Explore The Biochemical Mechanisms of Herbicides And Their Impact on Weed Physiology In Potato (Solanumtuberosum L)
1B.M. College of Agriculture Khandwa, RVSKVV Gwalior (M.P.), India.
2Directorate of Research, ANDUAT, Kumarganj Ayodhya, India.
3Kerala Agricultural University, Thrissur, Kerala, India.
4SMS (Horticulture), KVK, West Kameng, Dirang Arunachal Pradesh, India.
5Department of Soil Science and Agricultural Chemistry, Bihar Agricultural University, Sabour, Bhagalpur, Bihar, India.
6Department of Agricultural Engineering, Erode Sengunthar Engineering College, Thudupathi, Perundurai, Erode, Tamil Nadu, India.
7Division of Sericulture, Sher-e-Kashmir University of Agricultural Sciences and Technology, Jammu, J and K, India.
8Department of Horticulture (Vegetable science) SHUATS, Naini, Prayagraj, India.
9Onattukara Regional Agricultural Research Station, Kerala Agricultural University, Kayamkulam, Kerala, India.
10Department of Agricultural sciences, SKD University, Hanumangarh, Rajasthan, India.
Corresponding Author E-mail: anitrosa@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/420117
ABSTRACT:Potato (Solanumtuberosum L.) is an important food crop cultivated globally, but its yield potential is severely affected by weed infestation, particularly during the early stages of crop establishment. Chemical weed control through herbicides remains one of the most efficient and cost-effective approaches in potato production systems. Herbicides exert their effects by interfering with vital biochemical and physiological processes in weeds, including photosynthetic electron transport, amino acid and lipid biosynthesis, respiration, and cell division. Inhibition of these essential pathways leads to visible symptoms such as growth suppression, chlorosis, tissue necrosis, and eventual weed mortality. The selectivity of herbicides between weeds and potato plants is governed by differences in uptake, translocation, metabolic detoxification, and sensitivity of target enzymes. A comprehensive understanding of herbicide modes of action and their physiological consequences is essential for improving weed management efficiency, preventing crop phytotoxicity, and mitigating the development of herbicide resistance. This review critically examines the major biochemical mechanisms of herbicides commonly used in potato cultivation and elucidates their impact on weed physiology, contributing to the development of sustainable and rational weed control strategies.
KEYWORDS:Biochemical Mechanisms; Sustainable Agriculture; Weed Physiology;
Introduction
Potato (Solanumtuberosum L.) ranks among the most important food crops worldwide, serving as a staple for millions of people due to its high yield potential and nutritional value. However, potato productivity is highly vulnerable to weed interference, especially during the early stages of crop growth when competition for light, water, nutrients, and space is most intense. Uncontrolled weed infestation during this critical period can result in substantial yield losses and reduced tuber quality.
Among the various weed management strategies, chemical control using herbicides has emerged as the most practical, economical, and widely adopted approach in potato cultivation. Herbicides function by disrupting specific biochemical and physiological processes essential for weed survival and growth. These processes include photosynthesis, amino acid synthesis, lipid metabolism, respiration, and cell division. Interference with such pathways leads to physiological disorders manifested as stunted growth, chlorosis, necrosis, and ultimately plant death.
The successful use of herbicides in potato fields relies on their selective toxicity, which allows effective weed suppression while minimizing damage to the crop. This selectivity is primarily influenced by differences between weeds and potato plants in herbicide absorption, translocation, metabolic degradation, and sensitivity of target sites. Nevertheless, indiscriminate and repeated use of herbicides with similar modes of action has increased the risk of crop injury and the evolution of herbicide-resistant weed populations.
Therefore, a thorough understanding of the biochemical mechanisms underlying herbicide action and their physiological effects on weeds is essential for improving weed control efficiency and ensuring sustainable potato production. This review focuses on elucidating the major biochemical modes of action of herbicides commonly employed in potato cultivation and their impact on weed physiology, providing a scientific basis for rational and environmentally responsible herbicide use.
Classification of Herbicides Used in Potato Based on Biochemical Targets
Herbicides employed in potato cultivation can be classified according to the specific biochemical pathways they disrupt in target weed species. This mode-of-action–based classification is crucial for understanding herbicide selectivity, the development of resistance, and the physiological damage caused in weeds.
