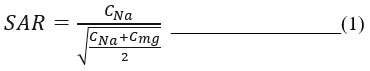Mitigating Abiotic Stress and Enhancing Crop Yield Through Micronutrient-Based Nano Fertilizer
1Department of Environmental Sciences, Maharshi Dayanand University, Rohtak, Haryana, India
2Environmental Microbiology Lab, Rohtak, Haryana, India
Corresponding Author E-mail: manishalohchab558@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/410511
ABSTRACT:Micronutrient deficiencies are a critical barrier to sustainable agriculture, significantly impacting crop yield and quality worldwide. Iron (Fe), copper (Cu), and zinc (Zn) deficiencies are particularly prevalent in plants grown in calcareous soils due to their high alkalinity, low organic matter content, and unfavourable texture, which limit nutrient bioavailability. Conventional fertilizers often exhibit poor nutrient efficiency due to leaching, fixation, and volatilization, necessitating advanced solutions such as micronutrient-based nano fertilizers (MNFs). These nanomaterials leverage unique physicochemical properties, including high surface-area-to-volume ratio, enhanced solubility, controlled nutrient release, and increased bioavailability, to optimize plant nutrient uptake and mitigate soil nutrient depletion. This review explores the role of ZnO, CuO, and Fe₂O₃ nanoparticles in improving nutrient use efficiency (NUE), enhancing photosynthetic activity, and promoting stress tolerance under various abiotic conditions, including salinity, drought, and heavy metal toxicity. Furthermore, it examines the environmental fate of nano-fertilizers, potential ecotoxicological concerns, and regulatory challenges associated with their large-scale application. While MNFs hold promise for revolutionizing precision agriculture and soil fertility management, further research is required to develop standardized formulations, assess long-term soil-microbe interactions, and integrate nano-enabled fertilizers into sustainable agricultural systems.
KEYWORDS:Abiotic stress mitigation; CuO; Fe₂O₃; Nano fertilizers; Sustainable agriculture; ZnO
Introduction
As global population growth surges, so does the demand for food grain production. However, several challenges complicate efforts to meet this demand sustainably, including mismanagement of agricultural land, excessive fertilizer usage, climate change, environmental degradation, over-reliance on non-renewable resources, and rapid urbanization. These factors contribute intensely to food shortages and intense pressure on agricultural systems. In India, where the population has exceeded 1.4 billion, it now surpasses China’s population.1 The need for record-breaking food grain production has reached unprecedented levels. Recently, India produced 342.3 million tons of food grains, surpassing the previous record by 5.32 million tons. Among the top ten food grain-producing states, Haryana ranks sixth with an output of 16.38 million tonnes over 4.47 million hectares.2 However, this increased agricultural productivity also depletes essential soil nutrients, particularly micronutrients critical for crop development and human health. Although required in small amounts, micronutrients play pivotal roles in critical metabolic functions, including photosynthesis, nitrogen fixation, cation balance, and amino acid synthesis. 3 Deficiencies of micronutrients, especially zinc (Zn), iron (Fe), and copper (Cu), are widespread across South Asia, affecting countries like India, Pakistan, Afghanistan, Nepal, Bhutan, Sudan, and Bangladesh. 4,5 Approximately 36.5% of Indian soils are estimated to be deficient in zinc, with iron and copper deficiencies affecting 12% and 3.3% of soils, respectively. Specifically in Haryana, analyses show significant deficiencies in zinc (15.3%), copper (5.2%), and iron (21.6%).6,7 Such deficiencies underscore the need for targeted nutrient management strategies to ensure optimal crop productivity in nutrient-deficient regions. Among essential micronutrients, zinc, copper, and iron are critical for plant health and productivity.
Micronutrient deficiencies hinder crop growth and lead to physiological disorders, such as stunted growth, chlorosis, and leaf deformation, which reduce yields and quality.8 To address nutrient deficiencies, farmers worldwide have traditionally used bulk fertilizers containing water-soluble salts, including sulfates or chelated forms such as ZnSO4, Fe2(SO4)3, ammoniated zinc, and Fe/Zn chelates like EDTA and DTPA.9 While effective, these fertilizers present challenges, including 40-70% nutrient leaching, runoff, nutrient fixation in soil, and reactivity, necessitating higher dosages.10 Additionally, the overuse of fertilizers and pesticides for crop growth and nutrient density enhancement often results in soil degradation, water contamination, and environmental damage, further exacerbated by rising fertilizer costs.
In this context, nanotechnology has emerged as a promising approach to tackling agricultural challenges like nutrient deficiencies, land quality decline, and nutrient loss through leaching. Agriculture increasingly uses it to enhance the efficiency of inputs such as fertilizers. Nano-fertilizers, due to their high surface area-to-volume ratio, controlled release, targeted delivery, and reduced environmental impact, represent an innovative approach to enhancing nutrient absorption efficiency.11,12 Nanoparticles like ZnO, CuO, and Fe₂O₃ have improved root size, chlorophyll content, protein production, and yield.11 In recent research,13 Demonstrated that spraying rice with zinc oxide nanoparticles can enhance yield and nutrient density under heat-stress conditions, showing a 22.1% yield increase compared to untreated plants. Controlled-release formulations and nanoscale coatings are gaining scientific interest, as evidenced by an upward trend in related publications, particularly since 2017.14
As the agricultural sector strives to enhance crop yields sustainably, it becomes crucial to compare conventional fertilizers and nano-fertilizers to understand their effectiveness, impact on soil quality, and potential to fulfill modern agricultural demands (Figure 2). Conventional fertilizers must be applied substantially due to low nutrient utilization efficiency and their adverse effects on soil quality. In contrast, nano fertilizers release nutrients gradually, potentially enhancing nutrient use efficiency without causing harmful side effects.15 Adding ZnO nanoparticles to other fertilizers in zinc-deficient soil led to a 91% increase in barley productivity and improved resource utilization efficiency compared to conventional ZnSO4, which only resulted in a 31% productivity increase relative to the control.16 Nano-composite fertilizers demonstrate beneficial impacts on rhizosphere bacteria by promoting the production of secondary compounds and enhancing the development of plants. Additionally, studies show that using controlled-release fertilizers increased wheat yield and residual soil mineral nitrogen by 6% and 10%, respectively, while reducing leaching and runoff losses by 25% and 22%, respectively, compared to bulk fertilizers.17 Conventional fertilizers are costly and can harm human health and the environment. In contrast, nano fertilizers are crucial for sustaining soil productivity and improving crop production.18 Nano fertilizers represent sophisticated farming resources created to deliver micronutrients to soils in a regulated way, and the advantage of nano fertilizers is their nanoscale size, enabling them to penetrate soil, water, and plants. Nano fertilizers are gradual-release agents that address soil acidity, moisture, and temperature fluctuations, promoting more effective plant growth19 and present significant potential to enhance resource utilization efficiency, optimize costs, and minimize environmental degradation.20 Therefore, this study investigates the efficacy of ZnO, CuO, and Fe₂O₃-based nano fertilizers in alleviating abiotic stress and enhancing crop yield, presenting a sustainable and efficient alternative to conventional bulk fertilizer applications.
Deficiency of micronutrients
While macronutrients like nitrogen, phosphorus, and potassium are vital for plant growth, micronutrients such as copper, zinc, and iron play equally crucial roles in enhancing crop quality, boosting yields, and ensuring sustainable agricultural practices.21
A 2017 study by ICAR identified significant micronutrient deficiencies across India. The ICAR-All India Coordinated Research Project on Micro and Secondary Nutrients and Pollutant Elements in Soils and Plants (AICRP-MSPE) analyzed over 200,000 geo-referenced soil samples from various districts as shown in Table 1. Micronutrient deficiencies are increasingly prevalent in extensively cultivated crops, including vegetables, oilseeds, pulses, and grains. While the widespread and consistent application of zinc fertilizers has significantly reduced zinc deficiencies, multi-micronutrient deficiencies are emerging as a critical challenge in modern agricultural systems.22 Nano-fertilizers are revolutionizing agricultural practices by enhancing the efficiency of micronutrient delivery. For example, zinc and copper oxide nanoparticles address soil deficiencies, promote root elongation, and plant growth.23 And improve crop stress tolerance.24 Iron nano-chelates offer a promising solution for chlorosis-prone soils, providing targeted and efficient nutrient delivery that minimizes environmental impacts.25 Future strategies should integrate traditional soil fertility management practices with advanced nano-technologies to create a synergistic approach. Precision agriculture tools like drones and IoT-based sensors can further optimize the application of micronutrients, ensuring sustainable and productive farming systems.
