Quality Control in Pharmaceutical Fused Deposition
1Department of Pharmacy, Sumandeep Vidyapeeth Deemed to be University, Vadodara, Gujarat, India
2Department of Pharmaceutical Chemistry, JSS College of Pharmacy, Mysuru, Karnataka, India
3Pioneer Pharmacy College, Ajwa-Nimeta road, Vadodara, Gujarat, India
4JNCT Professional University Bhopal (M. P.), India.
5Indukaka Ipcowala College of Pharmacy (IICP), CVM UNIVERSITY, New V V Nagar, Anand, Gujarat, India.
Corresponding Author E-mail: harshsvdu@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/420140
ABSTRACT:Fused Deposition Modelling (FDM) is a new 3D printing method that has a lot of promise for the pharmaceutical industry, especially for creating intricate and patient-specific drug delivery systems. Consistently producing high-quality products is still a major obstacle, though. This paper describes the key elements that affect pharmaceutical FDM quality, such as drug-polymer compatibility, polymer selection, and process parameter optimization, including extrusion temperature, infill density, and cooling rates. Dosage form dependability depends on post-printing evaluations of mechanical strength, dissolving behaviour, and drug content homogeneity. For their functions in assessing physical and chemical properties, sophisticated analytical methods such as Differential Scanning Calorimetry (DSC), Fourier Transform Infrared Spectroscopy (FTIR), X-ray Diffraction (XRD), Scanning Electron Microscopy (SEM), and Micro-Computerized Tomography (Micro-CT) are highlighted. The combination of artificial intelligence (AI), Process Analytical Technology (PAT), and real-time monitoring tools to facilitate predictive quality control is also examined in the paper. Future directions for using FDM in pharmaceutical practice as well as current regulatory considerations are covered. All things considered, this review highlights the necessity of strong quality control methods to support the secure and efficient application of FDM in customized medicine.
KEYWORDS:3D printing in pharmaceuticals; Drug-polymer compatibility; Fused Deposition Modelling (FDM); Personalized drug delivery; Quality control
Introduction
The development of personalized dosage forms and medicines using 3D printing technologies has emerged as a promising area of pharmaceutical research, drawing significant attention from the scientific community. This innovation has been increasingly embraced by the pharmaceutical industry, leading to a sharp rise in related publications and intellectual property filings. Interest in various 3D printing techniques for fabricating drug delivery systems surged following the U.S. Food and Drug Administration’s approval of Spritam™, the first 3D-printed tablet developed by Aprecia Pharmaceuticals. At the core of this advancement lies the goal of enabling tailored, patient-specific, and on-demand drug delivery solutions.1
The integration of 3D printing into pharmaceutical production, particularly through Fused Deposition Modeling (FDM), represents a major breakthrough. This additive manufacturing technique involves the extrusion of thermoplastic materials in layers to create dosage forms, providing opportunities for the precise control of drug release and dosing. Despite the advantages, applying FDM in the pharmaceutical sector necessitates rigorous quality control to ensure compliance with regulatory standards and therapeutic efficacy.2
Review Methodology
A thorough review of the literature was conducted using databases such as PubMed, ScienceDirect, and Google Scholar. Relevant articles from the years 2015 to 2024 were included, with an emphasis on peer-reviewed publications, regulatory guidelines, and case studies. Search terms like “Fused Deposition Modeling,” “3D printing in pharmaceuticals,” “quality control,” and “personalized drug delivery” were used to gather information.
FDM in Pharmaceutical Manufacturing
FDM operates by melting a thermoplastic filament and extruding it through a nozzle to build structures layer by layer. In drug manufacturing, the uniformity and stability of the drug-polymer blend are critical. Commonly used polymers such as polyvinyl alcohol (PVA), polylactic acid (PLA), and ethyl cellulose must be compatible with the active pharmaceutical ingredient (API) and capable of withstanding the printing process.3
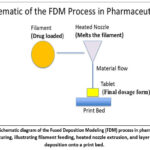 |
Figure 1: Schematic diagram of the Fused Deposition Modeling (FDM) process in pharmaceutical manufacturing, illustrating filament feeding, heated nozzle extrusion, and layer-by-layer deposition onto a print bed. |
Key Quality Attributes in FDM
Materials and Methods
Material Selection
The properties of the materials used heavily influence the performance of FDM-printed pharmaceuticals. Considerations include:
Thermal characteristics of the drug and polymer
The thermal properties of polymers are a critical consideration in FDM-based pharmaceutical applications, as they influence both the polymer’s processability and the incorporated drug’s thermal stability. Most commonly used thermoplastics in FDM, such as PLA, ABS, PETG, and Nylon, exhibit relatively low glass transition (Tg) and melting temperatures (Tm), which can be beneficial for processing temperature-sensitive drugs. However, their limited thermal and mechanical stability restricts their use in high-temperature applications.