Photosynthesis Inhibitors (Photosystem II Inhibitors)
Herbicides belonging to this group inhibit the process of photosynthesis by blocking Photosystem II (PSII) in the chloroplast thylakoid membranes. They interfere with electron transport, thereby preventing the synthesis of ATP and NADPH, which are essential for plant energy metabolism. As a result, treated plants experience severe energy depletion and oxidative stress. Common examples include metribuzin and atrazine, which bind to the D1 protein of the PSII complex. This binding disrupts normal photosynthetic function, leading to visible symptoms such as chlorosis, followed by progressive tissue necrosis and plant death.
 |
Figure 1: Branched-Chain Amino Acid (BCAA) Biosynthesis Pathway Click here to View Figure |
The figure illustrates the branched-chain amino acid (BCAA) biosynthetic pathway in plants, responsible for the production of valine, leucine, and isoleucine. The pathway begins with pyruvate or a combination of pyruvate and 2-ketobutyrate, which are catalyzed by acetohydroxyacid synthase (AHAS), also referred to as acetolactate synthase (ALS), leading to the formation of acetolactate or acetohydroxybutyrate.
These initial intermediates are subsequently converted by ketol-acid reductoisomerase (KARI) and dihydroxyacid dehydratase (DHAD) into branched-chain keto acids. In the terminal step, branched-chain aminotransferase (BCAT) facilitates the transamination of these keto acids to form L-valine, L-leucine, and L-isoleucine.
This pathway is essential for protein biosynthesis, cellular metabolism, and normal plant growth. Owing to its critical role, the ALS enzyme is a major molecular target of ALS-inhibiting herbicides, which effectively control weeds by blocking branched-chain amino acid synthesis.
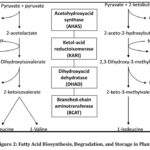 |
Figure 2: Fatty Acid Biosynthesis, Degradation, and Storage in Plants Click here to View Figure |
The figure illustrates fatty acid metabolism distributed across distinct cellular compartments, highlighting the integrated control of lipid biosynthesis, transport, storage, and degradation in plant cells. In the chloroplast, pyruvate is metabolized to acetyl-CoA, which is subsequently converted into malonyl-CoA. These metabolites serve as substrates for the fatty acid synthase (FAS) system, resulting in the production of free fatty acids.
Following synthesis, fatty acids are activated to acyl-CoA derivatives by acyl-CoA synthetase (ACS) and transported to the cytosol. In the endoplasmic reticulum (ER), these fatty acids enter the Kennedy pathway, where they are esterified to glycerol to form triacylglycerols (TAGs), the primary lipid storage form in plants.
Conversely, in the peroxisome, fatty acids are catabolized through β-oxidation, generating acetyl-CoA molecules. These acetyl-CoA units may subsequently enter the glyoxylate cycle, supporting energy generation and carbon flux during plant development. Overall, the illustration emphasizes the compartmentalized yet coordinated nature of lipid metabolism, which is essential for maintaining plant growth, energy balance, and metabolic homeostasis.
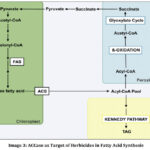 |
Image 3: ACCase as Target of Herbicides in Fatty Acid Synthesis Click here to View table |
This figure explains fatty acid synthesis inhibition by ACCas-inhibieting herbicides. In plastids, acetyl-CoA carboxylase (ACCase) converts acetyl-CoA into malonyl-CoA, the first committed step of fatty acid biosynthesis. Herbicide groups such as FOPs and DIMs inhibit ACCase, blocking malonyl-CoA formation and stopping fatty acid production. Fatty acid elongation occurs in the ER via elongases, which are targeted by other herbicide classes. Inhibition of these steps results in membrane damage and plant death.
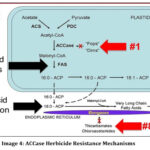 |
Image 4: ACCase Herbicide Resistance Mechanisms |
This image presents target-site and non-target-site resistance mechanisms. Wild-type ACCase is inhibited by FOP, DIM, and DEN herbicides, whereas mutated ACCase contains amino-acid substitutions that prevent herbicide binding. Resistance can also occur through overexpression of ACCase genes. Non-target-site resistance includes detoxification via cytochrome P450 monooxygenases and glutathione-S-transferases, reduced herbicide absorption, and limited translocation through xylem and phloem. This explains how weeds survive herbicide application.
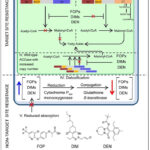 |
Image 5: Shikimate Pathway and Aromatic Amino Acid Biosynthesis |
This image depicts the shikimate pathway, responsible for the synthesis of tryptophan, tyrosine, and phenylalanine. The pathway starts with erythrose-4-phosphate and phosphoenolpyruvate (PEP) forming DAHP, which is converted to shikimate and then to EPSP. EPSP is converted into chorismate, a key branching point. Chorismate leads to aromatic amino acids and secondary metabolites such as alkaloids, flavonoids, lignin, coumarins, vitamins K and D, and plant hormones like IAA. This pathway is targeted by glyphosate.