Release mechanisms of coated micronutrient-based nano fertilizers
Several delivery routes exist for micronutrient nano fertilizers, such as applying nanoscale fertilizers through foliar and soil applications and integrating nanoscale additives and composites to enter plant systems. In soil, MtNPs can be taken up by root hairs where carrier proteins are attached, and nano fertilizers can enter cells by endocytosis, ion channels, or aquaporin.26 Several studies on the release of micronutrients from coated metal nanoparticles (MtNPs) suggest that a soil water concentration/pressure gradient plays a key role in nutrient release. The primary mechanism for nutrient release from polymer-coated fertilizers is the movement of nutrients from the fertilizer-polymer interface to the polymer surface. Initially, water enters the ZnO-gum Acacia, CuO-chitosan, or chitosan-hydrogel Fe2O3 nano-fertilizers as soil moisture. This causes the coating to swell, transforming the nano-fertilizer into a hydrogel.27 As water is absorbed, the soluble components of the fertilizer dissolve and spread into the nanocomposite core. The gel releases these dissolved nutrients into the soil while the undissolved fraction breaks down due to moisture and microbial activity. Plants then absorb nutrients through root hair, leading to apoplastic or symplastic movements, reaching various parts of the plant 27,28. Gum acacia/chitosan/hydrogel is essential for controlling the rate of nutrient release by preventing premature matrix breakdown and regulating water diffusion, ensuring that plants receive a steady supply of micronutrients over long periods. To optimize Nutrient utilization efficiency (NUE) and minimize ecological impact, controlled-release micronutrient fertilizers (CRNFs) provide a practical and promising solution by supplying plants with needed nutrients. These nano-fertilizers are engineered to deliver nutrients gradually at the targeted plant site, activated by moisture, pH, and temperature conditions. Integrating with carrier molecules enhances nutrient delivery to crops, minimizes environmental risks, and boosts crop yields. Nutrient release from coated nano fertilizers occurs in three phases from the fertilizer core: (1) the initial phase, (2) the steady release phase, and (3) the decline phase. During the initial phase, the difference in vapor pressure across the coating fuels allows water vapor to penetrate the granule, dissolving a small portion of the solid fertilizer. The difference in vapor pressure across the coating drives this reaction, with condensation occurring in the spaces between the core and its coating. The time it takes to fill the internal space of the granule with sufficient water is a crucial factor contributing to the lag period. After achieving an equilibrium state, the size of the granule remains almost constant. The efficiency of chitosan-coated nano urea as a slow-release fertilizer was investigated and compared with commercial urea. The coated nano urea significantly enhanced soil properties, improved plant growth, and reduced nitrate leaching. Nano-fertilizers play a significant role in sustainable agriculture. The present work proposes a new formulation of coated nano urea for the slow and sustainable release of nitrogen. Bare nano urea (NU) and a nano urea/chitosan nanocomposite (NUCNC) were synthesized, and their structural, morphological, and thermal characteristics were studied using X-ray diffraction (XRD), Fourier-transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), and thermogravimetry (TGA & DTA). In addition, the protein content, free radical scavenging activity, and phenolic content were also found to be significantly higher in NUCNC-fertilized plants. The doses of the applied fertilizer have been observed to play an important role in plant growth. This study demonstrated that NUCNC could be potentially utilized as an alternative to commercial urea for sustainable agriculture.27–29 When enough saturated solution accumulates within the granule, it triggers a continuous release of nutrients. The release rate remains steady as long as the saturated liquid within the granule is in equilibrium with the solid fertilizers. The consistent saturation level keeps the pushing force for fertilizer transport constant due to the reduced concentration in the adjacent liquid mixture. The level of the internal liquid mixture drops as the bulk fertilizer at the center dissolves during the third release phase, referred to as the decay phase. In the decline phase, nutrient discharge decreases as the granule’s internal liquid level drops due to nutrient dissolution and water movement, reducing the driving force for release 29. Nutrient release from controlled-release nitrogen fertilizers (CRNFs) is mainly driven by four mechanisms: absorption, swelling, osmotic pressure, and diffusion.30 Kumar et al. described a starch-based polyvinyl alcohol (PVA) polymer designed for the controlled release of copper and zinc nanoparticles embedded in carbon nanofibers (CNF). In a pot experiment with chickpea (Cicer arietinum), when delivered via foliar application, this nanoscale additive fertilizer offered benefits such as better water absorption, increased water retention, improved water transport, cell wall extension, and soil stabilization.31
Mode, translocation, and accumulation of nano fertilizers
Nano fertilizers can be applied through various methods, including soil treatment, seed priming, foliar treatment, aeroponics, coating, emulsion, etc. Choosing the proper method for applying nano fertilizer is vital for vegetative development, as it varies according to nutrient availability, environmental factors, and soil characteristics. The uptake of nanoparticles depends on plant species, the size, chemical configuration, and concentration of the nanoparticles.32–35 (Fig. 3) Proper application of nanoparticles can increase nutrient utilization efficiency, boost harvest output, minimize environmental harm, and promote sustainable agricultural practices. Soil nutrient deficiency can be managed effectively by using NPs for seed treatment and soil application instead of foliar application.36
Foliar spray
Foliar spray involves directly applying liquid fertilizers onto the leaves or foliage of plants. The absorption of nanoparticles can occur through stomata, hydathodes, cuticle penetration, and wounds, although it depends on particle size. Foliar fertilization provides rapid plant utilization and requires less time than soil application to address the observed deficiencies. The NF applied through this mechanism enters mainly via the stomatal pore and moves either apoplastically or symplastically to vascular bundles for translocation in the plant. Foliar spray presents several benefits over soil application, such as quicker responses, reduced leaching and runoff, enhanced nutrient absorption, and stimulated plant growth.37 Studies have shown its effectiveness in boosting plant growth and yield. For instance, applying micronutrients like iron, zinc, and manganese via foliar spray has enhanced snap bean growth, increased yield, improved pod morphology, and enhanced nutritional quality.38 Another study found that the foliar application of copper nanoparticles onto tomatoes led to an 80% enhancement in fruit yield while a 30% reduction in copper use compared to traditional copper-based fungicides.39 However, the time of application is critical (morning or evening) as the opening of stomata during the foliar application is crucial for nutrient absorption. Overuse can damage plants, and multiple applications may be necessary, as they can be washed away by rain, or the plant must possess adequate leaf surface area for absorption. 36
Soil application
Soil application, the prevalent method, involves mixing nano fertilizers with soil, facilitating interaction with plant roots for absorption onto the root surface and penetration into root cells via endocytosis, lenticels, and biotransformation. Nano fertilizers can be associated with soil particles and microorganisms, changing their interactions and effectiveness. Controlled-release or slow-release fertilizers ensure a consistent nutrient supply, promoting plant growth and yield. While this approach is dependable and can yield long-term effects of nanoparticles, it can be costly and poses regulatory hurdles.40 Several studies have indicated that the gradual and sustainable release of nutrients like copper, zinc, and iron from fertilizers enhances nutrient use efficiency in tomato plants more effectively than commercial fertilizers, resulting in higher yields.11
Seed priming
Seed priming, a pre-sowing technique, modifies the physiological characteristics of seeds, facilitating seed sprouting and improving vegetative growth and plant maturation through the regulation of signaling and metabolic pathways. This method entails soaking seeds in nano fertilizers, significantly improving the outcomes while reducing the fertilizer application by half.41 Nano-biofertilizer penetrates seed pores, internally disperses, and activates plant hormones that stimulate growth, operating as a stimulant, increasing germination rate and seed development. A study by Nciizah et al, demonstrated that using low concentrations (e.g., 0.01% B) and longer priming durations of Zn, B, and Mo significantly enhanced germination percentage, reduced seedling emergence time by 94% and increased seedling biomass and chlorophyll content by up to 58%, enhancing maize crop productivity.42
Nano fertilizer applications in mitigating abiotic stresses: saline, drought, and heavy metal conditions
Crop yield is often constrained by the quality of irrigation water, which is typically evaluated based on parameters such as salinity, sodium adsorption ratio (SAR), and calcium, magnesium, and sodium levels. 43 Furthermore, the presence of heavy metals such as arsenic (As), cadmium (Cd), chromium (Cr), copper (Cu), lead (Pb), iron (Fe), manganese (Mn), and zinc (Zn) has a substantial impact on water quality.44 High concentrations of salts and heavy metals in irrigation water degrade water quality, leading to their accumulation in cultivated plants and agricultural soils.44 The widespread use of wastewater, which often contains toxins, for irrigation has further exacerbated the problem. This practice introduces contaminants into the food chain, raising long-term health concerns for humans and animals.
Saline conditions
Sol saline soil impacts approximately 950 million hectares of productive fields, with 43 million hectares of arable land experiencing anthropogenic soil salinization, resulting in a 50% decline in global agricultural output.45 The concentration of salts in irrigation water directly influences crop growth and yield. However, its effect on soil permeability is more complex (Fig.4); higher salinity in irrigation water enhances soil permeability. In contrast, increased sodium (Na) concentration reduces soil permeability in response to sodic and saline conditions. (Anon, n.d.). Excessively high sodium (Na) levels lead to soil sodicity, crust formation, increased runoff, erosion, and poor aeration. It also negatively affects the soil’s hydraulic properties, including percolation rate and electrical conductivity. The potential risk of sodicity from irrigation water has traditionally been evaluated using the Sodium Adsorption Ratio (SAR) (Eq. 1), which relies on the cation configuration of the water.
