Nickel(II)-dithiocarbamate Mixed Ligand Complexes: Synthesis, Characterization, and Anti-bacterial properties
1Department of Chemistry, College of Science, Imam Mohammad Ibn Saud Islamic University, Riyadh 11623, Saudi Arabia.
2Department of Chemistry, College of Science, Tikrit University, Tikrit, Iraq
3Department of Chemistry, College of Science, King Faisal University, Al-Ahsa 31982, Saudi Arabia
4Department of Chemistry, Faculty of Science, Mansoura University, 35516 Mansoura, Egypt
Corresponding Author E-mail:dr.ahmed.chem@tu.edu.iq
DOI : http://dx.doi.org/10.13005/ojc/410210
ABSTRACT:Six homoleptic (1 and 2) and heteroleptic (3-6) nickel(II) chelates comprising dithiocarbamate and triphenylphosphine ligands were prepared and elucidated by various spectroscopic analysis. The dithiocarbamate ligand was bonded in a bidentate chelation manner via the sulfur atoms to furnish a square planar geometry. The antibacterial activity of mixed Ni(II) dithiocarbamate and triphenylphosphine ligand chelates was obtained against three bacterial strains. Among the complexes, the heteroleptic nickel(II) chelates comprising mixed dithiocarbamate and triphenylphosphine ligands (6) manifested the highest antibacterial activity against all tested bacteria. Our study demonstrated that the heteroleptic nickel(II) chelates might be further elaborated and improved for enhanced antimicrobial applications.
KEYWORDS:Biphenyl; Biological Activity; Dithiocarbamate; Biphenyl; Nickel; Ligand
Introduction
Dithiocarbamates (R2NCSS−) are unique amide compounds structurally characterised by a functional C=S bond and their ability to design stable chelates with a variety of cations, particularly transition metals.1-3 They have also emerged as a versatile class of ligands often employed to anchor metals in a variety of applications in materials chemistry.3-7 Since their discovery early in the 19th century, a vast number of R2NCSS− ligands and complexes as diverse as the metals and nonmetals constituting their core structure have fascinated chemists[8,9]. A general perception of R2NCSS− complexes is that they act solely as precursors for the formation of metal chalcogenides.8 In view of the rapid advances in chemistry and applications of R2NCSS−, there is a rising interest in obtaining high-purity R2NCSS−, as well as the additional need for enhanced facile synthetic routes. These research efforts have been directed mainly at new ligand design, improved preparation methods, and better structures towards better performance as stabilisers.1-14
R2NCSS− are considered valuable auxiliary ligands due to their strong σ-donating thiolate group(s) accompanied by a moderate π-back-bonding ability.1-3 These unique properties often lead to the formation of unconventional homoleptic and heteroleptic complexes,16-32 many of which are of size or shape well suited for a variety of applications. This, together with the increasing academic and industrial interest in R2NCSS−complexes, warrants a deeper understanding of the coordination character and nature of the R2NCSS−–metal bond.1-7 Another wide range of applications can be seen including, but not limited to, in the fields of agriculture (as biocides such as the diethyldithiocarbamates), material science, medicine (as antabuse), photo-sensor-based polymers, and organic synthesis.9-14 Critically, the produced insight could present new tactics in the rational design of these unconventional stress manifolds and, thus, their more efficacious applicability. There has consequently been a concerted effort to probe the nature of this coordination unit, in particular focusing on the detail of the metal–sulfur bonding interaction.1,2,32,33
This work continues previous work on the synthesis of mixed ligand chelates comprising R2NCSS− ligands and other ligands such as phosphines, amines, etc.16-26 Herein, we utilised the R2NCSS−Ni(II) complex of [1,1′-biphenyl]-4-amine or 4-(4-methylphenyl)aniline as a foundation to synthesize various mixed ligand complexes that include triphenylphosphine and additional ligands. Furthermore, evaluate the antibacterial activity of the prepared chelates against three bacterial pathogens.