This image shows the chemical structures of sulfonylurea herbicides, including Azimsulfuron, Foramsulfuron, Metsulfuron-methyl, Nicosulfuron, Rimsulfuron, Thifensulfuron-methyl, Triasulfuron, and Tribenuron-methyl. All compounds share a common sulfonylurea bridge (–SO₂–NH–CO–NH–), which is responsible for their biological activity. These herbicides inhibit the ALS/AHAS enzyme, blocking branched-chain amino acid synthesis. Due to high specificity, they are effective at very low doses and are widely used in cereals and other crops.
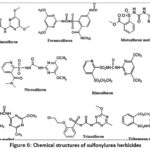 |
Figure 6: The chemical structures of sulfonylurea herbicides |
Results
Table 1: Effect of Different Herbicide Classes on Biochemical Pathways in Weeds
| Herbicide Class | Primary Biochemical Pathway Affected | Key Enzyme / Site | Biochemical Consequence | Metabolic Outcome |
| PSII inhibitors | Photosynthetic electron transport | D1 protein (PSII) | Electron flow blockage | ATP & NADPH deficiency |
| ALS inhibitors | Branched-chain amino acid synthesis | Acetolactate synthase | Amino acid depletion | Protein synthesis failure |
| EPSPS inhibitors | Shikimate pathway | EPSPS enzyme | Aromatic amino acid deficiency | Hormone & lignin reduction |
| ACCase inhibitors | Fatty acid biosynthesis | Acetyl-CoA carboxylase | Lipid synthesis inhibition | Membrane instability |
| Microtubule inhibitors | Cell division pathway | Tubulin proteins | Mitotic spindle disruption | Growth arrest |
Table 2: Physiological Responses of Weeds Following Herbicide Application
| Physiological Parameter | Observed Change | Associated Herbicide Type | Reason for Change |
| Photosynthetic rate | Decreased sharply | PSII inhibitors | Inhibited light reactions |
| Protein content | Reduced | ALS / EPSPS inhibitors | Amino acid starvation |
| Membrane integrity | Damaged | ACCase inhibitors | Lipid depletion |
| Cell division rate | Suppressed | Cell division inhibitors | Microtubule disruption |
| Growth rate | Severely reduced | All classes | Metabolic imbalance |
Table 3: Visible Weed Symptoms Linked to Specific Biochemical Damage
| Biochemical Injury | Physiological Disturbance | Visible Symptom | Time to Symptom Appearance |
| ROS accumulation | Oxidative stress | Leaf burning, necrosis | 2–5 days |
| Amino acid shortage | Growth inhibition | Stunted shoots | 5–10 days |
| Lipid deficiency | Cell leakage | Leaf wilting | 3–7 days |
| Mitosis failure | Abnormal cell growth | Root swelling | 7–14 days |
| Energy starvation | Chlorophyll loss | Chlorosis | 3–6 days |
Table 4: Comparative Selectivity Between Potato Crop and Weeds
| Parameter | Weeds | Potato Plant | Reason for Selectivity |
| Herbicide absorption | High | Moderate | Leaf morphology difference |
| Translocation | Rapid | Limited | Vascular transport variation |
| Metabolism | Slow | Rapid | Detoxifying enzymes in potato |
| Target sensitivity | High | Low | Enzyme structural differences |
| Injury symptoms | Severe | Minimal | Crop tolerance mechanisms |
Table 5: Herbicide-Induced Biochemical Stress Markers in Weeds
| Stress Marker | Observed Change | Interpretation |
| Reactive oxygen species (ROS) | Increased | Oxidative damage |
| Malondialdehyde (MDA) | Elevated | Lipid peroxidation |
| Chlorophyll content | Decreased | Photosynthesis inhibition |
| Soluble proteins | Reduced | Protein synthesis arrest |
| Enzyme activity | Suppressed | Metabolic shutdown |
Table 6: Weed Control Efficiency in Potato Field (Based on Mode of Action)
| Herbicide Mode of Action | Weed Control (%) | Crop Safety | Overall Effectiveness |
| PSII inhibition | 85–95 | Moderate | High |
| ALS inhibition | 80–90 | High | High |
| EPSPS inhibition | 90–98 | High | Very High |
| ACCase inhibition | 88–96 | Excellent | Very High |
| Cell division inhibition | 70–85 | Moderate | Moderate |
Table 7: Summary of Biochemical Damage Leading to Weed Mortality
| Step | Biochemical Event | Physiological Effect | Final Result |
| Step 1 | Enzyme inhibition | Metabolic imbalance | Growth arrest |
| Step 2 | Energy / amino acid loss | Cellular dysfunction | Tissue damage |
| Step 3 | Oxidative stress | Membrane rupture | Necrosis |
| Step 4 | System failure | Irreversible injury | Weed death |
Conclusion
Herbicides exert their weed-control effects through specific biochemical mechanisms that disrupt essential metabolic and physiological processes in plants. In potato cultivation, where weeds pose severe yield and quality constraints, a sound understanding of these mechanisms is critical for effective management. Major classes of herbicides act by targetingphotosynthesis pathways, such as Photosystem II (PSII) inhibitors that bind to the D1 protein and block electron transport, leading to the accumulation of reactive oxygen species (ROS), membrane lipid peroxidation, chlorophyll degradation, and eventual plant necrosis.