In contrast, high-performance polymers like PEEK, PEKK, and PEI offer superior thermal stability, with higher Tg and Tm values. These polymers are more suitable for sustained-release formulations or implants requiring mechanical strength and thermal durability. However, their elevated processing temperatures pose a risk of thermal degradation to heat-labile drugs, limiting their compatibility in certain formulations.
During FDM, maintaining the polymer temperature above Tg (for amorphous polymers) or close to Tm (for semi-crystalline polymers) is essential for proper extrusion and layer adhesion. However, these temperatures must remain below the degradation temperature (Tdeg) of the drug to prevent chemical degradation or loss of activity. For successful formulation, a narrow processing window is required, where the polymer is adequately flowable and the drug remains stable.
In composite systems, the inclusion of fillers or reinforcements can further modify the thermal behavior of the polymer, potentially increasing Tg and Tm, which may reduce compatibility with thermosensitive drugs. Hence, the selection of a polymer for FDM drug delivery must balance mechanical and thermal requirements with the stability profile of the active pharmaceutical ingredient (API)[4]
Table 1: lists the thermal characteristics of frequently used FDM polymers as well as the model medications’ thermal breakdown temperatures. Consequences for drug–polymer compatibility in FDM Processing.
(a) FDM Polymer Thermal Characteristics
| Polymer | Glass Transition Temp (Tg, °C) | Melting Temp (Tm, °C) | Typical Processing Temp (°C) | Thermal Stability | FDM Suitability for Thermolabile Drugs | References |
| PLA | ~60 | 150–180 | 180–210 [2] | Low to Moderate | High | [5] |
| ABS | ~105 | Amorphous | 210–250 [3] | Moderate | Moderate | [6] |
| PETG | ~85 | ~230 | 220–260 [4] | Moderate | Low to Moderate | [7] |
| Nylon (PA) | ~50 | 190–220 | 240–270 [5] | Moderate to High | Low | [8] |
| PEI | ~215 | Amorphous | 340–380 [6] | Very High | Very Low | [9] |
| PEEK | ~143 | ~343 | 360–400 [6] | Very High | Very Low | [9] |
| PEKK | ~160–165 | 300–360 | 350–390 [6] | Very High | Very Low | [9] |
(b) Thermal Degradation Temperatures of Selected Drugs
| Drug | Approx. Degradation Temp (Tdeg, °C) | Recommended FDM Polymer(s) | Notes on Compatibility | References |
| Ibuprofen | ~76–90 [10} | PLA | Only with low-temperature polymers | [13] |
| Paracetamol | ~170 [7] | PLA, ABS | Moderate stability; avoid PETG and above | [10] |
| sIndomethacin | ~160 [11] | PLA, ABS | Stable for standard low-temp FDM | [14] |
| Propranolol HCl | ~130 [8] | PLA | Slight degradation risk at moderate temps | [11] |
| Theophylline | ~270 [9] | PETG, PEI, PEEK | Suitable for high-temperature polymers | [12] |
Compatibility between the API and excipients
In preformulation studies, various analytical techniques are utilized to assess the compatibility between active pharmaceutical ingredients (APIs) and excipients. Derivative thermogravimetric analysis (DTG) and differential scanning calorimetry (DSC) are particularly useful for simulating accelerated aging conditions, allowing for the evaluation of phase transitions and chemical reactions that influence the stability of the drug formulation. These techniques measure enthalpy changes and mass loss, which are key indicators of the sample’s stability under virtual processing and storage conditions.
By combining thermal analysis with spectroscopic and diffractometric techniques, researchers can gain valuable insights into drug stability that would typically take months to determine through traditional stability studies. This integrated approach helps to anticipate the long-term stability of pharmaceutical products.
