |
Figure 6: Radar Diagram of the ligand and it metal complexes.Click here to View Figure |
Table 2: Pharmaco-kinetic data of the compounds
| Compounds | Mol.wt. | TPSA | H-Acceptor | H-Donor | No. of Rotatable Bonds | Bioactivityscore |
| ACP | 417.37 | 102.32 | 2 | 0 | 4 | 0.55 |
| ACP-1 | 510.32 | 108.1 | 2 | 2 | 0 | 0.55 |
| ACP-2 | 510.08 | 108.1 | 2 | 2 | 0 | 0.55 |
| ACP-3 | 514.94 | 108.1 | 2 | 2 | 0 | 0.55 |
| ACP-4 | 516.77 | 108.1 | 2 | 2 | 0 | 0.55 |
Table 3: Various parameters of the compounds
| Compounds | Gastrointestinal Absorption | BBB Permeation | P-glycoprotein Substrate | CYP3A4 inhibitor | Skin Permeation (log Kp) | Synthetic Accesibility score | |||
| ACP | Low | No | Yes | Yes | -4.53 | 2.74 | |||
| ACP-1 | Low | No | Yes | Yes | -5.57 | 4.27 | |||
| ACP-2 | Low | No | Yes | Yes | -5.57 | 4.17 | |||
| ACP-3 | Low | No | Yes | Yes | -5.60 | 4.24 | |||
| ACP-4 | Low | No | Yes | Yes | -5.61 | 4.32 | |||
Semi empirical Studies
Semi-empirical calculations were carried out with PM6 parameter. (Figure 7 & 8) Their energy minimisation were performed and values are tabulated in Table 4. The values of dipole moment were also calculated For compound ACP value is 3.3312 Debye, ACP-1 possess value of 2.322 Debye . In case of compound ACP-4 the value lies at 6.427 Debye. The energy difference between HOMO and LUMO were interpreted and revealed that the stability order is ACP> ACP-4> ACP-1. The conclusion is drawn on the basis of their energy difference calculations.
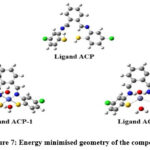 |
Figure 7: Energy minimised geometry of the compoundsClick here to View Figure |
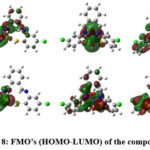 |
Figure 8: FMO’s (HOMO-LUMO) of the compoundsClick here to View Figure |
Table 4: Optimised energy and Energy difference between HOMO & LUMO
| Compounds | Minimised Energy (a.u.) | Energy (a.u.) HOMO | Energy (a.u.) LUMO | ∆E (a.u.) |
| ACP | 0.1632 a.u | -0.327 | -0.0327 | 0.294 |
| ACP-1 | -0.0501 a.u. | -0.254 | -0.1019 | 0.152 |
| ACP-4 | -0.0661 a.u. | -0.293 | -0.0504 | 0.242 |
Conclusion
Novel Schiff base and its metal complexes have been prepared by using 2-amino-4-chlorobenzenethiol and phthaldehyde. Ligand behave as tetradentate ligand. An distorted octahedral geometry may be proposed to all the metal complex on the bassis of spectroscopic and physico-analytical data. Good antimicrobial and pharmaco-kinetics efficacies were found in the compounds. Semi-empirical studies were carried out for energy minimisation and stability.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- R. Nair, A. Shah, S. Baluja and S. Chanda, J. Serb. Chem. Soc. 2006, 71 (7), 733.
CrossRef - Kargar , M. F. Mehrjardi , R. B.Ardakani , H. A. Rudbari , A. A. Ardakani , S. S. Khavidak , K. S. Munawar , M. Ashfaq , M. N. Tahir, Inorganica Chimica Acta, 2022,530, 120677
- K.Bhardwaj , M. S. Hundal , M. Corbella , V. Gómez , G. Hundal, Polyhedron 2012, 38, 224.
- Jafari , M. Salehi , M. Kubicki , A. Arab , A. Khaleghian, Inorganica Chimica Acta 2017, 462, 329.
- Kargar , A. A. Ardakani , M. N. Tahir , M. Ashfaq , K. S. Munawar, J. Mol. Struct. 2021, 1233, 130112.
- Biswas, A. Ghosh, Polyhedron, 2013, 65, 322.
CrossRef - Thakurta , C. Rizzoli , R.J. Butcher , C.J. GómezGarcía , E. Garribba, S. Mitra, Inorganica Chimica Acta, 2010, 363, 1395.
CrossRef - Bhattacharyya , S. Sen , K. Harms , S. Chattopadhyay, Polyhedron, 2015,88, 156.
CrossRef - Sani , S.M. Iliyasu , J. Pure and Applied Sciences, 2018 11(1), 214.
CrossRef - A.Z. El-Sonbati, W.H. Mahmoud, G.G. Mohamed, M.A. Diab, S.M. Morgan, S.Y. Abbas, J. Appl. Organometal Chem., 2019, e 5048.
- D. Maity, International J. Research and Analytical Reviews, 2019 6, 1269.
- M.N. Uddin, S.S. Ahmed, and S.M. Rahatul Alam, Biomedical applications of Schiff base metal complexes. J. Coordination Chem.,2020, 73(23), 3109.
CrossRef - S. Shahraki, F. Shiri, M. Saeidifar, J. Biomol. Struct. Dynam., 2017, 36, 1666.
CrossRef - J. Lin, D. C. Sahakian, S. De Morais, J. J. Xu, R. J. Polzer, S. M. Winter, Curr. Topics Med. Chem. 2003, 3, 1125.
CrossRef - V. Sangwan & D.P. Singh, J. Chin. Chem. Soc. 2020, 67, 592.
CrossRef - W.A. Zoubi, A.A.S.A. Hamdani, S.D. Ahmed, Y.G. Ko, J. Phys. Org. Chem, 2017, e3752.
- A. Sharma, N. Dhingra, H.L. Singh, S. Khaturia, U. Bhardwaj, J. Mol. Struct., 2024, 1261.
- G.G. Mohamed, M.M. Omar, A.M. Hindy, Turkish Journal of Chemistry,2006, 30, 361-382
- V. Sangwan & D.P. Singh, J Chin Chem Soc. 2020, 67, 1024
CrossRef - V. Sangwan & D.P. Singh, Polyhedron, 2024, 264, 117230
CrossRef
Accepted on: 29 Mar 2025

