Experimental part
Complexes [Ni(L1)2] (1) or [Ni(L2)2] (2)
These complexes were achieved according to the one-pot producer as follows:
A synthetic pathway involving three components for dithiocarbamates 34 was utilised to synthesis the ligand. In brief, [1,1′-biphenyl]-4-amine (0.196g, 1.000 mmol) (for ligand L1H) or 4-(4-methylphenyl)aniline (0.183g, 1.000 mmol) (for ligand L2H) reacted with CS2 (0.076 g, 1.000 mmol) at 0-5 oC for 0.5 h. Next, KOH (0.056 g, 1.000 mmol) was introduced, and the solution is lifted to stir for three hours to provide a yellowish-white solution. Then, NiCl2·6H2O (0.119g, 0.500 mmol) in (15 mL) H2O was introduced with stirring for three h to give a light green ppt formed. The produced solid was isolated, rinsed with H2O and then with (C2H5)2O ether.
Synthesis of complexes (3-6)
These complexes were prepared according to the one-pot producer as follows:
A mixture of complexes [Ni(L1)2] (1) (0.50 mmol) or [Ni(L2)2] (2) (0.001 mol), NiCl2 ·6H2O (0.50 mol), triphenylphosphine (PPh3) (1.00 mmol) and potassium thiocyanate (KSCN) (1.0 mol) was heated at 69 oC for three h in MeOH (50 mL). The produced material was isolated, washed with distilled water and then with (C2H5)2O ether. The physical properties, yield percentage, conductivity and elemental analysis are listed in Table 1.
Biological activity studies
The antibacterial efficacy of nickel(II) complexes with dithiocarbamate and other ligands was assessed toward three bacterial species: Bacillus subtilis, Pseudomonas aeruginosa, and Escherichia coli. The efficacy of the Ni(II)-dithiocarbamate complexes was evaluated using the agar disc diffusion method established by Biemer 35, with data compared to streptomycin as the typical antibiotic.
Results and Discussion
Chemistry and the synthesis protocols
Complexes (1) and (2) were prepared by one-pot method as follows:
First step
The solution of [1,1′-biphenyl]-4-amine dithiocarbamate (L1Na) or 4-(4-methylphenyl)aniline dithiocarbamate (L2Na) was prepared by the reaction of [1,1′-biphenyl]-4-amine or 4-(4-methylphenyl)aniline with CS2 in presence NaOH as a base below 5°C.
Second step
Nickel chloride hexa-hydrate was introduced to the fresh solution of [1,1′-biphenyl]-4-amine dithiocarbamate (L1Na) or 4-(4-methylphenyl)aniline dithiocarbamate (L2Na) in (1:2) molar ratio to afford complexes (1) and (2) as a product in good yield 81% and 87%, respectively (for more details and information see Scheme 1).
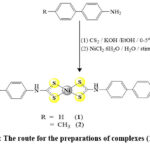 |
Scheme 1: The route for the preparations of complexes (1) and (2). |
Refluxed a mixture of equivalent moles of complex (1) or complex (2) and nickel chloride hexahydrate with two equivalent moles of triphenylphosphine and potassium thiocyanate or potassium cyanide afforded complexes of the type [Ni(SCN)(L)(PPh3)] (3,4) and [Ni(CN)(L)2(PPh3)] (5,6) (Scheme 2) in good yields (75- 90%). The products are soluble in DMSO and DMF, warm CHCl3 or warm CH2Cl2 and partially soluble in THF. We endeavored to obtain a single crystal for X-ray analysis but were unsuccessful. Consequently, it was impossible to investigate this kind of material by crystallography.