Furthermore, recent advancements in additive manufacturing, particularly in 3D printing using fused deposition modeling (FDM), have shown great promise in the pharmaceutical industry. This technique allows for the creation of dosage forms with highly customizable compositions, release profiles, shapes, and sizes to meet specific patient needs. To produce these 3D-printed dosage forms, APIs and excipients are first processed through hot-melt extrusion (HME) to form drug-loaded filaments. These filaments are then printed using 3D/FDM printers, where controlled heating and extrusion ensure precise layering and structure. Importantly, ensuring compatibility between the API and excipients in the filament is crucial for maintaining the efficacy and stability of the final dosage form.15
Mechanical properties and processability of the filament
The functional applicability of components made via rapid prototyping, especially Fused Deposition Modeling (FDM), in the engineering and biomedical fields is significantly influenced by their mechanical integrity. Numerous studies have shown that the composition of the material and the processing conditions significantly impact mechanical qualities, including tensile, flexural, and impact strength. Interlayer adhesion and overall structural performance are strongly influenced by construction orientation, raster angle, layer thickness, and extrusion temperature. The creation of composite filaments based on polymers has received a lot of interest to improve mechanical behavior. Increased wear resistance has been demonstrated by reinforced thermoplastics like acrylonitrile butadiene styrene (ABS) embedded with iron or copper particles and nylon matrices modified with solid lubricants like graphite, molybdenum disulfide (MoS₂), and polytetrafluoroethylene (PTFE).16
Process Optimization
Controlling process parameters is critical for ensuring consistent product quality. Key factors include:
Extrusion and bed temperatures
The mechanical properties of the parts produced by Fused Deposition Modelling (FDM) of Poly-ether-ether-ketone (PEEK) are greatly influenced by a number of processing parameters, two of which are crucial because of the high melting point of PEEK: the extrusion temperature (also known as the nozzle temperature) and the bedplate temperature, which should normally be between 350 and 440°C and 100 and 150°C, respectively, to ensure proper adhesion and minimize warping during the printing process; the chamber temperature is also important, with ideal conditions falling between 90 and 160°C, as it helps to maintain a stable thermal environment, reducing the thermal stresses that can result in defects like poor interlayer adhesion or cracks in the printed parts.
Printing speed and layer resolution
Important additional factors are the printing speed and layer resolution (also known as layer height). The rate at which material is deposited is determined by the printing speed, and this can have an immediate impact on the printed part’s surface finish and mechanical characteristics. While slower printing speeds can increase layer adhesion and strength, faster printing speeds may result in less interlayer bonding. The part’s strength, surface quality, and overall dimensional accuracy are all impacted by layer height, which specifies the vertical resolution of the printed layers. To balance print time and mechanical performance, layer height optimization is crucial.
Infill percentage and cooling rates
Additionally, the mechanical qualities like tensile strength and flexibility are influenced by the infill %, which determines the density of the internal structure. Stronger pieces are typically produced with higher infill percentages, but they also use more material and take longer to print. However, cooling rates are just as significant. While gradual cooling might result in better crystallization and enhanced mechanical characteristics, rapid cooling can produce unequal thermal stresses. To prevent flaws like warping or excessive shrinkage during the cooling phase, proper cooling regulation is essential.17
Adjusting these parameters can minimize issues such as deformation, drug degradation, and poor mechanical performance
Post-Printing Assessments
Once printed, the dosage forms undergo several evaluations, such as:
Consistency in size and weight
Pharmaceutical tablet manufacturing using Fused Deposition Modeling (FDM) 3D printing showed a high level of uniformity in tablet weight and size. Tribasic calcium phosphate (TCP) and other thermally stable fillers made it easier for the filament to flow smoothly and steadily during the printing process, guaranteeing regular tablet dimensions. For the precise manufacturing of mechanically stable tablets with specific physical characteristics, this consistency was essential.
Uniformity of drug content
Hot melt extrusion (HME) and FDM 3D printing are two thermal methods that were used to assess the consistency of the drug content in the 3D printed tablets. The following were the drug contents of the model drugs:
94.22% of 5-aminosalicylic acid (5-ASA)
88.53% for captopril
96.51% theophylline
93.04% prednisolone
Despite variations in the medicine’s characteristics, these percentages imply that the drug distribution in the tablets stayed constant throughout the formulation. To guarantee that every tablet contains the recommended dosage of the active pharmaceutical ingredient (API), homogeneity is essential.