Here, C denotes the ionic concentration measured in millimoles of charge per litre, Na denotes sodium, calcium (Ca), or magnesium (Mg).43
Salinity alters several physiological processes in plants, such as lowering photosynthetic efficiency, disrupting stomatal conductance, impairing gas exchange, and affecting plasma membrane integrity. Additionally, it causes the generation of toxic oxidative species such as Superoxide anion (O₂⁻), Hydrogen peroxide (H₂O₂), singlet oxygen (¹O₂), and hydroxyl radicals (OH*), Peroxyl radicals (ROO•)47 Various materials, such as hydrogel, biochar, magnetic bentonite, and nanomaterials or nanoparticles, have been studied to mitigate these effects and improve poor water quality.48 Nanomaterials have been utilized to eliminate pollutants from polluted water. Applying copper nanoparticles (CuNPs) improved plant growth under salt stress by boosting growth parameters, gas exchange metrics, and photosynthetic compounds. In addition, the accumulation of osmolytes, hydrogen peroxide (H2O₂), malondialdehyde (MDA), total phenols, and flavonoids was decreased by CuNPs, which lessened oxidative damage. At the same time, they increased the activity of antioxidant enzymes.48 Amira et al 2015 applied Zinc and iron nanoparticles through a foliar spray with a standard Hoagland solution. There was a decrease in sodium and chloride ion levels and an enhancement in nitrogen, phosphorus, potassium, magnesium, manganese, iron, and zinc.45 Total chlorophyll, carotenoids, proline, carbohydrates, crude protein levels, and non-enzymatic and enzymatic antioxidants were also elevated. Various studies show that ZnO nanoparticles increase the antioxidant enzyme activity (superoxide dismutase (SOD), ascorbate peroxidase (APX), and glutathione reductase (GR) system), and nutrient absorption.49
Drought stress conditions
Drought is one of the most significant abiotic stresses that negatively affect crop plants, causing a reduction in seed development, lower yields, and decreased nutritional quality.50 More than 40% of the global population faces drought stress annually, and by 2030, 7 billion people may be forced to migrate from arid regions. Due to drought-induced stress, plants experience oxidative stress and other physiological and biochemical reactions. Plants employ various mechanisms to cope with drought, including producing oxidative species, synthesizing stress hormones such as abscisic acid and ethylene, changes in root and shoot structure, stomatal closure, and reduced chlorophyll production.51,52 These responses lead to both short-term and long-term reactions. When the stress is short-lived, the plant’s responses are likewise temporary, including decreased carbon fixation, stomatal reduction, disrupted osmotic balance, growth, and water movement alterations, and the transmission of signals indicating cell dehydration. These short-term responses typically do not cause lasting damage if normal conditions are restored quickly. However, during prolonged drought stress, irreversible changes occur in the plant, ultimately leading to death.53 The primary ROS include superoxide anion radicals, hydroxyl radicals, hydrogen peroxide, and singlet oxygen. These molecules can harm cellular membranes, disrupt photosynthesis, and impair electron transfer in chloroplasts and mitochondria.50 The resulting disturbances under such unfavorable conditions cause a decline in crop yields. Plants use their antioxidant defense systems to scavenge the damaging reactive oxygen species (ROS) in response to these stressors.54 The severity of the water deficit, the plant’s developmental stage, and its genotype affect how strongly the plant defends itself.55 Antioxidant enzymes such as ascorbate peroxidase (APX), catalase (CAT), and peroxidase (POX) are part of the plant’s defense mechanism. Additionally, it contains suitable solutes such as proline and glycine betaine, as well as antioxidants like glutathione, α-tocopherols, and phenols.56 Samota et al, 2024 demonstrated that seed priming with iron (Fe), zinc (Zn), and methyl jasmonate (MeJA) under drought stress significantly enhanced several biochemical and physiological responses in plants: carotenoids (10–19%), abscisic acid (18–50%), proline (60–80%), superoxide dismutase activities (27–62%), ascorbate peroxidase (46–61%), catalase (50–80%), and NADPH oxidase (16–30%). When copper (Cu) and zinc (Zn) nanoparticles (NPs) are applied to wheat plants, oxidative stress defense enzyme activity is increased. The water retention capacity lowers the levels of thiobarbituric acid, influences the precipitation of reagents, stabilizes the levels of light-absorbing pigment in the leaves, and lessens the effects of stress.57 Furthermore, under dry conditions, Cu NPs applied to maize have been demonstrated to raise anthocyanin, chlorophyll, carotenoid, plant biomass, and leaf water content.58 The study by Ghani et al. increased the antioxidant defense system, nutrient absorption, and osmolyte accumulation in cucumber seedlings.59 Reduction in ROS lipid peroxidation is observed in ZnO NP-treated seedlings. In addition, the foliar spray of Zn and Fe nanoparticles increases antioxidant enzyme activity, proline and sugar content, and leaf size, and improves Pinto beans’ seed quality.60 Under drought stress conditions, applying foliar nano iron oxide enhances soybean seed production by 40.12 and 32.60% in water deficit conditions, increasing the oil content of seeds by 61%.
Heavy metal stress
Heavy metal (HM) strain impacts plants’ physiological and biochemical properties. Several approaches are employed to mitigate this stress in plants. These include:
Minimizing the concentration of bioavailable heavy metals in the topsoil.62
Regulating the functioning of genes related to the transport of heavy metals in plants.63
Strengthening the plant’s Oxidative stress defense system and optimizing physiological processes.64
Stimulating the synthesis of protective compounds like organic acids, metal-binding peptides, and rhizome secretion. 63,65
Nanoparticles (NPs) in the soil can absorb and transform heavy metals (HMs), reducing their movement and bioavailability. For instance, Fe3 O4 nanoparticles have been shown to reduce the transferability of cadmium and other HMs in the soil. Additionally, specific nanoparticles can enhance soil properties; for instance, hydroxyapatite nanoparticles can discharge phosphate and raise soil hydrogen ion concentration, thereby mitigating the detrimental impacts of heavy metals in the soil 66. The plant is protected by the apoplastic transport barriers in the rhizosphere, which regulate the flow of oxygen, cations, anions, and water 63. Heavy metal (HM) aggregation in the underground root may be restricted by the influence of nanoparticles (NPs) on the development of these barriers 67. However, as plant roots also have a variety of ion and protein channels within the cell wall that can simultaneously transport heavy metals, more than apoplastic polymeric barriers may be required to reduce HM stress. Additionally, specific NPs can transport and regulate metal in plants, strengthening the cell wall barriers to block HM entry. Most NPs build up in cell walls, forming compounds with metals to render them inert. These complexes stick to the cell’s surface 64, inhibiting the movement of heavy metals (HMs) within the plant and diminishing their biochemical function. Furthermore, natural acids that build up in root structure and foliage cell walls can chelate heavy metals, alleviating the detrimental impacts of stress. MNPs have also been shown to boost the production of protective structural agents.
An alternative strategy for alleviating heavy metal (HM) stress involves stimulating the Plant antioxidant defense system 68. Plants naturally produce Oxidative species through specific metabolic processes. For instance, ROS are continuously generated in chloroplasts and other cellular regions during respiration and photosynthesis. At low concentrations, ROS function as signaling molecules that contribute to growth, development, and defense 68. However, excessive accumulation of reactive oxygen species (ROS) can adversely affect cell walls, cytoplasm, mitochondria, chloroplast proteins, and other cellular structures under stress conditions. Plants utilize essential free radical-scavenging enzymes, including superoxide dismutase (SOD), peroxidase (POD), glutathione reductase (GR), ascorbate peroxidase (APX), catalase (CAT), and monodehydroascorbate reductase (MDHAR), to counteract ROS. Furthermore, ROS are scavenged by low-molecular-weight, non-catalytic substances such as ascorbic acid and tocopherol and carotenoids, polyphenols, and flavonoids69,70. Stress triggers the creation of shikimate-phenylpropanoid and the metabolism of galactose, alanine, aspartic acid, and ascorbate, among other metabolic pathways involved in ROS detoxification, which contribute to reducing oxidative stress in plants. In their 2019 study, Rizwan et al investigated how foliar application of ZnO nanoparticles (NPs) affected Zea mays’s cadmium (Cd) absorption and antioxidant enzyme activity. Their results demonstrated that ZnO NPs applied at 50, 75, and 100 mg/L concentrations significantly reduced Cd uptake in roots by 18%, 33%, and 53%, and in maize shoots by about 12%, 23%, and 61%, respectively. Additionally, ZnO NPs enhanced biomass in the roots and shoots of maize seedlings and elevated antioxidant enzyme activity 71.
Current and future outlook of nano fertilizer
After the 1960s, food grain production increased due to synthetic fertilizers and pesticides, severely affecting the environment. We must ensure that nano fertilizer decreases environmental damage rather than increases it. 72. Nano fertilizer holds the potential to enhance nutrient delivery to plants, thereby increasing yield production while mitigating the environmental drawbacks associated with chemical fertilizers. Diverse studies have investigated nanomaterials like nano chitosan and metal oxide nanoparticles, revealing improved soil nutrient absorption rate and retention capacity. Considerable advancements have been made in developing controlled-release fertilizers, which release nutrients gradually, thus reducing the need for frequent application.
Future studies on nano fertilizers should prioritize creating cost-effective and environmentally friendly methods for synthesizing nanomaterials, optimizing their physical and chemical properties, and decreasing the risk associated with their use. We need eco-friendly innovations and artificial intelligence technologies to create durable, decomposable, and future-oriented sustainable products. These technologies would significantly enhance natural resource applications and lessen environmental impact. 73 Nano sensors in Nano fertilizer could allow real-time monitoring, inform the right time to apply NFs, and decrease nutrient waste. 74,75. Precision agriculture incorporates drones equipped with multispectral cameras to capture images that detect nutrient levels in the soil. This technology enables farmers to optimize resource usage by avoiding over-applying nutrients to their crops.74,76
Furthermore, it is imperative for research efforts to comprehend the environmental and human health implications of nano fertilizers. Simultaneously, there is a need to establish a regulatory framework and standardized protocols to enhance their safety and appropriate utilization. 77 Research is progressing to form Nanocomposites to provide nutrients through intelligent delivery systems, which could aid in balancing the crop’s nutrient supply. 23. Research is required on how nutrients are delivered in the plant system and the effects and eventual destiny of nano fertilizer in the environment. 78.
