ORAI1 Gene and Calcium Deficiency in Oral Cancer: An Overview
1Department of Biotechnology, PSGR Krishnammal College for women, Coimbatore, Tamil Nadu, India.
2Department of Microbiology, PSG College of Arts and Science, Coimbatore, Tamil Nadu, India.
3Department of Botany, PSG College of Arts and Science, Coimbatore, Tamil Nadu, India.
4Department of Biochemistry, PSG College of Arts and Science, Coimbatore, Tamil Nadu, India.
Corresponding Author E-mail:shalini@psgrkcw.ac.in
DOI : http://dx.doi.org/10.13005/ojc/410224
ABSTRACT:Oral cancer, which includes malignancies of the lip, tongue, and mouth, ranks as the sixth most prevalent cancer globally, with approximately 355,000 new cases and 177,000 deaths annually. Over the past 25 years, its incidence has risen significantly among young individuals and women, while the decline in mortality rates remains minimal. Among these cases, oral squamous cell carcinomas (OSCCs) account for nearly 90%, originating primarily from squamous cells in the oral cavity and lips. Nutritional deficiencies, particularly inadequate levels of vitamins D, C, B, and A, along with protein-energy malnutrition (PEM), have been associated with various oral health issues, including enamel hypoplasia and periodontal disease. A lack of calcium, in particular, weakens alveolar bone density and compromises oral mucosal resistance, making tissues more vulnerable to carcinogenic agents. Recent studies suggest a link between calcium deficiency and the overexpression of the ORAI1 gene, which regulates store-operated calcium entry (SOCE) and plays a crucial role in tumorigenesis. Elevated ORAI1 expression in OSCC has been linked to prolonged calcium influx, disruptions in ion homeostasis, and increased cancer cell proliferation, migration, and pain modulation. The oncogenic activation of ORAI1 and its associated calcium channels (Orai-2 and Orai-3) interferes with key signalling pathways such as Akt, NF-κB, and mTOR, thereby promoting tumour progression. Furthermore, tobacco-derived carcinogens like 4-NQO and NNN have been shown to upregulate ORAI1 and Orai-2 expression in oral cancer cell lines. A deeper understanding of the connection between calcium deficiency and ORAI1 overexpression in OSCC may offer valuable insights for developing targeted therapies aimed at regulating abnormal calcium signalling in oral cancer cells.
KEYWORDS:Calcium deficiency; Oral cancer; oral squamous cell carcinoma (OSCC); ORAI1; store-operated calcium entry (SOCE)
Background of the study
Oral cancer presents a substantial global health concern, especially for dental surgeons 1, with 48% of all head and neck cancer cases being attributed to it. Histologically, 90% of oral cancer cases are oral squamous cell carcinomas (OSCCs), mainly from squamous cells in the mouth or lip.2,3 Deficiencies in vitamins D, C, B, and A, along with Protein Energy Malnutrition (PEM), are linked to oral tissue diseases, including enamel hypoplasia.4 Diet profoundly influences periodontal health, with calcium deficiency impacting the density of the alveolar bone.5 Dentists explore plant properties for their potential analgesic, antioxidant, and antimicrobial benefits.6 In oral cancer, heightened ORAI1 expression leads to prolonged calcium influx.7 The main intention of this work is to explore the relationship between calcium shortage and high ORAI1 gene expression in oral cancer.
The connection between oral cancers and calcium deficiency
Calcium is necessary for the healthy development and upkeep of calcified oral tissues, including jaw bones, bony sockets, and complex dental tissues. Mineralized tooth structures are not made of the same bone components, including cementum, dentin, and enamel. The coronal section of the tooth’s enamel layer comprises giant, closely spaced hydroxyapatite crystals with a unique structure.8 A decrease in bone mineralization exacerbates pathological periodontal changes, which provide less support for the teeth. Reduced dietary calcium and calcium-phosphorus intake may worsen both disorders’ symptoms by promoting bone resorption.9 The jaw bones (mostly the alveolar bones), cranial bones, ribs, vertebrae, and long bones are the bones affected by this form of bone loss in decreasing order. Studies have shown that increased calcium consumption reduces inflammation-related pain in gingivitis patients and improves tooth mobility—inadequate dietary calcium intake results in gingival and periodontal disease that is more severe.10 Low calcium levels can compromise the function of the oral mucosa and its resistance to carcinogenic agents. Furthermore, oxidative stress and inflammatory reactions can be made worse by a calcium deficit, which accelerates the carcinogenic process.