Dissolution behavior and stability testing
The physicochemical characteristics of the included model medications have an impact on the printed tablets’ dissolving behavior. X-ray powder diffraction (XRPD) study showed that captopril was present in an amorphous state, while 5-ASA, theophylline, and prednisolone were partially still crystalline. While 5-ASA, theophylline, and prednisolone may have slower dissolution profiles, which are common for crystalline substances, captopril’s amorphous nature may help explain its increased solubility and quicker rate of dissolution.
Stability testing of the 3D printed tablets involved evaluating the integrity of the drugs and the overall product under controlled conditions. The inclusion of thermally stable fillers like TCP, which allowed for consistent filament flow and minimized degradation during the printing process, suggests that the tablets maintain their mechanical stability and drug content stability throughout the manufacturing process. The optimized formulation, combining pharmaceutically approved polymers and fillers, offers a stable method for producing individualized dosage forms on demand, with minimal degradation during processing.18
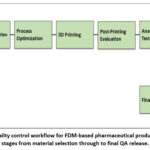 |
Figure 2: Quality control workflow for FDM-based pharmaceutical products, covering stages from material selection through to final QA release. |
Analytical Tools for Evaluation
The following analytical techniques are essential for evaluating the quality of 3D-printed pharmaceutical products:
Differential Scanning Calorimetry (DSC)
A thermal analysis method called DSC is used to quantify heat flow related to transitions such as glass transitions, melting, and crystallization. DSC is essential for assessing the compatibility and heat behavior of drug-polymer complexes in pharmaceutical FDM. It determines the drug-polymer miscibility, glass transition temperature (Tg), and melting point (Tm), all of which are essential for establishing processing parameters and forecasting the drug’s physical stability [19]. For example, a medication may have been molecularly distributed throughout the polymer matrix if its melting peak is absent from a DSC thermogram.19
Fourier Transform Infrared Spectroscopy (FTIR)
FTIR is useful for identifying molecular interactions between the active pharmaceutical ingredient (API) and excipients, such as hydrogen bonding or chemical incompatibility. It offers a distinct spectral fingerprint of the compound’s functional groups. Functional group interactions or degradation during the extrusion or printing process are indicated by any shift, disappearance, or development of distinctive peaks in the spectra. Because of this, FTIR is a useful technique for assessing the drug’s chemical integrity after processing.20
X-ray Diffraction (XRD): The crystalline or amorphous state of the medication in FDM-printed formulations is determined by XRD. Amorphous materials provide broad halos, while crystalline pharmaceuticals show sharp, well-defined diffraction peaks. Following FDM printing, a loss of crystallinity, as shown by the disappearance of particular diffraction peaks, may signal a change to the amorphous form, which might improve solubility but needs stability to avoid recrystallization.21
Scanning Electron Microscopy (SEM): High-resolution imaging of interior structure and surface topography is possible using SEM. It is frequently used to evaluate the morphology of dosage forms that are FDM printed, identifying flaws such layer separation, holes, and cracks [22]. Additionally, SEM helps assess the homogeneity and layer adherence of printed tablets and visualizes the dispersion of medication particles. SEM can also give elemental composition analysis when used in conjunction with Energy Dispersive X-ray Spectroscopy (EDS), which is useful for identifying unequal medication distribution.22
Mechanistic and Inspection Methods
Understanding and guaranteeing the quality of 3D printed things using Fused Deposition Modeling (FDM) requires the application of mechanistic and inspection techniques. The following are some FDM mechanistic and inspection techniques:
Mechanistic Techniques
Finite Element Analysis (FEA): A computational method used to model and analyse the mechanical behavior of 3D-printed components, predicting stresses, deformations, and failure locations.
Thermal Analysis: Examines thermal behavior during the FDM process to optimize parameters and understand temperature effects on layer bonding and material flow.
Material Characterization: Evaluates mechanical properties like modulus, elongation at break, and tensile strength under various conditions.
Rheological Analysis: Studies the flow and deformation behavior of materials to optimize extrusion and prevent issues like nozzle blockage.23
Inspection Methods
Visual Inspection Simple visual check for warping, surface imperfections, or layer adhesion issues.
Dimensional Inspection: Measures printed objects’ dimensions and tolerances using tools like callipers or CMM.