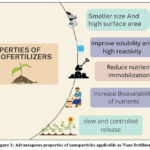 |
Figure 1: Advantageous properties of nanoparticles applicable as Nano fertilizers |
 |
Figure 2: Comparison between Conventional Fertilizers and nano fertilizers |
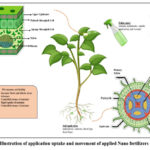 |
Figure 3: Illustration of application uptake and movement of applied Nano fertilizers (NFs) |
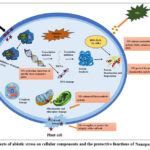 |
Figure 4: Effects of abiotic stress on cellular components and the protective functions of Nanoparticles (NPS) |
Table 1: Micronutrient deficiencies in Indian soils
| Micronutrient | Overall Deficiency (%) | Regions with Severe Deficiency (>50%) | Regions with Moderate Deficiency (30-50%) | Regions with Low Deficiency (<20%) | Contributing Factors |
| Zinc (Zn) | 36.5% | Rajasthan, Gujarat, Haryana, Punjab | Maharashtra, Telangana, Andhra Pradesh | Eastern states | Coarse-textured soils, high pH (>8.5), calcareous and sodic soils, low organic carbon |
| Iron (Fe) | 12.8% | Karnataka, Tamil Nadu, Maharashtra, Gujarat, Rajasthan | Haryana, Punjab | Eastern and northeastern regions | Alkaline soils (pH >7.5), calcareous, aerobic soils, high redox potential |
| Copper (Cu) | 4.2% | Southernmost regions of India | Southern and Western states | Tamil Nadu, Uttar Pradesh, Haryana, Punjab | Intensive farming practices, alkaline, calcareous soil, low organic carbon |
| Boron (B) | Varies regionally | Bihar, Gujarat, West Bengal, Odisha, Jharkhand | Madhya Pradesh, Maharashtra | Northwestern and some central areas | Highly calcareous and acidic soils, leaching in sandy loam soils |
Table 2: Role of key micronutrients in plants and the potential improvements achieved by using nano-fertilizers (NFs).
| Micronutrient | Role of elements | Improvement in plant growth by NFs | Physiological Toxicity | Reference |
| Zinc | · Activates enzymes (Carbonic dehydrogenase, Phosphorylase, Carboxylase)· Helps in tryptophan synthesis and precursor for auxin production | · Reduced reactive oxygen species and chlorophyll fluorescence activity· Improved photosynthetic rate, high chlorophyll and carotenoid content
· Increased level of protein and carbohydrates. |
Excess Zn enhances Fe deficiency. | 15,40,79–83 |
| Copper | · Associated with enzymes. Example – Lactase, oxidase· Synthesis of lignin
· Required for synthesis of vitamin C (Ascorbic acid), which controls cell division, elongation, and differentiation. |
· Increase antioxidant enzymes like oxidase, catalase, peroxidase and Superoxide Dimutase), and decrease malondialdehyde content.· Mitigate oxidative stress
· Promote stomata closure, which reduces water evaporation |
Growth inhibitionphotosynthesis inferences
Increase oxidative stress, chlorosis |
22,58,81,84 |
| Iron | · Constituents of enzymes like peroxidase and catalase (Present in peroxisomes) help in the formation and dissociation of peroxidase· Constituent of ferredoxin and cytochrome (a protein essential for electron transfer)
· Help in chlorophyll synthesis, energy production, Nitrogen reduction, and biological N2 fixation. |
· Increase in the level of photosynthesis· Increase physiological parameters, including root growth and antioxidant enzymes such as catalase, peroxidase, and SOD, polyphenyl oxidase. | Bronzing,Stunted root system,
Deficiency of other micronutrients |
85–89 |
| Manganese | · Helps in the production of chlorophyll during photosynthesis (transferring electrons through chlorophyll to convert CO2 into carbohydrates and release O2 from H2O)· Promotes faster seed germination and hastens the maturation process.
· Increase availability of P and Ca |
· Increased synthesis of flavonoids, amino acids, and lignin· Increase in the efficiency of herbicides and pesticides | Causing oxidative stress, chlorosis in leaves (interveinal and marginal)Necrotic leaf spot | 90–92 |
Table 3: Shows the comparative effects of nanoparticle-based fertilizers on plant growth, stress tolerance, and yield across different plant species and environmental conditions.
| Fertilizer composition | Method Used | Particle size |
Plant culture |
Stress | Experimental conditions | Experimental Result | References |
| Fe, Zn, and Mg | Green Synthesis | Zno-9.5nm,Fe2O3- 7.8nm and MnO-10.5nm | Cabbage and lupin plants | —– | Foliar Spray | Lupin Plants- 6% height increase, 19% root size increase, 3.5% increase in chlorophyll content, 300% increase in leaf area.For cabbage, root size increased by 10.3%, dry biomass increased by 55.1%, chlorophyll content increased by 7.1%, and leaf area increased by 25.6%. | 93 |
| Zn | Chemical method | ZnO (50 nm) | Cucumber | Drought | Foliar Spray | The antioxidant defense system is increased, and nutrient absorption and osmolytes accumulate.Reduction in ROS, lipid peroxidation | 24 |
| Zn | Chemical method | ZnO (30 nm) | Cotton | Drought | Foliar spray | Increase plant growth and yield, and increase antioxidant enzyme activity | 94 |
| Fe | Chemical method | Fe3O4 (50–100 nm) | Soyabean | Drought | foliar Spray | 40.12% and 32.60% increase seed yield under drought stress conditions,increased the oil content of seeds by 10.14 and 7.87% | 61 |
| Zn and Fe | Green Synthesis | Less than 100nm | Pinto bean | Drought | foliar Spray | Increase antioxidant activity, proline content, soluble sugar, and leaf areaImproved seed quality | 60 |
| Cu and Se | Chemical Reduction meth | SeNPs and CuNPs (5–18 and 35–42 nm), respectively, | Strawberry | Drought | Foliar Spray | Improving the growth, quality, and physiological characteristics | 95 |
| Zn and Fe | Chemical method | ZnO and Fe3O4 (30 nm and ranging from 10 to 30 nm) | Wheat (Triticum aestivum) | Drought | Foliar Spray | NPs increased the proline, soluble sugars, and enzyme activities, and 88% increased grain yield. | 96 |
| Zn and Fe | Chemical method | Less than 100nm | Dragonhead (Dracocephalum moldavica) | Drought | Foliar Spray | Enhanced level of total chlorophyll content, above-ground plant biomass, flavonoid, and anthocyanin.Oil production and quality improved. | 97 |
| Cu and Se | Green Synthesis | 100–300 nm for Se and 350–500 nm for CuO | Tomato | Salinity | Foliar Spray | Increased tomato output (2.07 kg/plant), 9.24% increase in soluble solids contentDecrease in Enzymatic antioxidants and soil biological activity | 98 |
| Cu | Green Synthesis | 9-18nm | Barley | Salinity | Seed Priming | Improved plant growth under salt stress, photosynthetic pigments, and gas exchange parameters | 48 |
| Polyvinylpyrrolidone-coated Cu Nps | Chemical Reduction Method | 420 nm | Wheat | Salinity | Foliar Spray | Reduced oxidative Stressenhanced the activity of antioxidant enzymes in stress conditions | 99 |
| Zn and Cu | Green Synthesis | Less than 100nm | Basil plant | Salinity | Foliar Spray | Significant effect on chlorophyll content,highest antioxidant activity and
increase flavonoid and phenolic levels. |
100 |
| Zn and Fe | Chemical Precipitation Method | ZnO (10-15 nm) and Fe3O4 (10 to 12 nm) | Moringa peregrine | Salinity | Foliar Spray | Decrease in sodium and chloride levels and increase the N, P, K⁺, Mg²⁺, Mn²⁺, Fe, and Zn concentrations and carotenoid levels.Higher total chlorophyll content, proline, carbohydrate levels, protein levels, and activity of non-enzymatic antioxidants | 45 |
| Zn | Chemical Method | 68.1 nm | Rice | Heavy Metal (As, Cd, And Micronutrients) | Foliar Spray | Highest excretion of root exudates and the most significant arsenic (As) uptake reduction.Enhancement of iron (Fe) and copper (Cu) uptake | 101 |
| S and Cu | Chemical Method | 37-61nm | oilseed rape | Heavy Metal (Cu) | Seed Priming | Increased shoot and root length and dry weight by up to 28.2% | 102 |
| Fe | Chemical co-precipitation method | Fe3O4 | Coriander | Cd and Pb toxicity | Seed Priming | Increase antibacterial activity | 103 |
| Zn | Green synthesis | (450 ppm and 600 ppm) | Maize (Zea mays) | Heavy Metal (Cd) | Foliar Spray | Reduction in plant height (35.24%), biomass (43.86%), and yield (37.62%) due to cadmium exposure) | 9 |
| Zn | Chemical Synthesis | Less than 100 nm | Rice and Serendipity Indica | Heavy Meta (Cd) | Seed Priming | Increase Antioxidant activities, specifically peroxidase (61.5 and 122.5% in Yi801 and Hz43 roots, respectively) | 104 |
| Fe, Zn, and Cu | Chemical Synthesis | Less than 100 nm | Wheat (Fayyad cultivar) and Dijla Al-Khair) | heavy metals | Foliar Spray | Increased tiller count, plant height, grain weight, yield, and biological yield | 105 |
Conclusions
Nanotechnology is transforming modern agriculture by addressing critical challenges such as nutrient deficiencies, soil degradation, and environmental sustainability. Micronutrient-based nano fertilizers, mainly those containing zinc, copper, and iron, exhibit superior efficacy in enhancing crop productivity, nutrient uptake, and stress resilience compared to conventional fertilizers. Their controlled-release mechanisms significantly reduce nutrient losses, enhance soil health, and mitigate the environmental impact of excessive fertilizer. Nano fertilizers also play a pivotal role in alleviating abiotic stress conditions, including drought, salinity, and heavy metal toxicity. These fertilizers enhance plant performance under adverse environmental conditions by optimizing nutrient absorption and improving physiological tolerance mechanisms. Their ability to modulate antioxidant activity, regulate osmolyte accumulation, and enhance enzymatic functions underscores their significance in stress management and sustainable agriculture. Despite the promising potential of nano fertilizers, further research is required to comprehensively assess their long-term environmental impacts, regulatory implications, and economic feasibility. The integration of precision agriculture methodologies, including nano sensors and AI-driven monitoring systems, will improve the efficiency of nano fertilizer applications and maximize their agronomic benefits. Future investigations should focus on developing safe and sustainable nano-fertilizer formulations to facilitate their role in advancing resilient and productive agricultural systems. By leveraging nanotechnology, it is possible to establish sustainable farming practices that address the increasing global food demand while minimizing ecological consequences.
Acknowledgment
The author would like to formally acknowledge the resources and infrastructure provided by Maharshi Dayanand University. Moreover, we thank the university for awarding Ms Manisha a university Research Scholarship.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Author Contribution
Manisha Lohchab: Major contribution in writing the manuscript, collection of data, and reviewing the research paper
Vaishali Arora: She contributed significantly to editing the manuscript.
Babita Khosla: She supervised manuscript development with insightful contributions and made the final edit to improve the quality of the manuscript.