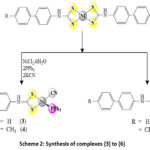 |
Scheme 2: Synthesis of complexes (3) to (6) |
The novel chelates were analysed through conductivity measurements, chemical analysis (CHN), infrared spectroscopy, UV-visible spectra, magnetic susceptibility, and NMR (1H and 31P) spectra. The molar conductivity of each complex has been studied in solution of DMSO (10-3 M). The low molar conductivity measurements, between 5.9 and 17.6 ohm⁻¹·cm²·mol⁻¹, indicate that all complexes behave as non-electrolytes. The CHN analysis supported the proposed geometries of the synthesised chelates. The data for characterisation can be found in Table (1).
Table 1: Spectral data of Ni(II) complexes.
| N | H | C | Λ (ohm-1. cm2. mol-1)in DMSO | Yield% | m.p(Co) | Colour | Complexes | Seq. |
| 5.12 (5.32) | 3.68 (3.89) | 57.05 (57.23) | 11.2 | 81 | 167-168 | Light green | [Ni(L1)2] | 1 |
| 4.87 (4.94) | 4.20 (4.43) | 58.44 (58.38) | 5.9 | 87 | 136-139 | Greenish yellow | [Ni(L2)2] | 2 |
| 4.49 (4.78) | 4.04 (4.30) | 61.65 (61.80) | 7.8 | 79 | 278-280 | Brownish-yellow | [Ni(SCN)(L1)2(PPh3)] | 3 |
| 4.39 (4.53) | 4.27 (4.20) | 62.18 (62.29) | 10.1 | 84 | 290-291 | Green | [Ni(SCN)(L2)2(PPh3)] | 4 |
| 4.39 (4.53) | 4.27 (4.20) | 62.18 (62.29) | 8.9 | 75 | 213-214 | Brownish-yellow | [Ni(CN)(L1)2(PPh3)] | 5 |
| 4.63 (4.53) | 4.50 (4.20) | 65.47 (62.29) | 12.9 | 90 | 193-195 | Green | [Ni(CN)(L2)2(PPh3)] | 6 |
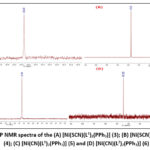 |
Figure 1: 31P NMR spectra of the (A) [Ni(SCN)(L1)2(PPh3)] (3); (B) [Ni(SCN)(L2)2(PPh3)] (4); (C) [Ni(CN)(L1)2(PPh3)] (5) and (D) [Ni(CN)(L2)2(PPh3)] (6). |
Spectroscopic data
The 31P-{1H} NMR spectra of products of the reaction of [Ni(L)2] (1 and 2) with triphenylphosphine in presence nickel chloride and KSCN or KCN (Figure 1A-D) displayed a single peak at δP= 24.32 ppm, 27.52ppm, 41.44ppm and 32.22ppm, for the complexes (3-6), correspondingly. This indicates the presence of a singular isomer each.
The 1H NMR spectrum of the dithiocarbamate chelate (1) displayed the protons of the Ph rings as three separated peaks, the first peak as a multiplet peak at dH 7.32ppm (6H), the second as a doublet peak dH 7.50ppm (3JHH 8:00Hz, 2H), and the third peak also as doublet peak dH 7.72ppm (3JHH 8:00Hz, 2H), whereas the proton of NH group displayed as a singlet peak at dH 12.88 ppm. Complex (2) (Figure 2) displayed the distinct four separated doublet peaks for the Ph rings, the first peak as at dH 7.67ppm (3JHH 8:00Hz, 2H), the second at dH 7.41ppm (3JHH 8:00Hz, 2H), the third at dH 7.24ppm (3JHH 8:00Hz, 2H), and the fourth at dH 7.04ppm (3JHH 8:00Hz, 2H), whereas the proton of the NH and methyl groups displayed as a singlet peak at dH 1.93 ppm and dH 12.41 ppm, respectively.