Microscopy: uses optical or SEM to examine layer adhesion, surface finish, and internal defects.
Non-destructive testing: Techniques like CT and X-ray identify internal voids or flaws without damaging the object.
Ultrasonic Testing: Detects internal flaws like delamination or vacancies using sound waves.
Thermographic Inspection: Uses infrared thermography to detect defects during or after printing.
Tensile Testing: Measures elasticity and strength of printed materials to determine mechanical performance.
Porosity Analysis: Uses gas or liquid penetration tests to detect pores or voids in the object.
Integrating inspection and mechanical techniques is crucial to comprehending Fused Deposition Modelling procedures and printed part quality as a whole. These techniques aid in making sure the components fit the intended uses and adhere to design specifications.24
Real-Time Monitoring and Process Analytical Technology (PAT)
Real-time monitoring tools are being used more and more in modern pharmaceutical FDM processes to ensure constant quality throughout production. The drug composition of each printed unit can be analysed in-line without the requirement for destructive testing, thanks to technologies like near-infrared (NIR) spectroscopy. When combined with the FDM printer nozzle, these probes can scan tablets just after they are made, generating spectra that can be compared to calibrated models to determine the accuracy of the content.25-27
In addition to NIR, sensors embedded in the extrusion assembly, such as torque or pressure sensors, help detect irregularities in filament flow, viscosity shifts, or nozzle blockages.28 Coupled with microbalances for real-time weighing, these systems allow each dosage form to be evaluated for weight uniformity during production.29 Such techniques support the emerging paradigm of “real-time release testing,” where batch testing is minimized in Favor of continuous quality verification.30
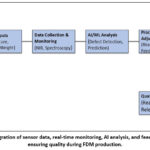 |
Figure 3: Integration of sensor data, real-time monitoring, AI analysis, and feedback loops in ensuring quality during FDM production. Click here to View Figure |
Artificial Intelligence and Machine Learning in Quality Control
Artificial intelligence (AI) is increasingly being used in pharmaceutical FDM to automate inspection and prediction tasks. Machine learning models, including convolutional neural networks (CNNs), have been trained on image data to classify printed dosage forms, detect defects, or predict successful printability based on formulation parameters.31,32
Researchers have shown the efficacy of synthetic training for tablet and capsule recognition using computer-generated image datasets. These technologies are capable of quickly detecting problems such as shape aberrations, under-extrusion, and layer misalignment. In addition to eye examination, machine learning models have been created to forecast process outcomes using past data, including mechanical integrity and medication release characteristics.33
Advanced Analytical Technologies
Nowadays, several high-resolution instruments are employed in addition to conventional analytical methods for quality evaluation in FDM pharmaceutical applications. Active pharmaceutical ingredients (APIs) on the outside and inside of dosage forms can be mapped using Raman Confocal Microscopy.34 It sheds light on the uniformity and solid-state form of medications. Micro-Computerized Tomography (Micro-CT) provides a non-invasive way to see inside structures. It can measure void distribution, porosity, and layer adhesion—all of which affect how drugs are released.35 Following printing, terahertz spectroscopy and Fourier transform infrared (FTIR) imaging provide comprehensive chemical characterization that helps identify polymorphic transitions or degradation products.36
These advanced techniques support comprehensive quality assessments, helping to verify consistency and compliance in printed medicines.
Real-World Applications and Case Studies
Numerous real-world applications have shown how useful FDM is in pharmaceutical environments. Hospitals utilizing desktop 3D printers have effectively created on-demand dosage forms using pre-formulated filaments in a decentralized production paradigm. An in-line NIR probe is used to scan each tablet, allowing for an instant assessment of the medication content.
With relative standard deviations less than 1%, these systems have demonstrated outstanding consistency in tablet weight, size, and API concentration. Process sensors and automated decision systems that pause or stop printing when anomalies are identified are integrated to provide such precision.
The use of microbalances in 3D printers, where each tablet is weighed after printing, is an additional example. Only conforming items are sent to packaging, since any deviation from acceptable weight criteria is noted.