References
- FAO. The Future of Food and Agriculture: Trends and Challenges; Food and Agriculture Organization of the United Nations: Rome, 2017.
- Agricultural-Statistics-at-a-Glance-2022.Pdf.
- Raiesi Ardali, T.; Ma’mani, L.; Chorom, M.; Motamedi, E.; Fathi Gharebaba, M. A Biocompatible NPK+Fe+Zn Slow Release Fertilizer: Synthesis and Its Evaluation in Tomato Plant Growth Improvement. Sci Rep 2024, 14 (1), 4640. https://doi.org/10.1038/s41598-024-55152-z.
CrossRef - Abdel Latef, A. A. H.; Abu Alhmad, M. F.; Abdelfattah, K. E. The Possible Roles of Priming with ZnO Nanoparticles in Mitigation of Salinity Stress in Lupine (Lupinus Termis) Plants. J Plant Growth Regul 2017, 36 (1), 60–70. https://doi.org/10.1007/s00344-016-9618-x.
CrossRef - Khan, S. T.; Malik, A.; Alwarthan, A.; Shaik, M. R. The Enormity of the Zinc Deficiency Problem and Available Solutions; an Overview. Arabian Journal of Chemistry 2022, 15 (3), 103668. https://doi.org/10.1016/j.arabjc.2021.103668.
CrossRef - Singhal, V. K. Addressing Micronutrient Deficiencies in Indian Soil: Challenges and Strategies. Int. J. Res. Agron. 2024, 7 (1S), 225–229. https://doi.org/10.33545/2618060X.2024.v7.i1Sc.801.
CrossRef - Shukla, A. K.; Malik, R. S.; Tiwari, P. K.; Prakash, C.; Behera, S. K.; Yadav, H.; Narwal, R. P. Status of Micronutrient Deficiencies in Soils of Haryana. 2015.
- Ekanayake, S. A.; Godakumbura, P. I. Synthesis of a Dual-Functional Nanofertilizer by Embedding ZnO and CuO Nanoparticles on an Alginate-Based Hydrogel. ACS Omega 2021, 6 (40), 26262–26272. https://doi.org/10.1021/acsomega.1c03271.
CrossRef - Ahmed, R.; Zia-ur-Rehman, M.; Sabir, M.; Usman, M.; Rizwan, M.; Ahmad, Z.; Alharby, H. F.; Al-Zahrani, H. S.; Alsamadany, H.; Aldhebiani, A. Y.; Alzahrani, Y. M.; Bamagoos, A. A. Differential Response of Nano Zinc Sulphate with Other Conventional Sources of Zn in Mitigating Salinity Stress in Rice Grown on Saline-Sodic Soil. Chemosphere 2023, 327, 138479. https://doi.org/10.1016/j.chemosphere.2023.138479.
CrossRef - Bandyopadhyay, S.; Ghosh, K.; Varadachari, C. Multimicronutrient Slow-Release Fertilizer of Zinc, Iron, Manganese, and Copper. International Journal of Chemical Engineering 2014, 2014, 1–7. https://doi.org/10.1155/2014/327153.
CrossRef - Tarafder, C.; Daizy, M.; Alam, Md. M.; Ali, Md. R.; Islam, Md. J.; Islam, R.; Ahommed, Md. S.; Aly Saad Aly, M.; Khan, Md. Z. H. Formulation of a Hybrid Nanofertilizer for Slow and Sustainable Release of Micronutrients. ACS Omega 2020, 5 (37), 23960–23966. https://doi.org/10.1021/acsomega.0c03233.
CrossRef
- Monreal, C. M.; DeRosa, M.; Mallubhotla, S. C.; Bindraban, P. S.; Dimkpa, C. Nanotechnologies for Increasing the Crop Use Efficiency of Fertilizer-Micronutrients. Biol Fertil Soils 2016, 52 (3), 423–437. https://doi.org/10.1007/s00374-015-1073-5.
CrossRef
- Guo, S.; Hu, X.; Wang, Z.; Yu, F.; Hou, X.; Xing, B. Zinc Oxide Nanoparticles Cooperate with the Phyllosphere to Promote Grain Yield and Nutritional Quality of Rice under Heatwave Stress. Proc. Natl. Acad. Sci. U.S.A. 2024, 121 (46), e2414822121. https://doi.org/10.1073/pnas.2414822121.
CrossRef
- Semenova, N. A.; Burmistrov, D. E.; Shumeyko, S. A.; Gudkov, S. V. Fertilizers Based on Nanoparticles as Sources of Macro- and Microelements for Plant Crop Growth: A Review. Agronomy 2024, 14 (8), 1646. https://doi.org/10.3390/agronomy14081646.
CrossRef
- Raliya, R.; Tarafdar, J. C. ZnO Nanoparticle Biosynthesis and Its Effect on Phosphorous-Mobilizing Enzyme Secretion and Gum Contents in Clusterbean (Cyamopsis Tetragonoloba L.). Agric Res 2013, 2 (1), 48–57. https://doi.org/10.1007/s40003-012-0049-z.
CrossRef
- P. Kale, A.; N. Gawade, S. STUDIES ON NANOPARTICLE INDUCED NUTRIENT USE EFICIENCY OF FERTILIZER AND CROP PRODUCTIVITY. gctl 2016, 2 (2), 88–92. https://doi.org/10.18510/gctl.2016. 226.
CrossRef
- Panichikkal, J.; Thomas, R.; John, J. C.; Radhakrishnan, E. K. Biogenic Gold Nanoparticle Supplementation to Plant Beneficial Pseudomonas Monteilii Was Found to Enhance Its Plant Probiotic Effect. Curr Microbiol 2019, 76 (4), 503–509. https://doi.org/10.1007/s00284-019-01649-0.
CrossRef
- Congreves, K. A.; Van Eerd, L. L. Nitrogen Cycling and Management in Intensive Horticultural Systems. Nutr Cycl Agroecosyst 2015, 102 (3), 299–318. https://doi.org/10.1007/s10705-015-9704-7.
CrossRef
- Vaseghi, Z.; Nematollahzadeh, A. Nanomaterials: Types, Synthesis, and Characterization. In Green Synthesis of Nanomaterials for Bioenergy Applications; Srivastava, N., Srivastava, M., Mishra, P. K., Gupta, V. K., Eds.; Wiley, 2020; pp 23–82. https://doi.org/10.1002/9781119576785.ch2.
CrossRef
- Chhipa, H. Nanofertilizers and Nanopesticides for Agriculture. Environ Chem Lett 2017, 15 (1), 15–22. https://doi.org/10.1007/s10311-016-0600-4.
CrossRef
- Anguissola Scotti, I.; Silva, S.; Botteschi, G. Effect of Fly Ash on the Availability of Zn, Cu, Ni and Cd to Chicory. Agriculture, Ecosystems & Environment 1999, 72 (2), 159–163. https://doi.org/10.1016/ S0167-8809(98)00170-4.
CrossRef
- Singh, M. V. Micronutrient Deficiencies in Crops and Soils in India. In Micronutrient Deficiencies in Global Crop Production; Alloway, B. J., Ed.; Springer Netherlands: Dordrecht, 2008; pp 93–125. https://doi.org/10.1007/978-1-4020-6860-7_4.
CrossRef
- Choudhary, R. C.; Kumaraswamy, R. V.; Kumari, S.; Pal, A.; Raliya, R.; Biswas, P.; Saharan, V. Synthesis, Characterization, and Application of Chitosan Nanomaterials Loaded with Zinc and Copper for Plant Growth and Protection. In Nanotechnology; Prasad, R., Kumar, M., Kumar, V., Eds.; Springer Singapore: Singapore, 2017; pp 227–247. https://doi.org/10.1007/978-981-10-4573-8_10.
CrossRef
- Ghani, M. I.; Saleem, S.; Rather, S. A.; Rehmani, M. S.; Alamri, S.; Rajput, V. D.; Kalaji, H. M.; Saleem, N.; Sial, T. A.; Liu, M. Foliar Application of Zinc Oxide Nanoparticles: An Effective Strategy to Mitigate Drought Stress in Cucumber Seedling by Modulating Antioxidant Defense System and Osmolytes Accumulation. Chemosphere 2022, 289, 133202. https://doi.org/10.1016/ j.chemosphere. 2021. 133202.
CrossRef
- Ghasemi, S.; Khoshgoftarmanesh, A. H.; Hadadzadeh, H.; Jafari, M. Synthesis of Iron-Amino Acid Chelates and Evaluation of Their Efficacy as Iron Source and Growth Stimulator for Tomato in Nutrient Solution Culture. J Plant Growth Regul 2012, 31 (4), 498–508. https://doi.org/10.1007/ s00344-012-9259-7.
CrossRef
- Rico, C. M.; Majumdar, S.; Duarte-Gardea, M.; Peralta-Videa, J. R.; Gardea-Torresdey, J. L. Interaction of Nanoparticles with Edible Plants and Their Possible Implications in the Food Chain. J. Agric. Food Chem. 2011, 59 (8), 3485–3498. https://doi.org/10.1021/jf104517j.
CrossRef
- Sharma, A.; Kumar, S.; Singh, R. Synthesis and Characterization of a Novel Slow-Release Nanourea/Chitosan Nanocomposite and Its Effect on Vigna Radiata L. Environ. Sci.: Nano 2022, 9 (11), 4177–4189. https://doi.org/10.1039/D2EN00297C.
CrossRef
- Liang, R.; Liu, M.; Wu, L. Controlled Release NPK Compound Fertilizer with the Function of Water Retention. Reactive and Functional Polymers 2007, 67 (9), 769–779. https://doi.org/10.1016/ j.reactfunctpolym.2006.12.007.
CrossRef
- Shaviv, A.; Raban, S.; Zaidel, E. Modeling Controlled Nutrient Release from Polymer Coated Fertilizers: Diffusion Release from Single Granules. Environ. Sci. Technol. 2003, 37 (10), 2251–2256. https://doi.org/10.1021/es011462v.