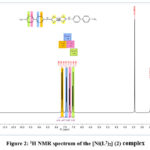 |
Figure 2: 1H NMR spectrum of the [Ni(L2)2] (2) complex |
In the case of the dithiocarbamate complexes (3-6)(Figure 3) displayed the protons of the Ph in the dithiocarbamate and triphenylphosphine ligands as multiplet peaks at dH 7.56ppm, dH 7.64ppm, dH 7.49ppm, and dH 7.61ppm, whereas the proton of the NH group displayed as a singlet peak at dH 12.71ppm, dH 12.39ppm, dH 12.43ppm and dH 12.28ppm, for the four complexes, respectively. At the same time, the methyl group displayed a singlet peak at dH 2.09 ppm and dH 2.01 ppm, respectively.
 |
Figure 3: 1H NMR spectrum of the [Ni(SCN)(L1)2(PPh3)] (3) complex |
The IR spectra of the dithiocarbamate chelates (1) and (2) displayed a strong band at position 1504 cm-1 and 1512 cm-1 referred to the stretching frequency of the u(C=C) group of the Ph system in the two complexes, respectively, further, the spectra displayed band at 3254 cm-1 and 3210 cm-1 as medium to broadband due to the stretching frequency of the u(N-H) group. Also, t, the spectra displayed bands 1464cm-1 and 1451 cm-1 due to the stretching frequency of the u(C-N) group.17-19
In the spectra of the complexes (3-6), three bands were observed that were not present in the spectra of (1) and (2) complexes, located in the ranges of (1429-1442) cm-1, (1109-1130) cm-1, and (493-513) cm-1, attributed to the u(P-Ph), u(P-C) stretching, and bending of the u(P-C) band, respectively.22-25 The CSS thiocarbonyl stretching showed as a strong to medium band within (1043-1078) cm-1 range for complexes (3-6) [20-30]. Our data indicates that the dithiocarbamate ligands were chelated in a bidentate chelating manner via the sulfur atoms with nickel(II) ions.28-31,34 Additional groups are documented in Table 2.
Table 2: IR data in (cm-1).
| Comps. | n(N-H) | n(C-H) | Phosphine ligands | n(C=C)n(C-N) | n(CSS) | n(M-P) or n(M-S) | |||
| Aliph. | Arom. | n(P-Ph) | n(P-C) | d(P-C) | |||||
| 1 | 3254m | 3062w | – | – | – | – | 1504s1464m | 1076m | -431w |
| 2 | 3210m | 3038w | 2962w | – | – | – | 1512s1451m | 1069m | -414w |
| 3 | 3255m | 3078w | – | 1430s | 1109s | 498s | 1512s1451m | 1078m | 451w423w |
| 4 | 3210w | 3023w | 2897w | 1434s | 1123s | 503s | 1512s1451m | 1059s | 467w410w |
| 5 | 3189w | 3087w | – | 1442s | 1130s | 513s | 1554m1429s | 1070m | 478w437w |
| 6 | 3209w | 3051w | 2913w | 1429s | 1116s | 493s | 1560s1476s | 1043m | 459w420w |
Anti-bacterial activity
The antibacterial properties of the mixed ligand chelates of nickel (II) dithiocarbamate and triphenylphosphine ligands. The outcomes were attained against three bacterial strains (see Table 3) usingtThe standard agar disc diffusion method. The diameter of the inhibition zone (DIZ) was compared to that of streptomycin as the positive control. The relative activity index (%) was calculated as shown below:
Table 3: DIZ (mm) and activity index (%) of synthetic compounds (3-6).
| compounds | DIZ (mm) | Activity index (%) | ||||
| p. aeruginosa | B. subtitles | E. Coli | p. aeruginosa | B. subtitles | E. Coli | |
| 3 | 16 | 14 | 20 | 59 | 50 | 67 |
| 4 | 18 | 19 | 24 | 67 | 68 | 80 |
| 5 | 17 | 17 | 22 | 63 | 61 | 73 |
| 6 | 22 | 20 | 26 | 81 | 71 | 87 |
| Streptomycin | 27 | 28 | 30 | 100 | 100 | 100 |
* DMSO is used as a negative control.
