Synthesis Development, Spectroscopy Characterizations, and Molecular Docking Study of Some New Bis-1,3-Oxazepine and Bis-1,3-Benzoxazepine Derivatives
Department of Chemistry, College of Science, University of Zakho, Zakho, Iraq.
Corresponding author E-mail:Bashar.salih@staff.uoz.edu.krd
DOI : http://dx.doi.org/10.13005/ojc/410227
ABSTRACT:Several novel Bis-Oxazepine and Bis-Benzoxazepine derivatives have been produced by condensation and cyclization processes over two effective practical stages. The first stage involved utilizing the Dean-Stark apparatus to create imine intermediates through the condensation reactions of 3-hydroxy benzaldehyde with various aromatic diamine substrates in the presence of glacial acetic acid as a catalyst. The second stage required treatment of bis-Schiff base intermediate with maleic- or phthalic anhydride in dry benzene to produce the desired Bis-Oxazepine and Bis-Benzoxazepine derivatives with 70-95% yields using microwave irradiation. The molecular docking of the produced chemicals was conducted against the progesterone receptor. The results indicate potential anticancer properties for three compounds, which show the most promising findings. These compounds obtained higher docking scores (ΔG −9.58, −9.28, and −9.11 kcal/mol), forming three hydrogen bonds with the target protein.
KEYWORDS:Bis-Schiff bases; Bis-Oxazepine; Bis-Benzoxazepine; Cyclization process; Dean-Stark apparatus; 3-hydroxy benzaldehyde; Microwave irradiation
Introduction
Heterocyclic compounds are essential organic molecules that are used in various applications.1 Oxazepine is a seven-member heterocyclic molecule with one nitrogen atom, one oxygen atom, and five carbon atoms.2 Meanwhile, benzoxazepine is a bicyclic heterocyclic molecule composed of a benzene ring fused with an Oxazepine ring. Oxazepine has three different isomers: (1,2), (1,3), and (1,4), depending on the position of the nitrogen atom. Atoms of oxygen and nitrogen are precisely positioned in the seven-ring, which is the basis for this numbering.3 (Fig.1).
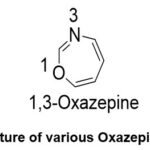 |
Figure 1: Structure of various Oxazepine Derivatives. |
Various Bis-Oxazepine and Bis-Benzoxazepine compounds were synthesized and assessed for different biological activities.4 Some chemical derivatives have demonstrated strong biological activity, like anticonvulsant5, antimicrobial6, anticancer7, antipsychotic agents8, calcium antagonists, and neuroprotective.9 Because of the significance of oxazepine and benzoxazepine, several synthetic procedures for their production have been developed.10
Due to the preparation and biological consequences of Bis-Oxazepine and Bis-Benzoxazepine compounds generating significant attention, our current study is the development, synthesis, and characterization of new Bis-oxazepine and Bis-benzoxazepine derived from Schiff bases11–13, using two critical approaches: condensation1315 and cyclization reaction processes17 (Scheme 1).
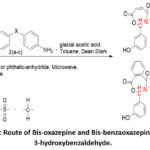 |
Scheme 1: Synthetic Route of Bis-oxazepine and Bis-benzaoxazepine Derivatives from 3-hydroxybenzaldehyde. |
Results and Discussion
Chemistry
This research presents a novel approach for synthesizing bis-oxazepine and Bis-benzoxazepine compounds from bis-Schiff base over two practical processes. First step involved the treatment of 3-hydroxybenzaldehyde with various aromatic diamines 2(a-c) with a few drops of glacial acetic acid. under refluxing with toluene as a solvent and using Dean-Stark apparatus18–20. This step demonstrated a successful technique and supplied pure desired products 3(a-c) (73-88%) followed by a purification process through recrystallization from ethanol (Scheme 2 & Table 1). IR spectra of Schiff bases compounds 3(a-c) Medium bands appeared at 1627-1625 cm-1 attributed to the vibration of stretching of the imine group (C=N). The absorption bands were observed within the A spectrum of (1599-1593), (3047-3036), (2920-2910) cm-1 about the motion of stretching aromatic. (C=N), aromatic(C=N), and aliphatic(C-H) bonds, constantly Fig. 2. The 1HNMR spectra of Schiff bases intermediate 3(a-c) detected new peaks with chemical imine signal positions at (8.57–8.51) ppm, which were compatible with the (CH=N) functional group. The appearance of additional signals for protons in the aromatic region, resulting in chemical shift at (8.01-6.92) ppm due to the aromatic structure of these compounds, Figure 3,4.