CrossRef
- Khadiran, N. F.; Hussein, M. Z.; Ahmad, R.; Khadiran, T.; Zainal, Z.; Kadir, W. R. W. A.; Hashim, S. S. Preparation and Properties of Zinc Layered Hydroxide with Nitrate and Phosphate as the Counter Anion, a Novel Control Release Fertilizer Formulation. J Porous Mater 2021, 28 (6), 1797–1811. https://doi.org/10.1007/s10934-021-01122-z.
CrossRef
- Kumar, R.; Ashfaq, M.; Verma, N. Synthesis of Novel PVA–Starch Formulation-Supported Cu–Zn Nanoparticle Carrying Carbon Nanofibers as a Nanofertilizer: Controlled Release of Micronutrients. J Mater Sci 2018, 53 (10), 7150–7164. https://doi.org/10.1007/s10853-018-2107-9.
CrossRef
- Gajjar, P.; Pettee, B.; Britt, D. W.; Huang, W.; Johnson, W. P.; Anderson, A. J. Antimicrobial Activities of Commercial Nanoparticles against an Environmental Soil Microbe, Pseudomonas Putida KT2440. J Biol Eng 2009, 3 (1), 9. https://doi.org/10.1186/1754-1611-3-9.
CrossRef
- Collins, D.; Luxton, T.; Kumar, N.; Shah, S.; Walker, V. K.; Shah, V. Assessing the Impact of Copper and Zinc Oxide Nanoparticles on Soil: A Field Study. PLoS ONE 2012, 7 (8), e42663. https://doi.org/10.1371/journal.pone.0042663.
CrossRef
- Pallavi; Mehta, C. M.; Srivastava, R.; Arora, S.; Sharma, A. K. Impact Assessment of Silver Nanoparticles on Plant Growth and Soil Bacterial Diversity. 3 Biotech 2016, 6 (2), 254. https://doi.org/10.1007/s13205-016-0567-7.
CrossRef
- Cao, J.; Feng, Y.; Lin, X.; Wang, J. Arbuscular Mycorrhizal Fungi Alleviate the Negative Effects of Iron Oxide Nanoparticles on Bacterial Community in Rhizospheric Soils. Front. Environ. Sci. 2016, 4. https://doi.org/10.3389/fenvs.2016.00010.
CrossRef
- Fageria, N. K.; Filho, M. P. B.; Moreira, A.; Guimarães, C. M. Foliar Fertilization of Crop Plants. Journal of Plant Nutrition 2009, 32 (6), 1044–1064. https://doi.org/10.1080/01904160902872826.
CrossRef
- Fernández, V.; Sotiropoulos, T.; Brown, P. H. Foliar Fertilization: Scientific Principles and Field Pratices; International fertilizer industry association: Paris, 2013.
- Marzouk, N. M.; Abd-Alrahman, H. A.; EL-Tanahy, A. M. M.; Mahmoud, S. H. Impact of Foliar Spraying of Nano Micronutrient Fertilizers on the Growth, Yield, Physical Quality, and Nutritional Value of Two Snap Bean Cultivars in Sandy Soils. Bull Natl Res Cent 2019, 43 (1), 84. https://doi.org/10.1186/ s42269-019-0127-5.
CrossRef
- Lopez-Lima, D.; Mtz-Enriquez, A. I.; Carrión, G.; Basurto-Cereceda, S.; Pariona, N. The Bifunctional Role of Copper Nanoparticles in Tomato: Effective Treatment for Fusarium Wilt and Plant Growth Promoter. Scientia Horticulturae 2021, 277, 109810. https://doi.org/10.1016/j.scienta.2020.109810.
CrossRef
- Raliya, R.; Tarafdar, J. C.; Biswas, P. Enhancing the Mobilization of Native Phosphorus in the Mung Bean Rhizosphere Using ZnO Nanoparticles Synthesized by Soil Fungi. J. Agric. Food Chem. 2016, 64 (16), 3111–3118. https://doi.org/10.1021/acs.jafc.5b05224.
CrossRef
- Do Espirito Santo Pereira, A.; Caixeta Oliveira, H.; Fernandes Fraceto, L.; Santaella, C. Nanotechnology Potential in Seed Priming for Sustainable Agriculture. Nanomaterials 2021, 11 (2), 267. https://doi.org/10.3390/nano11020267.
CrossRef
- Nciizah, A. D.; Rapetsoa, M. C.; Wakindiki, I. IC.; Zerizghy, M. G. Micronutrient Seed Priming Improves Maize (Zea Mays) Early Seedling Growth in a Micronutrient Deficient Soil. Heliyon 2020, 6 (8). https://doi.org/10.1016/j.heliyon.2020.e04766.
CrossRef
- Qadir, M.; Sposito, G.; Smith, C. J.; Oster, J. D. Reassessing Irrigation Water Quality Guidelines for Sodicity Hazard. Agricultural Water Management 2021, 255, 107054. https://doi.org/10.1016/ j.agwat.2021.107054.
CrossRef
- Zakir, H. M.; Sharmin, S.; Akter, A.; Rahman, Md. S. Assessment of Health Risk of Heavy Metals and Water Quality Indices for Irrigation and Drinking Suitability of Waters: A Case Study of Jamalpur Sadar Area, Bangladesh. Environmental Advances 2020, 2, 100005. https://doi.org/10.1016/ j.envadv.2020.100005.
CrossRef
- Amira, S. S.; Souad, A. E. F.; Essam, D. Alleviation of Salt Stress on Moringa Peregrina Using Foliar Application of Nanofertilizers. J. Hortic. For. 2015, 7 (2), 36–47. https://doi.org/10.5897/ JHF2014.0379.
CrossRef
- Response_of_Soils_to_Sodic_and_Saline_Conditions.Pdf.
- Negacz, K.; Vellinga, P.; Barrett-Lennard, E.; Choukr-Allah, R.; Elzenga, T. Future of Sustainable Agriculture in Saline Environments, 1st ed.; CRC Press: Boca Raton, 2021. https://doi.org/ 10.1201/9781003112327.
CrossRef
- Shaikhaldein, H. O.; Al-Qurainy, F.; Nadeem, M.; Khan, S.; Tarroum, M.; Salih, A. M.; Al-Hashimi, A. Biosynthesis of Copper Nanoparticles Using Solenostemma Argel and Their Effect on Enhancing Salt Tolerance in Barley Plants. Sci Rep 2024, 14 (1), 12701. https://doi.org/10.1038/s41598-024-63641-4.
CrossRef
- Suleiman, M.; Al-Masri, M.; Ali, A. A.; Aref, D.; Hussein, A.; Saadeddin, I.; Warad, I. Synthesis of Nano-Sized Sulfur Nanoparticles and Their Antibacterial Activities. 2015.
- Chaves, M. M.; Flexas, J.; Pinheiro, C. Photosynthesis under Drought and Salt Stress: Regulation Mechanisms from Whole Plant to Cell. Annals of Botany 2009, 103 (4), 551–560. https://doi.org/10.1093/aob/mcn125.
CrossRef
- Bhat, M. A.; Mir, R. A.; Kumar, V.; Shah, A. A.; Zargar, S. M.; Rahman, S.; Jan, A. T. Mechanistic Insights of CRISPR /Cas‐mediated Genome Editing towards Enhancing Abiotic Stress Tolerance in Plants. Physiologia Plantarum 2021, 172 (2), 1255–1268. https://doi.org/10.1111/ppl.13359.
CrossRef
- Chiappero, J.; Cappellari, L. D. R.; Sosa Alderete, L. G.; Palermo, T. B.; Banchio, E. Plant Growth Promoting Rhizobacteria Improve the Antioxidant Status in Mentha Piperita Grown under Drought Stress Leading to an Enhancement of Plant Growth and Total Phenolic Content. Industrial Crops and Products 2019, 139, 111553. https://doi.org/10.1016/j.indcrop.2019.111553.
CrossRef
- Kaur, G.; Asthir, B. Molecular Responses to Drought Stress in Plants. Biologia plant. 2017, 61 (2), 201–209. https://doi.org/10.1007/s10535-016-0700-9.
CrossRef
- Mittler, R. Abiotic Stress, the Field Environment and Stress Combination. Trends in Plant Science 2006, 11 (1), 15–19. https://doi.org/10.1016/j.tplants.2005.11.002.
CrossRef
- Nuñez Barrios, A.; Hoogenboom, G.; Nesmith, D. S. Drought Sress and the Distribution of Vegetative and Reproductive Traits of a Bean Cultivar. Sci. agric. (Piracicaba, Braz.) 2005, 62 (1), 18–22. https://doi.org/10.1590/S0103-90162005000100004.
CrossRef
- Vendruscolo, E. C. G.; Schuster, I.; Pileggi, M.; Scapim, C. A.; Molinari, H. B. C.; Marur, C. J.; Vieira, L. G. E. Stress-Induced Synthesis of Proline Confers Tolerance to Water Deficit in Transgenic Wheat. Journal of Plant Physiology 2007, 164 (10), 1367–1376. https://doi.org/10.1016/j.jplph.2007.05.001.
CrossRef
- Semida, W. M.; Abdelkhalik, A.; Mohamed, Gamal. F.; Abd El-Mageed, T. A.; Abd El-Mageed, S. A.; Rady, M. M.; Ali, E. F. Foliar Application of Zinc Oxide Nanoparticles Promotes Drought Stress Tolerance in Eggplant (Solanum Melongena L.). Plants 2021, 10 (2), 421. https://doi.org/10.3390/ plants10020421.
CrossRef
- Van Nguyen, D.; Nguyen, H. M.; Le, N. T.; Nguyen, K. H.; Nguyen, H. T.; Le, H. M.; Nguyen, A. T.; Dinh, N. T. T.; Hoang, S. A.; Van Ha, C. Copper Nanoparticle Application Enhances Plant Growth and Grain Yield in Maize Under Drought Stress Conditions. J Plant Growth Regul 2022, 41 (1), 364–375. https://doi.org/10.1007/s00344-021-10301-w.