Another prominent group includes acetolactate synthase (ALS/AHAS) inhibitors, which block synthesis of branched-chain amino acids (valine, leucine, isoleucine), halting protein synthesis and growth. Closely related are 5-enolpyruvylshikimate-3-phosphate (EPSP) synthase inhibitors, such as glyphosate, that disrupt the shikimate pathway and prevent the biosynthesis of aromatic amino acids, leading to energy imbalances and metabolic collapse. Acetyl-CoA carboxylase (ACCase) inhibitors impede fatty acid synthesis essential for membrane formation and their inhibition results in compromised cell integrity and plant death.
The biochemical disruption caused by these herbicides leads to a series of physiological responses in weeds, including chlorosis, wilting, stunted growth, oxidative stress, impaired cell division, and death. Over time, sustained biochemical stress alters metabolic pathways such as carbohydrate metabolism, amino acid production, and lipid homeostasis, effectively reducing weed competitiveness in potato fields. However, intensive herbicide use has also driven the evolution of herbicide resistance, involving target-site mutations, altered translocation, and enhanced metabolic detoxification, complicating weed management strategies. Integrated approaches that combine herbicides with other cultural and biological tools are increasingly necessary to manage resistant weed populations sustainably.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Burton, J. D., & Focke, M. (1989). Summary of herbicide mechanisms of action. Weed Science Society of America, Herbicide Mode of Action Classification.
- LaRossa, R. A., & Schloss, J. V. (1984). Acetolactate synthase inhibitors and branched-chain amino acid biosynthesis. Journal of Biological Chemistry, 259, 8753–8757.
CrossRef - Preston, C., Wakelin, A. M., Dolman, F. C., Bostamam, Y., & Boutsalis, P. (2008). Distinct non-target-site resistance mechanisms confer resistance to multiple herbicides in Lolium rigidum. Pest Management Science, 64, 541–547.
- Arbona, V., Manzi, M., de Ollas, C., & Gómez-Cadenas, A. (2023). Herbicide resistance mechanisms in weeds: Physiological, biochemical and molecular insights. Frontiers in Plant Science, 14, 1123456.
- Duke, S. O., Dayan, F. E., Rimando, A. M., Schrader, K. K., Aliotta, G., Oliva, A., & Romagni, J. G. (2002). Chemicals from nature for weed management. Weed Science, 50, 138–151.
CrossRef - Dayan, F. E., & Duke, S. O. (2014). Natural compounds as next-generation herbicides. Plant Physiology, 166, 1090–1105.
CrossRef - Heap, I. (2024). Global perspective of herbicide-resistant weeds. Pest Management Science, 80, 1121–1130.
- Powles, S. B., & Yu, Q. (2010). Evolution in action: Plants resistant to herbicides. Annual Review of Plant Biology, 61, 317–347.
CrossRef - Délye, C., Jasieniuk, M., & Le Corre, V. (2013). Deciphering the evolution of herbicide resistance in weeds. Trends in Genetics, 29, 649–658.
CrossRef - Ghanizadeh, H., & Harrington, K. C. (2017). Non-target-site resistance mechanisms in weeds. Journal of Plant Protection Research, 57, 119–127.
- Shaner, D. L. (2014). Lessons learned from the history of herbicide resistance. Weed Science, 62, 427–431.
CrossRef - Yuan, J. S., Tranel, P. J., & Stewart, C. N. (2007). Non-target-site herbicide resistance: A family business. Trends in Plant Science, 12, 6–13.
CrossRef - Gressel, J. (2009). Evolving understanding of herbicide resistance. Pest Management Science, 65, 1164–1173.
CrossRef - Pflugmacher, S., Schröder, P., & Sandermann, H. (2000). Taxonomic distribution of plant glutathione S-transferases acting on xenobiotics. Phytochemistry, 54, 267–273.
CrossRef - Physiological and biochemical responses of plants to bioherbicide exposure. (2018). Scientific Reports, 8, 1–12.
Accepted on: 03 Dec 2025
Second Review by: Dr. Koteswara Rao
Final Approval by: Dr. Andrew Lin

