 |
Scheme 2: Synthesis of Bis-Schiff Bases from 3-hydroxybenzaldehyde. |
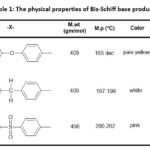 |
Table 1: The physical properties of Bis-Schiff base products 3(a-c). |
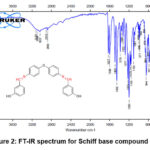 |
Figure 2: FT-IR spectrum for Schiff base compound (3a). |
 |
Figure 3: 1H-NMR (400 MHz, DMSO) Spectrum for Compound (3b). |
 |
Figure 4: 1H-NMR (400 MHz, DMSO) Spectrum for Compound (3c). |
While the Bis-1,3-Oxazepine 6(a-c) and Bis-1,3-Benzoxazepine 7(a-c) products were created at the last step by using microwave irradiation protocol, by reaction of Bis-imines 3(a-c) with maleic- or phthalic-anhydride and a small amount of drying benzene as a solvent after applying the purification method. This step (cyclization process) produced the expected products with acceptable yields 6(a-c) and 7 (a-c) (70-95%) (Scheme 3 and Table 2,3). The 1H-NMR analysis showed new and extra proton Signal peaks related to chemical shifts at (8.61-8.49) and (8.01-6.91) ppm which belong to the CH peak inside the Oxazepine and Benzoxazepine heterocyclic ring and aromatic ring region correspondingly Fig. 5. Moreover, In13C-NMR the appearance peaks of the new carbon signal with the corresponding chemical changes at (169.11-160.11) and (167.98-158.14) ppm for C=O of lactone and lactam respectively, as well as, the appearance of new signal with chemical changes at (114.65-114.64) ppm which belong to the CH in the 1,3-Oxazepine and 1,3-Benzoxazepine heterocyclic ring. Finally, the appearance of extra signal peaks with the chemical shifts at (160.63-115.11) ppm due to the aromatic region, Fig.6, which sustained the structures of the desired products in 6(a-c) and 7(a-c), Scheme 3.
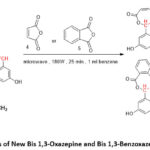 |
Scheme 3: Synthesis of New Bis 1,3-Oxazepine and Bis 1,3-Benzoxazepine from Schiff Bases. |
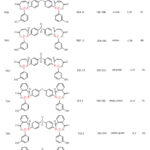 |
Table 2: The physical properties of the synthesized Bis 1,3-Oxazepine 6(a-c) and Bis1,3-Benzoxazepine 7(a-c) from 3-hydroxybenzaldehyde. |
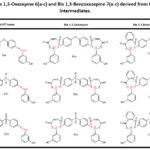 |
Table 3: The Bis 1,3-Oxazepine 6(a-c) and Bis 1,3-Benzoxazepine 7(a-c) derived from Bis-Schiff bases intermediates. |
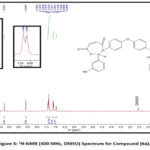 |
Figure 5: 1H-NMR (400 MHz, DMSO) Spectrum for Compound (6a). |
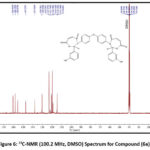 |
Figure 6: 13C-NMR (100.2 MHz, DMSO) Spectrum for Compound (6a). |
Analysis and interpretation of molecular Docking.
A comprehensive set of progesterone receptor (PDB ID: 4OAR) was subjected to docking, leading to the identification of three compounds. Notably, the docking poses of these three hit compounds exhibited interactions remarkably similar to those of other anti-breast cancer compounds. Molecular docking analysis was conducted to assess the potential of these molecules in combating breast cancer. The docking process focused on a phytosterol isolated from Lagerstroemia speciosa seed ethanolic extract, aiming at the 4OAR binding pocket, a protein associated with breast cancer. All tested molecules exhibited binding scores greater than –9.11 when bound to 4OAR, with hydrophobic interactions observed with residues such as LEU718, CYS891, ASN719, ARG899, SER712, LEU887, MET759, and MET756. Notably, Diosgenin, a powerful anti-breast cancer chemical, and its derivatives reached an upper binding score of –9.58, forming hydrogen bonds with 4OAR at GLN725 and GLN916
It is important to note which ligand-protein complexes display the lowest number of hydrophobic interactions when their binding scores are highest. These two- and three-dimensional ligand structures bind most strongly (7c) in complex with the protein presented in (Table 4).21–23 The docking analysis reveals that all compounds in Figures (7,8, and 9) bind to the amino acid active site in breast cancer.24–26 Various types of bonds are involved, including van der Waals, carbon-hydrogen, π-alkyl, and most importantly, hydrogen bonds. Fig. (7,8, and 9).