CrossRef
- Ghani, M. I.; Saleem, S.; Rather, S. A.; Rehmani, M. S.; Alamri, S.; Rajput, V. D.; Kalaji, H. M.; Saleem, N.; Sial, T. A.; Liu, M. Foliar Application of Zinc Oxide Nanoparticles: An Effective Strategy to Mitigate Drought Stress in Cucumber Seedling by Modulating Antioxidant Defense System and Osmolytes Accumulation. Chemosphere 2022, 289, 133202. https://doi.org/10.1016/j.chemosphere. 2021.133202.
CrossRef
- Fatollahpour Grangah, M.; Rashidi, V.; Mirshekari, B.; Khalilvand Behrouzyar, E.; Farahvash, F. Effects of Nano-Fertilizers on Physiological and Yield Characteristics of Pinto Bean Cultivars under Water Deficit Stress. Journal of Plant Nutrition 2020, 43 (19), 2898–2910. https://doi.org/10.1080/ 01904167.2020.1799000.
CrossRef
- Dola, D. B.; Mannan, Md. A.; Sarker, U.; Mamun, Md. A. A.; Islam, T.; Ercisli, S.; Saleem, M. H.; Ali, B.; Pop, O. L.; Marc, R. A. Nano-Iron Oxide Accelerates Growth, Yield, and Quality of Glycine Max Seed in Water Deficits. Front. Plant Sci. 2022, 13, 992535. https://doi.org/10.3389/fpls.2022.992535.
CrossRef
- Moharem, M.; Elkhatib, E.; Mesalem, M. Remediation of Chromium and Mercury Polluted Calcareous Soils Using Nanoparticles: Sorption –Desorption Kinetics, Speciation and Fractionation. Environmental Research 2019, 170, 366–373. https://doi.org/10.1016/j.envres.2018.12.054.
CrossRef
- Cao, F.; Dai, H.; Hao, P.-F.; Wu, F. Silicon Regulates the Expression of Vacuolar H+-Pyrophosphatase 1 and Decreases Cadmium Accumulation in Rice (Oryza Sativa L.). Chemosphere 2020, 240, 124907. https://doi.org/10.1016/j.chemosphere.2019.124907.
CrossRef
- Wang, K.; Wang, Y.; Wan, Y.; Mi, Z.; Wang, Q.; Wang, Q.; Li, H. The Fate of Arsenic in Rice Plants (Oryza Sativa L.): Influence of Different Forms of Selenium. Chemosphere 2021, 264, 128417. https://doi.org/10.1016/j.chemosphere.2020.128417.
CrossRef
- Lian, J.; Zhao, L.; Wu, J.; Xiong, H.; Bao, Y.; Zeb, A.; Tang, J.; Liu, W. Foliar Spray of TiO2 Nanoparticles Prevails over Root Application in Reducing Cd Accumulation and Mitigating Cd-Induced Phytotoxicity in Maize (Zea Mays L.). Chemosphere 2020, 239, 124794. https://doi.org/10.1016/ j.chemosphere.2019.124794.
CrossRef
- Sebastian, A.; Nangia, A.; Prasad, M. N. V. Cadmium and Sodium Adsorption Properties of Magnetite Nanoparticles Synthesized from Hevea Brasiliensis Muell. Arg. Bark: Relevance in Amelioration of Metal Stress in Rice. Journal of Hazardous Materials 2019, 371, 261–272. https://doi.org/10.1016/ j.jhazmat.2019.03.021.
CrossRef
- Rossi, L.; Zhang, W.; Schwab, A. P.; Ma, X. Uptake, Accumulation, and in Planta Distribution of Coexisting Cerium Oxide Nanoparticles and Cadmium in Glycine Max (L.) Merr. Environ. Sci. Technol. 2017, 51 (21), 12815–12824. https://doi.org/10.1021/acs.est.7b03363.
CrossRef
- Wu, H.; Tito, N.; Giraldo, J. P. Anionic Cerium Oxide Nanoparticles Protect Plant Photosynthesis from Abiotic Stress by Scavenging Reactive Oxygen Species. ACS Nano 2017, 11 (11), 11283–11297. https://doi.org/10.1021/acsnano.7b05723.
CrossRef
- Zhao, L.; Lu, L.; Wang, A.; Zhang, H.; Huang, M.; Wu, H.; Xing, B.; Wang, Z.; Ji, R. Nano-Biotechnology in Agriculture: Use of Nanomaterials to Promote Plant Growth and Stress Tolerance. J. Agric. Food Chem. 2020, 68 (7), 1935–1947. https://doi.org/10.1021/acs.jafc.9b06615.
CrossRef
- Ullah, S.; Adeel, M.; Zain, M.; Rizwan, M.; Irshad, M. K.; Jilani, G.; Hameed, A.; Khan, A.; Arshad, M.; Raza, A.; Baluch, M. A.; Rui, Y. Physiological and Biochemical Response of Wheat (Triticum Aestivum) to TiO2 Nanoparticles in Phosphorous Amended Soil: A Full Life Cycle Study. Journal of Environmental Management 2020, 263, 110365. https://doi.org/10.1016/j.jenvman.2020.110365.
CrossRef
- Rizwan, M.; Ali, S.; Zia Ur Rehman, M.; Rinklebe, J.; Tsang, D. C. W.; Bashir, A.; Maqbool, A.; Tack, F. M. G.; Ok, Y. S. Cadmium Phytoremediation Potential of Brassica Crop Species: A Review. Science of The Total Environment 2018, 631–632, 1175–1191. https://doi.org/10.1016/j.scitotenv.2018.03.104.
CrossRef
- Babu, S.; Singh, R.; Yadav, D.; Rathore, S. S.; Raj, R.; Avasthe, R.; Yadav, S. K.; Das, A.; Yadav, V.; Yadav, B.; Shekhawat, K.; Upadhyay, P. K.; Yadav, D. K.; Singh, V. K. Nanofertilizers for Agricultural and Environmental Sustainability. Chemosphere 2022, 292, 133451. https://doi.org/10.1016/ j.chemosphere.2021.133451.
CrossRef
- Konappa, N.; Krishnamurthy, S.; Arakere, U. C.; Chowdappa, S.; Akbarbasha, R.; Ramachandrappa, N. S. Nanofertilizers and Nanopesticides: Recent Trends, Future Prospects in Agriculture. In Advances in Nano-Fertilizers and Nano-Pesticides in Agriculture; Elsevier, 2021; pp 281–330. https://doi.org/10.1016/B978-0-12-820092-6.00012-4.
CrossRef
- Hemalatha, M.; Vinita; Sravanalakshmi, G.; Kotibagar, B. C.; Megha. Nanotechnology for Sustainable Agriculture: Current Trends and Future Prospects. In Modern Nanotechnology; Malik, J. A., Sadiq Mohamed, M. J., Eds.; Springer International Publishing: Cham, 2023; pp 43–75. https://doi.org/10.1007/978-3-031-31111-6_3.
CrossRef
- Sagadevan, S.; Periasamy, M. RECENT TRENDS IN NANOBIOSENSORS AND THEIR APPLICATIONS – A REVIEW.
- Verma, K. K.; Song, X.-P.; Joshi, A.; Tian, D.-D.; Rajput, V. D.; Singh, M.; Arora, J.; Minkina, T.; Li, Y.-R. Recent Trends in Nano-Fertilizers for Sustainable Agriculture under Climate Change for Global Food Security. Nanomaterials 2022, 12 (1), 173. https://doi.org/10.3390/nano12010173.
CrossRef
- Zahra, Z.; Habib, Z.; Hyun, H.; Shahzad, H. M. A. Overview on Recent Developments in the Design, Application, and Impacts of Nanofertilizers in Agriculture. Sustainability 2022, 14 (15), 9397. https://doi.org/10.3390/su14159397.
CrossRef
- Mittal, D.; Kaur, G.; Singh, P.; Yadav, K.; Ali, S. A. Nanoparticle-Based Sustainable Agriculture and Food Science: Recent Advances and Future Outlook. Front. Nanotechnol. 2020, 2, 579954. https://doi.org/10.3389/fnano.2020.579954.
CrossRef
- Narendhran, S.; Rajiv, P.; Sivaraj, R. INFLUENCE OF ZINC OXIDE NANOPARTICLES ON GROWTH OF SESAMUM INDICUM L. IN ZINC DEFICIENT SOIL. 8 (3).
- Prasad, A. S. Discovery of Human Zinc Deficiency: 50 Years Later. Journal of Trace Elements in Medicine and Biology 2012, 26 (2–3), 66–69. https://doi.org/10.1016/j.jtemb.2012.04.004.
CrossRef
- Yruela, I. Copper in Plants. Braz. J. Plant Physiol. 2005, 17 (1), 145–156. https://doi.org/10.1590/S1677-04202005000100012.
CrossRef
- Gupta, A.; Bharati, R.; Kubes, J.; Popelkova, D.; Praus, L.; Yang, X.; Severova, L.; Skalicky, M.; Brestic, M. Zinc Oxide Nanoparticles Application Alleviates Salinity Stress by Modulating Plant Growth, Biochemical Attributes and Nutrient Homeostasis in Phaseolus Vulgaris L. Front. Plant Sci. 2024, 15, 1432258. https://doi.org/10.3389/fpls.2024.1432258.
CrossRef
- Nazir, M. A.; Hasan, M.; Mustafa, G.; Tariq, T.; Ahmed, M. M.; Golzari Dehno, R.; Ghorbanpour, M. Zinc Oxide Nano-Fertilizer Differentially Effect on Morphological and Physiological Identity of Redox-Enzymes and Biochemical Attributes in Wheat (Triticum Aestivum L.). Sci Rep 2024, 14 (1), 13091. https://doi.org/10.1038/s41598-024-63987-9.