Correlation of ORAI1 to OSCC
One of the leading causes of cancer development is oncogenic transformation-induced abnormal expression of ion channel-coding genes. This is happening because channels like these regulate ion concentrations, which have an impact on key processes in biology such as cancer cell growth.11 More specifically, changes in Ca2+ homeostasis have been linked to the development of tumor phenotypes and the epithelial-to-mesenchymal transition (EMT).12 The Ca2+ release-activated Ca2+ (CRAC) channels control store-operated Ca2+ entry (SOCE) and have been connected to some diseases, including the advancement of cancer.11 The Ca2+ channel ORAI1 is a crucial regulator of pain associated with mouth cancer. Significant expression of ORAI1 was seen in tumour samples from oral cancer patients, and ORAI1 activation resulted in prolonged Ca2+ influx in human oral cancer cells. RNA analysis from earlier research showed that ORAI1 regulated a large number of genes, including metalloproteases (MMPs) and pain modulators, that encoded markers of oral cancer. Mouth cancer cells lacking-ORAI1 produced primary tumours and less allodynia when injected into mice’s feet than control cells. Trigeminal ganglion neurons’ action potentials grew after being exposed to MMP1. These imply that MMP1 abundance is regulated by ORAI1, which may be important in the development of mouth cancer and pain.13
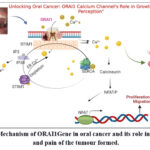 |
Figure 1: Mechanism of ORAI1Gene in oral cancer and its role in the growth and pain of the tumour formed. |
Overexpression of Gene in Oral Oncology
Store-operated calcium entry (SOCE) is triggered by SOCE channels (SOCC) in a variety of cells from mammals as a survival strategy in response to ER calcium depletion. Orai-1, Orai-2, and Orai-3 are found on the plasma membrane, and two proteins called stromal interaction molecules (STIM-1 and STIM-2) detect calcium in the ER membrane and regulate SOCC. Cancer of the mouth tissues and cell lines (SAS) expressed more Orai-1 and Orai-2 proteins than normal epithelial tissues and cell lines. Furthermore, Orai-1 and Orai-2 silencing using pharmacological SOCE regulators and siRNAs reduced calcium absorption and decreased mouth cancer cell proliferation, growth of colonies, and motility.Moreover, the Akt, NF-κB, and mTOR pathways were inhibited in mouth cancer cells when Orai-1 and Orai-2 were silenced. Remarkably, the carcinogens 4-NQO and NNN from tobacco enhanced the expression of Orai-1 and Orai-2 in SAS cells.14
Conclusion
Calcium is vital for the growth and maintenance of calcified oral structures like jaw bones and dental tissues. Comparing tissues and cell lines (such as SAS) to regular epithelial counterparts in oral cancer revealed heightened expression of Orai-1 and Orai-2 proteins. Orai-1 governs extracellular calcium influx in cancer cells, thereby modulating various signalling pathways. This dysregulation suggests a potential role of calcium signalling in oral cancer progression. Understanding these mechanisms could pave the way for targeted therapeutic interventions to disrupt aberrant calcium signalling and its downstream effects in oral cancer cells.
Acknowledgement
The authors acknowledge the ICSSR, DST FIST, New Delhi for the infrastructure provided. Department of Biotechnology, PSGR Krishnammal College for Women, Coimbatore for providing technical assistance during the study.
Funding Sources
This work was supported by Indian Council of Social Science Research (ICSSR) Grant no : ICSSR/RPD/MN/2023-24/Sc/55 and Author Dr.Shalini G has received research support from PSGR Krishnammal College for women, Coimbatore.