Table 4: Molecular Docking result data for Bis 1,3-Oxazepine and Bis1,3-Benzoxazepine.
| Comp. No | G[Keal/mol] | Residue |
| 7a | -9.28 | LEU718, CYS891, ASN719, ARG899, SER712, LEU887, MET759 and MET756. |
| 6b | -9.11 | LEU716, LEU715, LEU718, GLN725, MET759, LEU797 and ARG899 |
| 7c | -9.58 | LEU718, CYS891, PHE895, GLN916, ALA915, VAL912, MET759, MET801 and GLN725 |
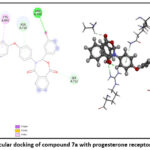 |
Figure 7: Molecular docking of compound 7a with progesterone receptor in 2D and 3D. |
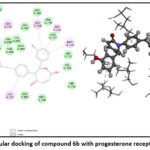 |
Figure 8: Molecular docking of compound 6b with progesterone receptor in 2D and 3D. |
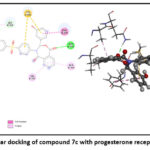 |
Figure 9: Molecular docking of compound 7c with progesterone receptor in 2D and 3D. |
Experimental Materials and Methods
All reactions took place in anhydrous, dry conditions with the use of solvents. Commercial reagents weren’t purified before usage. As an absolute solvent, ethanol was utilized. Electrothermal apparatus has been used for melting points determination, which may be uncorrected. Bruker DPX-300FT-NMR spectrometers have been used to generate 1H- and 13C-NMR spectra at 400 and 100.2 MHz. The Spectrum was recorded in CDCl3. IR spectra were recorded using a 1710-FTIR Perkin-Elmer spectrometer. Merck Kiese gel 60 F254 Thin Layer Chromatography (TLC) on aluminum foil from Macherey-Nagel. UV light at 254 & 365 nanometers was used for detection.
Synthesis
General procedure for synthesizing Bis-Schiff bases 3(a-c)
A solution of 3-hydroxybenzaldehyde 1(0.073 g, 0.6 mmol, 2 eq.) was added by dropping it to an aromatic diamine substitutes solution (2-4) (0.059 g, 0.3 mmol, 1 eq.) in dry toluene (60ml) with stirring using Dean-Stark apparatus at room temp. Subsequently, the mixture was refluxed overnight after being treated with a few drops of glacial acetic acid. It was used to monitor the progress of the reaction by TLC (PE: EtOAc 60:40). The crude products were obtained after evaporating the solvent under the vacuum followed by purification twice using ethanol, Hot filtration, and recrystallizing protocols using ethanol absolute were employed to generate the required Bis-Schiff base products 3(a-c) (Table 1).27–29
3,3′-((oxybis(4,1-phenylene))bis(azanylylidene))bis(methanylylidene))diphenol 3(a)
Pale-yellow, yield (81%), Rf = 0.43, m.p. (165-166) °C. IR (cm-1) = 3126 (O-H broad),3037 (C-Har), 2910 (C-Haliph), 1627 (C=N), 1599-1552 (C=Car), 1200 (C-Ostr), 1HNMR (400 MHz, DMSO) (ppm) = 9.73(s, 2H, 2OH), 8.57 (s, 2H, 2(N=CH)), 7.39-7.30 (m, 10H, CHaro), 7.08 (d, 4H, J = 8 Hz, CHaro), 6.94 ( d.,2H, J = 7.6 Hz, CHaro).