The mixed ligand complexes of nickel(II) of dithiocarbamate and triphenylphosphine showed moderate to effective activity towards the examined pathogenic bacterial species. The results achieved may be described as follows:
The synthetic nickel (II) compounds showed good activity towards the Coli species, whilst the minimal activity was observed against B. subtills.
[Ni(CN)(L2)2(PPh3)] (6) compound exhibited a high activity towards examined bacteria matched with other tested compounds. Whereas the [Ni(SCN)(L1)2(PPh3)] (3) exhibited the lowest activity.Inhibition order against the aeruginosa, B. subtilis, and E coli bacteria species of the compound are as follows:
3 > 5 > 4 > 6
increase of inhibition zone→
 |
Figure 4: The anti-bacterial activities. * DMSO is used as a negative control. |
Conclusion
Mixed ligand Ni(II) complexes that involve the dithiocarbamate ligand, triphenylphosphine, thiocyanate, and cyanide ligand were synthesized. The complexes have been completely characterised through CHN analysis, molar conductivity measurements, as well as NMR (1H and 31P) and IR methods. The spectroscopic findings indicated that the thione ligand coordinates bidentate via S atoms with Ni(II). The study of biological activity against Escherichia coli, Pseudomonas aeruginosa, and Bacillus subtilis was conducted, revealing that the Ni(CN)(L2)2(PPh3)] (6) complex exhibited superior inhibition against all tested organisms to other chelates evaluated. In contrast, the [Ni(SCN)(L1)2(PPh3)] (3) displayed the least activity.
Acknowledgement
The authors extend their appreciation to the Deanship of Scientific Research, Imam Mohammad Ibn Saud Islamic University (IMSIU), Saudi Arabia, for funding this research work through Grant No. (221412015).
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Heard, P. J. Inorg. Chem. 2005, 53, 1-69.
- Hogarth, G Inorg. Chem.2005, 53, 71-561.
CrossRef - Hogarth, G. Mini-Rev. Med. Chem. 2012, 12(12), 1202-1215.
CrossRef - Magee, R. J.; Hill, J. O. Anal. Chem.,1985, 8(1-2), 5-72.
- Adeyemi, J. O.; Onwudiwe, D. C. Molecules, 2018, 23(10), 2571.
CrossRef - Sarker, J. C.; Hogarth, G. Rev.2021, 121(10), 6057-6123.
CrossRef - Ajiboye, T. O.; Ajiboye, T. T.; Marzouki, R.; Onwudiwe, D. C. J. Mol. Sci., 2022, 23(3), 1317.
CrossRef - Odularu, A. T.; Ajibade, P. A. Chem. Appl., 2019, 2019(1), 8260496.
CrossRef - Hill, J. O.; Magee, R. J.; Liesegang, J. Inorg. Chem., 1985, 5(1), 1-27.
CrossRef - Fujii, S.; Yoshimura, T. Chem. Rev., 2000, 198(1), 89-99.
CrossRef - Bond, A. M.; Martin, R. L. Chem. Rev., 1984, 54, 23-98.
CrossRef - Adeyemi, J. O.; Onwudiwe, D. C. Chim. Acta, 2020, 511, 119809.
CrossRef - Buac, D.; Schmitt, S.; Ventro, G.; Rani Kona, F.; Ping Dou, Q. Mini-Rev. Med. Chem., 2012, 12(12), 1193-1201.
CrossRef - Ahmed, A. J. Asian J. Chem, 2018, 30, 2595-2602.
CrossRef - Alshdoukhi, I. F.; Al-barwari, A. S.; Aziz, N. M.; Khalil, T.; Faihan, A. S.; Al-Jibori, S. A.; Al-Janabi, A. S. Chem. Soc. Ethiop, 2024, 38(4), 889-899.