These case studies highlight the feasibility of integrating real-time quality control in decentralized and hospital-based pharmaceutical manufacturing.37
Regulatory Framework
The International Council for Harmonization (ICH) guidelines Q8 (Pharmaceutical Development), Q9 (Quality Risk Management), and Q10 (Pharmaceutical Quality System) are among the frameworks that are guiding the evolution of the regulatory environment for 3D-printed pharmaceuticals. Pharmaceutical items, especially those made using cutting-edge manufacturing techniques like 3D printing, may be guaranteed to be safe, effective, and of high quality thanks to these principles.38
The U.S. Food and Drug Administration’s (FDA) 2015 approval of Sprit am® (levetiracetam), the first 3D-printed medication to be approved, was a major turning point in this field.39 Sprit am® uses the exclusive Zip Dose® technology from Aprecia Pharmaceuticals, which uses a powder bed fusion 3D printing method to produce quickly dissolving tablets that make it easier for patients who have trouble swallowing to take them.40
The FDA has started working to offer more focused recommendations in recognition of the particular difficulties and factors that additive manufacturing presents. The FDA published a guidance document in 2017 called “Technical Considerations for Additive Manufactured Medical Devices,” which lays the foundation for future recommendations tailored to 3D-printed medications, even though it is primarily focused on medical devices.41
Regulatory bodies are anticipated to create more thorough guidelines addressing the unique hazards and controls related to 3D printing in pharmaceutical manufacturing as the technology develops. The goal of these upcoming recommendations is to promote innovation in customized medicine while guaranteeing patient safety and product quality.
Advances in Quality Control
Recent developments in digital technologies in several fields, such as communications, imaging, architecture, engineering, and industrial technology, have significantly advanced production techniques. Because of its high speed and dimensional accuracy, additive manufacturing (AM), which builds objects layer by layer from a material feedstock, has attracted a lot of attention. Engineers use Computer-Aided Design (CAD) files to create and manufacture bespoke components for industries including aerospace, medical implants, and automobile manufacturing to meet the growing need for quick and accurate fabrication.
Nowadays, several AM methods are used, such as Stereolithography (SLA), Direct Metal Deposition (DMD), Fused Deposition Modeling (FDM), Inkjet Modeling (IJM), 3D printing, Selective Laser Sintering (SLS), and Direct Metal Deposition (DMD). The ability of fused deposition modeling to process thermoplastics to produce intricate, highly precise pieces has made it stand out among the others. The FDM technique involves heating a material to a molten state and then using a nozzle to extrude it layer by layer onto a heated substrate. Support structures are frequently made of materials like polycarbonate (PC), acrylonitrile butadiene styrene (ABS), or PC-ABS blends to guarantee the dimensional accuracy of the printed pieces. With an emphasis on improving accuracy and processing speed, this analysis looks at current developments in FDM technology and how they are used in product development.42
Challenges and Limitations of Fused Deposition Modeling
Fused Deposition Modeling (FDM) has great promise for changing medication release profiles by creating intricate geometries and for producing customized dosage forms on a small scale for individualized treatment. Nonetheless, many issues need to be resolved with FDM. These difficulties fall into one of three primary parameter categories. The physicochemical features of the filament, including its mechanical attributes, thermal behavior, and viscosity, are included in material-specific parameters. These qualities have a direct impact on the printing process and the quality of the finished output. They ascertain whether printing is a feasible procedure. The final quality and the dissolving behavior of the objects are affected by operation-specific characteristics, such as printing temperature and infill density, which are connected to the printing processing circumstances. The printing equipment is defined by the particular parameters of each machine. A few modifications to this device also enhance the way the printing process is carried out.43
This session discusses the challenges and considerations that must be made in continuous manufacturing processes, such as HME-coupled 3D printing, where control systems are crucial for managing interruptions. Because of the high temperatures, some materials cannot be employed in these procedures. Finding pharmaceutical-grade polymers suitable for 3D printing is difficult, and the addition of drugs can alter the properties of the polymers.
There are regulations governing 3D-printed medical devices, but not other 3D-printed goods. The FDA released draft rules in 2016, and because there aren’t many 3D printers that comply, pharmaceutical companies are wary. Using additive manufacturing technology to produce medical products: best practices were explored by an FDA working group in 2014. Regulatory organizations closely watch developments in this area.44
Conclusion
FDM-based 3D printing is emerging as a robust platform for producing personalized drug delivery systems. Advances in real-time monitoring, AI-driven inspection, and sophisticated analytical tools have significantly improved quality control capabilities. These developments, along with supportive regulatory initiatives, make it feasible to ensure consistent product quality even in small-scale, on-demand manufacturing.