3,3′((methylenebis(4,1phenylene))bis(azanylylidene))bis(methanylylidene))diphenol 3(b)
White, yield = (88 %), Rf .= 0.42, m.p. (197-199) °C. IR (cm-1)= 3273 (O-H broad), 3024 (C-Har), 2907 (C-Haliph), 1624 (C=N), 1591-1576 (C=Caro), 1HNMR (400 MHz, DMSO) (ppm) = 9.71 (s, 2H, 2OH), 8.52 (s, 2H, 2(N=CH)), 7.37-7.20 (m, 14H, CHaro), 6.93 (d, 2H, J= 6.8 Hz, CHaro), 3.98 (s, 2H, CH2).
3,3′-((sulfonylbis(4,1-phenylene))bis(azanylylidene))bis(methanylylidene))diphenol 3(c)
Pink, yield = (73 %), Rf =0.27, m.p. .( 200-202 ) °C. IR. .(cm-1) = 3286 (O-H broad), 3025 (C-Har),2977 (C-Haliph), 1626 (C=N), 1596-1578 (C=Car), 1HNMR (400 MHz, DMSO) (ppm) = 9.79(s, 2H, 2OH), 8.51(s, 2H, 2( N=CH ) ), 7.99 ( d, 4H, J = 8.4 Hz, CHaro), 7.41-7.33 ( m, 10H.CHaro), 6.97 (t, 2H, J= 6.4Hz CHaro).
General procedure for preparation of Bis-Oxazepine 6(a-c) and Bis-Benzoxazepine 7(a-c).
In the crucible with dry benzene (1 ml), appropriate Schiff bases (0.123 mmole, 1eq.) 3(a-c), maleic- or phthalic anhydride (0.246 mmole, 2eq.) were dissolved. The solution was exposed to microwave irradiation at 180W for about 25 min. The reaction process was monitored using TLC (PE: EtOAc 60:40). The crude products were obtained after evaporating the solvent below the vacuum, followed by purification twice using ethanol absolute.to give the expected products 6(a-c) and 7(a-c). The physical properties of Bis-Oxazepine 6(a-c) and Bis-Benzoxazepine 7(a-c) are shown in (Table 2).30–34
3,3′-(oxy bis(4,1-phenylene)) bis(2-(3-hydroxyphenyl)-2,3-dihydro- 1,3-oxazepine-4,7-dione) (6a)
cumin color, yield = (70 % ), Rf = 0.47,m.p. ( 196-198 )°C. IR (cm-1) = 3126 (O-H broad), 3025 (C-Har), 2880 (C-Haliph), 1711 ( C=Olactone),1675 ( C=Olactam), 1598-1451 ( C=Caro), 1HNMR (400 MHz, DMSO) (ppm) = 9.76(s, 2H, 2OH), 8.61 ( s, 2H, 2 (N-CH) ), 7.42-7.43 (m, 13H, CH), 7.13 ( d, 5H. J= 8.4 Hz CHaro), 6.98-6.96 (d, 2H, J= 7.2,CHar).13C NMR (100.2, DMSO) = 160.46 (C=Olactone), 158.15 (C=Olactam),155.58(Car), 147.27(Caro), 137.92(Caro), 130.79(Caro), 130.32(Caro), 123.22(2Caro), 122.28(Caro), 121.73(Caro), 120.71(Caro), 119.72(2Caro), 119.13(Caro), 115.11(Caro), 114.64(C oxazepine ring).
3,3′-(methylenebis(4,1-phenylene))bis(2-(3-hydroxyphenyl)-2,3-dihydro-1,3 oxazepine-4,7-dione) (6b)
Yellow, yield = 83%, Rf = 0.6, m.p.(204-206) °C. IR (cm-1.)= 3273 (O-H broad), 3025 (C-Har),2921 (C-Haliph), 1703 (C=O lactone),1667 (C=O lactam), 1589-1452 (C=Caro), 1HNMR (400 MHz, DMSO) (ppm) = 9.70(s, 2H, 2OH), 8.52 (s, 2H, 2 (N-CH), 7.54 (d, 2H, J= 8.4Hz CHaro), 7.37-7,17 ( m, 16H, CHaro), 6.925 (d, 2H, J= 6.4Hz CHaro).13C NMR (100.2, DMSO) = 160.63 (C=Olactone), 158.14 (C=Olactam),149.85(Caro), 139.71(Caro), 137.93(Caro), 130.31(Caro), 129.94(2Caro), 129.38 (Caro), 128.69(Caro), 125.80(Caro), 121.63(2Caro), 120.69(Caro), 120.19(Caro), 119.11(Caro), 114.64(C oxazepine ring), 40.60(C methane).