CrossRef - Al-Jibori, S. A.; Al-Janabi, A. S.; Al-Sahan, S. W.; Wagner, C. Mol. Struct., 2021, 1227, 129524.
CrossRef - Al‐Janabi, A. S.; Kadhim, M. M.; Al‐Nassiry, A. I.; Yousef, T. A. Organomet. Chem., 35(2), e6108.
- Abdullah, T. B.; Behjatmanesh-Ardakani, R.; Faihan, A. S.; Jirjes, H. M.; Abou-Krisha, M. M.; Yousef, T. A.; Kenawy S. H. ; Al-Janabi, A. S. Molecules, 2023, 28(5), 2305.
CrossRef - Al‐Janabi, A. S.; Saleh, A. M.; Hatshan, M. R. Chin. Chem. Soc., 2021, 68(6), 1104-1115.
CrossRef - Al‐Nassiry, A. I.; Al‐Janabi, A. S.; Thayee Al‐Janabi, O. Y.; Spearman, P.; Alheety, M. A. Chin. Chem. Soc., 2020, 67(5), 775-781.
CrossRef - Salman, M. M.; Al-Dulaimi, A. A.; Al-Janabi, A. S.; Alheety, M. A. Today Proc., 2021, 43, 863-868.
CrossRef - Abdullah, K. T.; Al-Janabi, A. S.; Hussien, N. J.; Yousef, T. A., Shaaban, S.; Attia, M. I.; & Al Duaij, O. K. Chem. Soc. Ethiop, 2024, 79-90..
- Al-Janabi, E. M.; Alheety, M. A.; Al-Jibori, S. A.; Faihan, A. S.; Al-Janabi, A. S. J Iran Chem. Soc., 2023, 20(8), 1781-1790.
CrossRef - Al-Janabi, E. M.; Hatshan, M. R.; Adil, S. F.; Kadhum, W. R.; Al-Jibori, S. A., Faihan, A. S.; Al-Janabi, A. S. Mol. Struct., 2022, 1252, 132227.
CrossRef - Abdullah, T. B.; Jirjes, H. M.; Faihan, A. S.; Al-Janabi, A. S. Mol. Struct., 2023, 1276, 134730.
CrossRef - Kadhim, M. M.; Juber, L. A. A.; Al-Janabi, A. S.Iraqi J. Sci., 2021, 3323-3335.
CrossRef - Pal, S. K.; Singh, B.; Yadav, J. K.; Yadav, C. L.;Drew, M. G.; Singh, N.; Kumar, K. Dalton Trans. 2022, 51(34), 13003-13014.
CrossRef - Bobinihi, F. F.; Osuntokun, J.; Onwudiwe, D. C. Saudi Chem. Soc., 2018, 22(4), 381-395.
CrossRef - Singh, S. K.; Kumar, V.; Drew, M. G.; Singh, N. Chem. Commun., 2013, 37, 151-154.
CrossRef - Bobinihi, F. F.; Onwudiwe, D. C.; Hosten, E. C. Mol. Struct., 2018, 1164, 475-485.
CrossRef - Rani, P. J.; Thirumaran, S.; Ciattini, S. Phosphorus, Sulfur Relat. Elem. 2013, 188(6), 778-789.
CrossRef - Kondo, T.; Mitsudo, T. A. Rev., 2000, 100(8), 3205-3220.
CrossRef - Abdalrazaq, E. A.; Abu-Yamin, A. A.; Taher, D., Hassan, A. E.; Elhenawy, A. A. Sulf. Chem.,2024, 45(5), 714-739.
CrossRef - Bobinihi, F. F.; Onwudiwe, D. C.; Hosten, E. C. Mol. Struct., 2018, 1164, 475-485.
CrossRef - Biemer, J. J. Clin. Lab. Sci., 1973, 3(2), 135-140.
Second Review by: Dr. Deependra Yadav
Final Approval by: Dr. B.K Sharma

