As these innovations mature, the pharmaceutical industry is poised to integrate FDM into mainstream production, supported by frameworks that promote real-time quality assurance and digital validation.
Acknowledgement
All authors acknowledge the management and principal of Sumandeep Vidyapeeth Deemed to be University, Vadodara & JSS College of Pharmacy, Mysuru, for their encouragement, complete support, and for creating excellent infrastructure facilities to carry out this work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Aftab, M.; Ikram, S.; Ullah, M.; Khan, N.; Naeem, M.; Khan, M. A.; ogle, R. B.; Qizi, K. S. S.; Ugli, O. O.; Abdurasulovna, B. M.; Qizi, O. K. A.J. Manuf. Mater. Process. 2025, 9(4), 129.
CrossRef - Barbosa, A.; Silva, J. M.; Pereira, J.; Rodrigues, M. P.; Correia, L. J. Pharm. Biomed. Anal. 2022, 207, 114331.
- Díaz-Torres, R.; Pérez-Llana, P.; Rodríguez-González, L.; Blanco-López, M. C. Eur. J. Pharm. Sci 2020, 155, 105553.
- Deno, N. C.; Richey, H. G.; Liu, J. S.; Lincoln, D. N.. J. Am. Chem. Soc.1965, 87, 4533-4538.
CrossRef - Ennotte, O.; Lechanteur, A.; Evrard, B.; Parulski, C. Adv. Drug Deliv. Rev. 2021, 175, 113820.
- Fanous, M.; Boudy, P.; Douard, E.; Tissot, M.; Chaminade, P. Int. J. Pharm. 2021, 597, 120321.
- Goyanes, A.; Basit, A. W.; Gaisford, S. Int. J. Pharm.2014, 478(1), 88–92.
CrossRef - Goyanes, A.; Madla, C. M.; Umerji, A.; Gaisford, S.; Basit, A. W. Int. J. Pharm. 2017, 527(1-2), 21–30
- Huan Butta, K.; Burapapadh, K.; Sriamornsak, P.; Sangnim, T. Polymers. 2023, 15(7), 1877.
CrossRef - Impro Precision. 2019, May 30. Inspection method for 3D printed parts (Part 3). Retrieved from https://www.improprecision.com/inspection-method-for-3d-printed-parts//
- Iqbal, H.; Fernandes, Q.; Idoudi, S.; Basin Eni, R.; Billa, N. Polymers.2024, 16(3), 386.
CrossRef - Jamróz, W.; Szafraniec, J.; Kurek, M.; Jachowicz, R. Pharm. Res. 2018, 35(9), 176.
CrossRef - Jennotte, O.; Lechanteur, A.; Evrard, B.; Parulski, C. Adv. Drug Deliv. Rev. 2021, 175, 113820.
- Jørgensen, M. R.; Mønsted, M.; Larsen, S. W.; Rades, T.; Müllertz, A. Eur. J. Pharm. Sci. 2020, 146, 105241.
- Khaled, S. A.; Burley, J. C.; Alexander, M. R.; Yang, J.; Roberts, C. J.J. Controlled Release. 2014, 217, 308–314.
- Khan, M. I.; Iqbal, K.; Khan, S.; Ullah, H.; Alam, S.; Rahman, S. J. Microsc. Micro anal.2021, 27(4), 823–836.
- Kulkarni, V. R.; Chakka, J.; Alkadi, F.; Maniruzzaman, M. Pharmaceutics. 2023, 15(5), 1324.
CrossRef - Mora-Castaño, G.; Giraldo-Restrepo, M.; Jaramillo-Isaza, A.; Arias-Trujillo, J. Addit. Manuf. .2021, 39, 101872.
- Sadia, M.; Sośnicka, A.; Arafat, B.; Isreb, A.; Ahmed, W.; Kelarakis, A.; Alhnan, M. A..Int. J. Pharm. 2016, 513(1-2), 659-668.
CrossRef - Ngo, T. D.; Kashani, A.; Panesar, A.; Nguyen, P.; Hua, T.; Lu, J.; Imbalzano, G.; Dao, T. V. Compos. Part B. 2018, 143, 172-196.
CrossRef - Paul, S. Measurement. 2021, 178, 109402.