3,3′-(sulfonylbis(4,1-phenylene))bis(2-(3-hydroxyphenyl)-2,3-dihydro-1,3-oxazepine-4,7-dione) (6C).
Off-white, yield = 95%, Rf = 0.55, m.p.(230-232)°C. IR (cm-1.)= 3388 (O-H broad), 3049 (C-Haro), 2958 (C-Haliph), 1724 (C=Olactone),1694 (C=Olactam), 1597-1557 (C=Caro), 1HNMR (400 MHz, DMSO) (ppm) = 9.78(s, 2H, 2OH), 8.50. (s, 2H, 2(N-CH)
) , 7.99 (d, 2H, J= 8Hz CHaro), 7.90-7.81 (m, 3H, CHaro), 7.58-6.96 (m, 15H, CHaro). 13C NMR (100.2, DMSO) = 165.06 (C=Olactone), 158.45 (C=Olactam),156.55(Caro), 139.81(Caro), 137.83(Caro), 130.86(Caro), 129.18(2Caro),126.13(Caro), 125.79 (Caro),123.11 (2Caro), 122.49(Caro), 121.17(Caro), 119.50(Caro), 116.18(Caro), 113.47(C-N oxazepine ring).
4,4′-(oxybis (4,1-phenylene))bis(3-(3-hydroxyphenyl) -3,4-dihydrobenzo [e][1,3] oxazepine-1,5-dione) (7a)
Deep Yellow, yield = 75%, Rf =0.41, m.p. (190-192) °C. IR (cm-1) = 3126 (O-H broad), 3025 (C-Haro), 2977 (C-Haliph), 1716 (C=O lactone),1684 (C=O lactam), 1597-1557 (C=Caro). 1H-NMR: d (ppm) = 9.69 (s,2H,2OH), 8.57 (s, 2H, 2(N=CH)), 7.38-7.25 (m, 16H.CHaro), 7.09 (d, 5H, J= 8.4Hz CHaro), 6.95-6.92 (m, 2H, CHaro). 13C NMR (100.2, DMSO) = 160.46 (C=Olactone), 158.15 (C=Olactam) 155.58(Caro), 147.28(Caro), 137.93(Caro), 130.31(Caro), 129.38(Caro), 128.68(Caro), 125.79 (Caro), 123.22 (2Caro), 122.95(Caro),122.27(Caro), 121.30(Caro), 120.70(Caro), 119.72(2Caro), 119.12(Caro), 117.54(Caro), 115.40(Caro), 114.65(C-N oxazepine ring).
4,4′-(methylenebis(4,1-phenylene))bis(3-(3-hydroxyphenyl)-3,4-dihydrobenzo[e] [1,3]oxazepine-1,5-dione) (7b)
Yellow-green, yield =83%, Rf = 0.6, m.p.(202-204) °C. IR (cm-1) 3273 (O-H broad), 3025 (C-Har),2977 (C-Haliph), 1714 (C=O lactone),1667 (C=O lactam), 1597-1557 (C=Car).1H-NMR: d (ppm) = 9.71 (s, 2H, 2OH), 8.52 (s, 2H, 2(N-CH)), 7.36-7.18 (m, 21H, CHaro), 6.92 (d, 3H, J= 6.4Hz, CHaro), 3.98 (s, 2H, CH2). 13C NMR (100.2, DMSO) = 169.11 (C=Olactone) ,167.98 (C=Olactam), 160.63(Caro), 158.15(Caro), 149.85(Caro), 139.71(Caro), 137.93(Caro), 135.16(Caro), 130.31 (Caro), 129.94 (Caro), 129.38(Caro),128.69(Caro), 125.80(Caro),122.28(Caro), 121.62(Caro), 120.70(Caro), 120.10(Caro), 119.11(Caro), 114.64(C-N oxazepine ring), 40.59 (CH2).