CrossRef
- Tiwari, V.; Bambharoliya, K. S.; Bhatt, M. D.; Nath, M.; Arora, S.; Dobriyal, A. K.; Bhatt, D. Application of Green Synthesized Copper Oxide Nanoparticles for Effective Mitigation of Fusarium Wilt Disease in Roots of Cicer Arietinum. Physiological and Molecular Plant Pathology 2024, 131, 102244. https://doi.org/10.1016/j.pmpp.2024.102244.
CrossRef
- Feng, Y.; Kreslavski, V. D.; Shmarev, A. N.; Ivanov, A. A.; Zharmukhamedov, S. K.; Kosobryukhov, A.; Yu, M.; Allakhverdiev, S. I.; Shabala, S. Effects of Iron Oxide Nanoparticles (Fe3O4) on Growth, Photosynthesis, Antioxidant Activity and Distribution of Mineral Elements in Wheat (Triticum Aestivum) Plants. Plants 2022, 11 (14), 1894. https://doi.org/10.3390/plants11141894.
CrossRef
- Gobinath, R.; Datta, S.; Vijayakumar, S.; Singh, R.; Sharma, V.; Surekha, K.; Manasa, V. Influence of Iron Nanoparticles on Rice (Oryza Sativa) Plant Growth and Yields. Oryza 2023, 60 (3), 442–447. https://doi.org/10.35709/ory.2023.60.3.7.
CrossRef
- Ghafari, H.; Razmjoo, J. Effect of Foliar Application of Nano-Iron Oxidase, Iron Chelate and Iron Sulphate Rates on Yield and Quality of Wheat. 2013.
- Elbasuney, S.; El-Sayyad, G. S.; Abdelaziz, A. M.; Rizk, S. H.; Tolba, M. M.; Attia, M. S. Stable Colloidal Iron Oxide Nanoparticles: A New Green Nanofertilizer and Therapeutic Nutrient for Eggplant Immune Response Against Fusarium Wilt Disease. J Clust Sci 2024, 35 (4), 983–997. https://doi.org/10.1007/s10876-023-02527-3.
CrossRef
- Samsoon, S.; Azam, M.; Khan, A.; Ashraf, M.; Bhatti, H. N.; Alshawwa, S. Z.; Iqbal, M. Green-Synthesized MnO2 Nanofertilizer Impact on Growth, Photosynthetic Pigment, and Non-Enzymatic Antioxidant of Vigna Unguiculata Cultivar. Biomass Conv. Bioref. 2024, 14 (21), 26943–26952. https://doi.org/10.1007/s13399-022-03686-5.
CrossRef
- Moroni, J. S.; Scott, B. J.; Wratten, N. [No Title Found]. Plant and Soil 2003, 253 (2), 507–519. https://doi.org/10.1023/A:1024899215845.
CrossRef
- Millaleo, R.; Reyes- Diaz, M.; Ivanov, A. G.; Mora, M. L.; Alberdi, M. MANGANESE AS ESSENTIAL AND TOXIC ELEMENT FOR PLANTS: TRANSPORT, ACCUMULATION AND RESISTANCE MECHANISMS. J. Soil Sci. Plant Nutr. 2010, 10 (4), 470–481. https://doi.org/10.4067/S0718-95162010000200008.
CrossRef
- Kasote, D. M.; Lee, J. H. J.; Jayaprakasha, G. K.; Patil, B. S. Manganese Oxide Nanoparticles as Safer Seed Priming Agent to Improve Chlorophyll and Antioxidant Profiles in Watermelon Seedlings. Nanomaterials 2021, 11 (4), 1016. https://doi.org/10.3390/nano11041016.
CrossRef
- Murgueitio-Herrera, E.; Falconí, C. E.; Cumbal, L.; Gómez, J.; Yanchatipán, K.; Tapia, A.; Martínez, K.; Sinde-Gonzalez, I.; Toulkeridis, T. Synthesis of Iron, Zinc, and Manganese Nanofertilizers, Using Andean Blueberry Extract, and Their Effect in the Growth of Cabbage and Lupin Plants. Nanomaterials 2022, 12 (11), 1921. https://doi.org/10.3390/nano12111921.
CrossRef
- El-Beltagi, H. S.; El-Waraky, E. A.; Almutairi, H. H.; Al-Daej, M. I.; El-Nady, M. F.; Shehata, W. F.; Belal, E. B.; El-Mogy, M. M.; El-Mehasseb, I.; Metwaly, M. M. S. Morphophysiological and Biochemical Responses of Cotton ( Gossypium Barbadense L.) to Nano Zinc (ZnO-NPs) and Azospirillum Sp. under Water Deficit Stress Conditions. Journal of Plant Nutrition 2025, 48 (2), 326–344. https://doi.org/10.1080/01904167.2024.2402882.
CrossRef
- Liu, A.; Xiao, W.; Lai, W.; Wang, J.; Li, X.; Yu, H.; Zha, Y. Potential Application of Selenium and Copper Nanoparticles in Improving Growth, Quality, and Physiological Characteristics of Strawberry under Drought Stress. Agriculture 2024, 14 (7), 1172. https://doi.org/10.3390/agriculture14071172.
CrossRef
- Seyed Sharifi, R.; Khalilzadeh, R.; Pirzad, A.; Anwar, S. Effects of Biofertilizers and Nano Zinc-Iron Oxide on Yield and Physicochemical Properties of Wheat under Water Deficit Conditions. Communications in Soil Science and Plant Analysis 2020, 51 (19), 2511–2524. https://doi.org/10.1080/00103624.2020.1845350.
CrossRef
- Nekoukhou, M.; Fallah, S.; Pokhrel, L. R.; Abbasi-Surki, A.; Rostamnejadi, A. Foliar Co-Application of Zinc Oxide and Copper Oxide Nanoparticles Promotes Phytochemicals and Essential Oil Production in Dragonhead (Dracocephalum Moldavica). Science of The Total Environment 2024, 906, 167519. https://doi.org/10.1016/j.scitotenv.2023.167519.
CrossRef
- Saffan, M. M.; Koriem, M. A.; El-Henawy, A.; El-Mahdy, S.; El-Ramady, H.; Elbehiry, F.; Omara, A. E.-D.; Bayoumi, Y.; Badgar, K.; Prokisch, J. Sustainable Production of Tomato Plants (Solanum Lycopersicum L.) under Low-Quality Irrigation Water as Affected by Bio-Nanofertilizers of Selenium and Copper. Sustainability 2022, 14 (6), 3236. https://doi.org/10.3390/su14063236.
CrossRef
- Ekim, R.; Arikan, B.; Alp-Turgut, F. N.; Koyukan, B.; Ozfidan-Konakci, C.; Yildiztugay, E. Polyvinylpyrrolidone-Coated Copper Nanoparticles Dose-Dependently Conferred Tolerance to Wheat under Salinity and/or Drought Stress by Improving Photochemical Activity and Antioxidant System. Environmental Research 2024, 241, 117681. https://doi.org/10.1016/j.envres.2023.117681.
CrossRef
- Abbasifar, A.; Shahrabadi, F.; ValizadehKaji, B. Effects of Green Synthesized Zinc and Copper Nano-Fertilizers on the Morphological and Biochemical Attributes of Basil Plant. Journal of Plant Nutrition 2020, 43 (8), 1104–1118. https://doi.org/10.1080/01904167.2020.1724305.
CrossRef
- Sharifan, H.; Ma, X. Foliar Application of Zn Agrichemicals Affects the Bioavailability of Arsenic, Cadmium and Micronutrients to Rice (Oryza Sativa L.) in Flooded Paddy Soil. Agriculture 2021, 11 (6), 505. https://doi.org/10.3390/agriculture11060505.
CrossRef
- Yuan, H.; Liu, Q.; Fu, J.; Wang, Y.; Zhang, Y.; Sun, Y.; Tong, H.; Dhankher, O. P. Co-Exposure of Sulfur Nanoparticles and Cu Alleviate Cu Stress and Toxicity to Oilseed Rape Brassica Napus L. Journal of Environmental Sciences 2023, 124, 319–329. https://doi.org/10.1016/j.jes.2021.09.040.
CrossRef
- Fahad; Balouch, A.; Agheem, M. H.; Memon, S. A.; Baloch, A. R.; Tunio, A.; Abdullah; Pato, A. H.; Jagirani, M. S.; Panah, P.; Gabole, A. A.; Qasim, S. Efficient Mitigation of Cadmium and Lead Toxicity in Coriander Plant Utilizing Magnetite (Fe3 O4 ) Nanofertilizer as Growth Regulator and Antimicrobial Agent. International Journal of Environmental Analytical Chemistry 2022, 102 (16), 3868–3879. https://doi.org/10.1080/03067319.2020.1776861.
CrossRef
- Adil, M. F.; Sehar, S.; Ma, Z.; Tahira, K.; Askri, S. M. H.; El-Sheikh, M. A.; Ahmad, A.; Zhou, F.; Zhao, P.; Shamsi, I. H. Insights into the Alleviation of Cadmium Toxicity in Rice by Nano-Zinc and Serendipita Indica: Modulation of Stress-Responsive Gene Expression and Antioxidant Defense System Activation. Environmental Pollution 2024, 350, 123952. https://doi.org/10.1016/j.envpol.2024.123952.
CrossRef
- Hamagharib Rojbayany, S. N.; Aziz Aljoburi, J. M. The Effect of Spraying Iron, Zinc and Copper Nanoparticles on the Components of the Crop of Two Cultivars of Bread Wheat. IOP Conf. Ser.: Earth Environ. Sci. 2024, 1371 (3), 032011. https://doi.org/10.1088/1755-1315/1371/3/032011.
CrossRef
Accepted on: 08 Sep 2025
Second Review by: Dr. Anwar Qasim Ahmad Hameed
Final Approval by: Dr. Ayssar Nahle