CrossRef - Peers Alley Media. 2025. Regulatory landscape for 3D printed pharmaceuticals. Retrieved from https://advanced-3dprinting.peersalleyconferences.com/tracks/regulatory-landscape-for-3d-printed-pharmaceuticals
- Sikder, P.; Challa, B. T.; Gummadi, S. K. Materialia.2022, 22.
CrossRef - Rayna, T.; Striukova, L. Technol. Forecast. Soc. Change.2016, 102, 214–224.
CrossRef - Reddy Dumpa, N.; Bandari, S.; Repka, M. Pharmaceutics.2020, 12(1), 52.
CrossRef - Sadia, M.; Sosnicka, A.; Arafat, B.; Isreb, A.; Ahmed, W.; Kelarakis, A.; Alhnan, M. A.Int. J. Pharm 2016, 513, 258-268.
CrossRef - Schmid, M.; Amado, A.; Wegener, K. AIP Conf. Proc. 2014, 1593, 640–645.
- Seoane-Viaño, I.; Janus Kaite, P.; Goyanes, A.; Basit, A. W.; Gaisford, S. Int. J. Pharm. 2021, 593, 120147.
- Shanmugam, V.; Babu, K.; Kannan, G.; Mensah, R. A.; Ray, S. K. S.; Das, Polymer. Degrad. Stab. 2024, 228, 110902.
CrossRef - Sharma, N.; Jindal, A.; Gupta, R.; Singh, H.; Sharma, R. Pharm. Res. 2023, 40(1), 150–162.
- Skowyra, J.; Parra, E.; Quintans, J.; Saura-Calixto, F.; López-Ramos, J.J. Therm. Anal. Calorim. 2015, 119, 1069–1079.
- Singh, J.; Singh, R.; Sharma, S. Mater. Today: Proc. 2022, 50(Part 5), 886-892.
CrossRef
- Sun, Y.; Zhao, H.; Li, S.; Wang, Y.; Li, L.; He, H.; Hu, Y.; Zhao, L.; Guo, Z.; Cao, D.J. Pharm. Sci. 2023, 112(1), 104–112.
- Tiwari, R. V.; Gupta, R.; Nayak, A.; Patel, A.; Singh, B. Anal. Bioanal. Chem 2022, 414(6), 2671–2682.
- Torrado, A. R.; Torrado-Santiago, S.; Grande, T.; Diez-Sales, O. Addit. Manuf. 2015, 6, 16-29.
- Trenfield, S. J.; Madan, J.; Haddad, F.; Wells, C. M.; Goyanes, A.; Gaisford, S.; Basit, A. W. Adv. Drug Deliv. Rev.. .2018, 132, 79–89.
- Trenfield, S. J.; Madan, J.; Gaisford, S.; Basit, A. W. Adv. Drug Deliv. Rev. 2019, 134, 85–97.
- U.S. Food and Drug Administration (FDA). 2022. Process analytical technology: A framework for innovative pharmaceutical development, manufacturing, and quality assurance. https://www.fda.gov/media/71012/download
- U.S. Food and Drug Administration (FDA). 2022. M10 bioanalytical method validation and study sample analysis. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/m10-bioanalytical-method-validation-and-study-sample-analysis
- Shanmugam, V.; Babu, K.; Kannan, G.; Mensah, R. A.; Ray, S. K. S.; Das, O. Polym. Degrad. Stab. 2024, 228.
CrossRef - Wang, X.; Yu, Y.; Chen, J.; Liu, H.; Wang, Y.; Lv, Y.; Lu, J.; Yu, C. 2020, 113, 101324. Prog. Polym. Sci.
- Xu, X.; Li, S.; Fan, Y.; Lv, H.; Su, J.; Zhang, Y.; Zhang, C.Int. J. Pharm. 2021, 597, 120329
- Yang, Y.; Zheng, R.; Lin, L.; Zhang, S.; Chen, C. Pharmaceutics. 2022, 14(3), 611.
- Zhang, Y.; Wang, X.; Li, S.; Hu, Y.; Ma, Z.; Yang, H.; Cai, G. Int. J. Pharm, 2023, 586, 119508.
Accepted on: 27 Oct 2025
Second Review by: Dr. Panda N
Final Approval by: Dr. Tanay Pramanik

