4,4′-(sulfonyl bis (4,1-phenylene))bis (3-(3-hydroxyphenyl)-3,4 dihydrobenzo[e] [1,3]oxazepine-1,5-dione) (7C)
Pale-Yellow, yield. = 93%, Rf = 0.41, m.p. (216-218) °C. IR (cm-1) = 3374 (O-H broad), 3025 (C-Har), 2958 (C-Haliph), 1690 (C=O lactone),1686 (C=O lactam), 1597-1557 (C=Caro). 1HNMR: d ppm. = 9.76 (s, 2H, 2OH), 8.50. (s, 2H, 2(N-CH)), 7.84 (d, 3H, J= 8 Hz, CHaro), 7.56 (d, 3H, J= 8, CHaro), 7.45-7.33 (m,17H, CHaro). 13C NMR (100.2, DMSO) = 165.06 (C=Olactone), 158.45 (C=Olactam),158.19(Caro), 147.28(Caro),138.47(Caro), 137.22(Caro), 130.80(Caro), 129.18(Caro), 129.14(2Caro), 128.33(Caro), 126.13(Caro), 125.79 (Caro), 123.11 (2Caro),122.46(Caro), 121.17(Caro), 120.49(Caro), 119.12(Caro), 115.06(Caro), 114.43(C-N oxazepine ring)
Molecular Docking
All stationary points involved in the studied reactions were optimized with DFT method using the B3LYP functional in conjunction with 6-31G(d) basis set35 The calculations were done using Gaussian 16 software.36,37 Molecular docking of the synthesized compounds against progesterone receptor (PDB ID: 4OAR) was executed utilizing Auto Dock Vina.38 The geometry of the X-ray crystal of the progesterone receptor for docking purposes, it was obtained as a PDB file through the Protein Data Bank. 39 Thereafter, to eliminate water molecules and initial inhibitors from the structure of the protein of interest. Following the elimination of all the molecules of water, polar hydrogen molecules were included, and charges were allocated utilizing the Kollman unified atom library via Auto Dock Vina Tools (ADT; version 1.5.4).40 The poses of the docked protein-ligand complexes in 2D and 3D have been observed using the Discovery Studio Client visualization software.41
Conclusion
In conclusion, we have disclosed an effective approach to the synthesis of Bis-Oxazepine and Bis-Benzoxazepine derivatives. This process started by synthesizing different derivatives of Bis-Schiff bases through the reaction of 3-hydroxybenzaldehyde with several aromatic diamine substrates over the condensation process and using the Dean-Stark apparatus. Secondly, the cyclization of Bis-Schiff bases with maleic- and phthalic anhydride afforded the desired oxazepine and benzoxazepine products using microwave irradiation, which gives high yields, short time, safety, and without using high amounts of solvent. The synthesized compounds were exposed to molecular docking against the progesterone receptor. The three final compounds 6b, 7a, and 7c indicate potential anticancer properties with higher docking scores (ΔG −9.58, −9.28, and −9.11 kcal/mol), forming three hydrogen bonds with the target protein.
Acknowledgment
The authors express thanks to the University of Zakho, College of Science, and Department of Chemistry for giving the essential benefits.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Kabir E, Uzzaman M. A review on biological and medicinal impact of heterocyclic compounds. Results Chem. 2022;4:100606.
CrossRef - Alobaidy RM. Synthesis and Spectroscopic Identification of New bis-Oxazepines. Coll Basic Educ Res J. 2020;16(4):1075-1088.
CrossRef - Mahapatra DK, Shivhare RS, Gupta SD. Anxiolytic activity of some 2, 3-dihydrobenzo [b][1, 4] oxazepine derivatives synthesized from Murrayanine-Chalcone. Asian J Res Pharm Sci. 2018;8(1):25-29.
CrossRef - Alja’afreh IY, Qasem IAS, Tarawneh AH, Fronczek FR, Ashram M. Synthesis, Biological Activity and Crystal Structure Studies of Some Benzoxazepine Derivatives. Russ J Bioorganic Chem. 2023;49(2):376-383.
CrossRef - Garg N, Chandra T, Jain AB, Kumar A. Synthesis and evaluation of some new substituted benzothiazepine and benzoxazepine derivatives as anticonvulsant agents. Eur J Med Chem. 2010;45(4):1529-1535.
CrossRef - Sager AG, Abaies JK, Katoof ZR. Molecular Docking, Synthesis and Evaluation for Antioxidant and Antibacterial Activity of New Oxazepane and Benzoxazepine Derivatives. Baghdad Sci J. Published online 2023.
CrossRef - Odame F, Schoeman R, Krause J, Hosten EC, Tshentu ZR, Frost C. Synthesis, characterization, crystal structures, and anticancer activity of some new 2, 3-dihydro-1, 5-benzoxazepines. Med Chem Res. 2021;30:987-1004.