Conflict of interest
The authors declare that they have no conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Author contributions
Nithya Thangam S,Vino U, Sankaravel, Vinitha and Saravanan: Writing –original draft. Conceptualization, Supervision: Review, Editing and Formal analysis: Shalini G, Rajan T & Nirmal kumar R
References
- Rivera, C. (2015). Essentials of oral cancer. International journal of clinical and experimental pathology, 8(9), 11884. PMCID: PMC4637760.
- Nakashima, T., Tomita, H., Hirata, A., Ishida, K., Hisamatsu, K., Hatano, Y., … & Hara, A. (2017). Promotion of cell proliferation by the proto‐oncogene DEK enhances oral squamous cell carcinogenesis through field cancerization. Cancer medicine, 6(10), 2424-2439. https://doi.org/10.1002/cam4.1157
CrossRef - Lingen, M. W., Kalmar, J. R., Karrison, T., & Speight, P. M. (2008). Critical evaluation of diagnostic aids for the detection of oral cancer. Oral oncology, 44(1), 10-22. https://doi.org/10.1016/j.oraloncology.2007.06.011
CrossRef - Psoter, W. J., Reid, B. C., & Katz, R. V. (2005). Malnutrition and dental caries: a review of the literature. Caries research, 39(6), 441-447. https://doi.org/10.1159/000088178
CrossRef - Ehizele, A. O., Ojehanon, P. I., & Akhionbare, O. (2009). Nutrition and oral health. Benin Journal of Postgraduate Medicine, 11(1). https://doi.org/10.4314/bjpm.v11i1.48830
CrossRef - Sinha, D. J., & Sinha, A. A. (2014). Natural medicaments in dentistry. AYU (An international quarterly journal of research in Ayurveda), 35(2), 113-118. 4103/0974-8520.146198
CrossRef - Zhang, Q., Wang, C., & He, L. (2024). ORAI Ca2+ Channels in Cancers and Therapeutic Interventions. Biomolecules, 14(4), 417. https://doi.org/10.3390/biom14040417
CrossRef - Rathee, D. M., Singla, D. S., & Tamrakar, D. A. K. (2013). Calcium and Oral Health: A Review. International Journal of Scientific Research, 2(9), 335-336.
CrossRef - Ortega, R. M., Requejo, A. M., Sotillos, A. E., Andres, P., Lopez-Sobaler, A. M., & Quintas, E. (1998). Implication of calcium deficiency in the progress of periodontal diseases and osteoporosis. Nutricion hospitalaria, 13(6), 316-319. PMID: 9889558.
- Nishida, M., Grossi, S. G., Dunford, R. G., Ho, A. W., Trevisan, M., & Genco, R. J. (2000). Calcium and the risk for periodontal disease. Journal of periodontology, 71(7), 1057-1066. https://doi.org/10.1902/jop.2000.71.7.1057
CrossRef - Monteith, G. R., Prevarskaya, N., & Roberts-Thomson, S. J. (2017). The calcium–cancer signalling nexus. Nature Reviews Cancer, 17(6), 373-380. https://doi.org/10.1038/nrc.2017.18
CrossRef - Prevarskaya, N., Skryma, R., & Shuba, Y. (2010). Ion channels and the hallmarks of cancer. Trends in molecular medicine, 16(3), 107-121. https://doi.org/10.1016/j.molmed.2010.01.005
CrossRef - Son, G. Y., Tu, N. H., Santi, M. D., Loya Lopez, S., Souza Bomfim, G. H., Vinu, M., … & Lacruz, R. S. (2023). The Ca2+ channel ORAI1 is a regulator of oral cancer growth and nociceptive pain. Science signaling, 16(801), eadf9535. https://doi.org/10.1126/scisignal.adf9535
CrossRef - Singh, A. K., Roy, N. K., Bordoloi, D., Padmavathi, G., Banik, K., Khwairakpam, A. D., … & Sukumar, P. (2020). Orai-1 and Orai-2 regulate oral cancer cell migration and colonisation by suppressing Akt/mTOR/NF-κB signalling. Life Sciences, 261, 118372. https://doi.org/10.1016/j.lfs.2020.118372
CrossRef
Accepted on: 30 Mar 2025
Second Review by: Dr. Shaikh Azam
Final Approval by: Dr. B. K Sharma

