CrossRef - Merabtine T, Tarhini Z, Preux PM, Christou N, Jost J. Effects of antidepressant and antipsychotic medication on peripheral brain-derived neurotrophic factor concentration: Systematic review and meta-analysis. Psychiatry Res. Published online 2024:115946.
CrossRef - Chatterjee I, Ali K, Panda G. A Synthetic Overview of Benzoxazines and Benzoxazepines as Anticancer Agents. ChemMedChem. 2023;18(5):e202200617.
CrossRef - Stefaniak M, Olszewska B. 1, 5‐Benzoxazepines as a unique and potent scaffold for activity drugs: A review. Arch Pharm (Weinheim). 2021;354(12):2100224.
CrossRef - HAJI RB, ALI MK, MOHAMMED SR. Synthesis and Spectroscopy Characterizations of Some New Bis 1, 3-thiazolidin-4-ones Derived from 4-hydroxybenzaldehyde Substrate. Orient J Chem. 2023;39(5).
CrossRef - Rojin B, Maher K, Shireen M. Synthesis of Some New Bis-4-Thiazolidinones Derived from 2-Hydroxy-1-naphthaldehyde. Indian J Heterocycl Chem. 2022;32(03):357-362.
- Beniazza R, Liautard V, Poittevin C, et al. Free‐radical carbo‐alkenylation of olefins: Scope, limitations and mechanistic insights. Chem Eur J. 2017;23(10):2439-2447.
CrossRef - Hanoon HD, Abd Al Hussain HA, Abass SK. Synthesis and characterization of azetidin-2-one and 1, 3-oxazepine derivatives using Schiff bases derived from 1, 1’-biphenyl-4, 4’-diamine. In: Journal of Physics: Conference Series. Vol 2063. IOP Publishing; 2021:12010.
CrossRef - Othman LA, Mohammed SR, Khalid M. Synthesis and Characterization of Some New Quinoline Derivatives Derived from 2-Amino Benzonitrile. Indian J Heterocycl Chem. 2022;32(04):487-492.
- OTHMAN LA, MOHAMMED SR, ALI MK. A Flexible Route to Synthesis and Molecular Docking of Some New Quinoline Derivatives through Imine and Cyclization Processes. Orient J Chem. 2023;39(2).
CrossRef - Hanoon HD. Synthesis and characterization of new seven-membered heterocyclic compounds from reaction of new Schiff-bases with maleic and phthalic anhydrides. Iraqi Natl J Chem. 2011;11(41):77-89.
- Bakht MA. Lemon juice catalyzed ultrasound assisted synthesis of Schiff’s base: a total green approach. Bull Env Pharmacol Life Sci. 2015;4(10):79-85.
- Mohammed S. A Novel Synthetic Route of Fused Tricyclic Framework Quinoline Derivatives from Readily Available Aliphatic Amino Carboxylic Acid Substrates. Orient J Chem. 2019;35(2):611.
CrossRef - Mohammed SR. Development of new radical processes: approaches toward the synthesis of Eucophylline. Published online 2014.
- Ramkumar S, Ramarajan R. Design, Synthesis, Spectral Characterization, Antioxidant Activity, Molecular Docking and in silico ADMET Studies of 1, 3 Oxazepines. ChemistrySelect. 2023;8(9):e202204818.
CrossRef - Hassan SA, Aziz DM, Kader DA, Rasul SM, Muhamad MA, Muhammedamin AA. Design, synthesis, and computational analysis (molecular docking, DFT, MEP, RDG, ELF) of diazepine and oxazepine sulfonamides: Biological evaluation for in vitro and in vivo anti-inflammatory, antimicrobial, and cytotoxicity predictions. Mol Divers. Published online 2024:1-23.
CrossRef - Noser AA, Abdelmonsef AH, Salem MM. Design, synthesis and molecular docking of novel substituted azepines as inhibitors of PI3K/Akt/TSC2/mTOR signaling pathway in colorectal carcinoma. Bioorg Chem. 2023;131:106299.
CrossRef - Azzawi MH, Hussein MS. Synthesis and Cytotoxicity Evaluation of Pyrazole Compounds Bearing Oxazepine Core Against Breast Cancer Cells. Int J Health Sci (Qassim). 6(S6):6984-6996.
CrossRef - Muhee AA, Kadhim MI. Synthesis, antibacterial and Molecular docking of some new tetrazole, oxazepine and thiazolidine derivatives contacting with aromatic nucleus. Chem Rev Lett. 2024;7(6):647-654.
- Sunil D, Ranjitha C, Rama M, KSR P. Oxazepine derivative as an antitumor agent and snail1 inhibitor against human colorectal adenocarcinoma. Int J Innov Res Sci Eng Technol. 2014;3(8):15357-15363.
CrossRef - Jagrut VB, Kale AD, Sadawarte GP, Phase RP. Micelle promoted synthesis of schiff base ligands derived from 4-methyl-1, 2, 3-thiadiazoles-5-carboxylic acid hydrazide in bio based green media. Chem Rev Lett. 2024;7(6):957-963.
- Gharge S, Alegaon SG, Ranade SD, Kavalapure RS, Kumar BRP. Novel rhodanine–thiazole hybrids as potential antidiabetic agents: a structure-based drug design approach. RSC Med Chem. Published online 2025.
CrossRef - Kaur M, Sood K, Yempally V, Kaur H. Cytotoxicity Profile of Schiff Base Organotin (IV) Complexes: Experimental and Theoretical Approach. J Fluoresc. Published online 2025:1-13.
CrossRef - Taha NI. Synthesis of 1, 3-oxazepine derivatives derived from 2-(1H-benzo [d][1, 2, 3] triazol-1-yl) acetohydrazide by using microwave irradiation. Int J Org Chem. 2017;7(03):219-228.
CrossRef - Mollo MC, Orelli LR. Microwave-Assisted Synthesis of 2-Aryl-2-oxazolines, 5, 6-Dihydro-4 H-1, 3-oxazines, and 4, 5, 6, 7-Tetrahydro-1, 3-oxazepines. Org Lett. 2016;18(23):6116-6119.
CrossRef - Dalaf AH, Jumaa FH, Yass IA. Synthesis, characterization, biological evaluation, molecular docking, assess laser efficacy, thermal performance and optical stability study for new derivatives of bis-1, 3-oxazepene and 1, 3-diazepine. In: AIP Conference Proceedings. Vol 2394. AIP Publishing; 2022.
CrossRef - Hassan SA, Aziz DM, Abdullah MN, et al. Design and synthesis of oxazepine derivatives from sulfonamide Schiff bases as antimicrobial and antioxidant agents with low cytotoxicity and hemolytic prospective. J Mol Struct. 2023;1292:136121.
CrossRef - Muhammad FM, Khairallah BA, Albadrany KA. Synthesis, characterization and Antibacterial Evaluation of Novel 1, 3-Oxazepine Derivatives Using A Cycloaddition Approach. J Angiother. 2024;8(3):1-5.
CrossRef - Hehre WJ. Ab initio molecular orbital theory. Acc Chem Res. 1976;9(11):399-406.
CrossRef - Becke AD. Density‐functional thermochemistry. I. The effect of the exchange‐only gradient correction. J Chem Phys. 1992;96(3):2155-2160.
CrossRef - Lee C, Yang W, Parr RG. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B. 1988;37(2):785.
CrossRef - Tiwari A, Tiwari V, Sharma A, et al. Tanshinone-I for the treatment of uterine fibroids: Molecular docking, simulation, and density functional theory investigations. Saudi Pharm J. 2023;31(6):1061-1076.
CrossRef - Acharya R, Chacko S, Bose P, Lapenna A, Pattanayak SP. Structure based multitargeted molecular docking analysis of selected furanocoumarins against breast cancer. Sci Rep. 2019;9(1):15743.
CrossRef - Pathoor R, Bahulayan D. MCR-click synthesis, molecular docking and cytotoxicity evaluation of a new series of indole–triazole–coumarin hybrid peptidomimetics. New J Chem. 2018;42(9):6810-6816.
CrossRef - Baroroh U, Biotek M, Muscifa ZS, Destiarani W, Rohmatullah FG, Yusuf M. Molecular interaction analysis and visualization of protein-ligand docking using Biovia Discovery Studio Visualizer. Indones J Comput Biol. 2023;2(1):22-30.
CrossRef
Second Review by: Dr. Pinak Dutta
Final Approval by: Dr. Abdelwahab Omri

















