Synthesis and Characterization of Excess and Deficit Of Iron in LaFeO3 [LaFe1.05O3 and LaFe0.95O3] Inorganic Perovskite Materials Using Simple and Efficient Sol-gel Technique with Magnetic Properties
1Department of Chemistry, University College of Science, Osmania University, Hyderabad, Telangana, India
2Department of Chemistry, Veeranari Chakali Ilamma Women’s University, Koti, Hyderabad, Telangana, India
3Department of Integrated Chemistry, University P.G. College, Palamuru University, Mahbubnagar, Telangana, India
4Department of Chemistry, Vardhaman College of Engineering, Hyderabad, Telangana, India.
Corresponding Author E-mail:patriji2095@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/410207
ABSTRACT:The sol-gel method was used to create LaFeO₃ (LFO) perovskites with abundant iron and deficit iron (LaFe1.05O3 and LaFe0.95O3). Vibrating sample magnetometry (VSM) analysis, X-ray Photoelectron Spectroscopy (XPS), UV-Visible Diffuse Reflectance Spectrometry,Powder X-ray Diffraction and Field Emission Scanning Electron Microscopy and Energy Dispersive Spectroscopy were used to thoroughly describe the materials.The orthorhombic-shaped iron-modified LFO perovskites that crystallised in the Pnma space group were successfully synthesised, as proven by the p-XRD. The FESEM investigation revealed uneven morphology and phase purity. EDS and XPS examination of the following perovskites elemental composition, surface structure, and valence state of the chemical. UV-Vis DRS was used to determine the energy band gap of LaFe1.05O3 (1.98 eV) and LaFe0.95O3 (2.03 eV). Iron-deficient LaFeO3 (LaFe0.95O3) showed paramagnetic behaviour with increased saturation magnetisation (MS), while LaFe₁.₀₅O₃ (with abundant iron) shows weak ferromagnetic behaviour according to VSM research.
KEYWORDS:Iron Excess; Iron Deficiency; Magnetic characteristics; Perovskites; Paramagnetism; VSM analysis
Introduction
Both too much and too little iron using powder XRD, X-Ray Photoelectron Spectroscopy, FESEM-EDS, UV-Visible Diffuse Reflectance spectrometry, VSM. LaFeO3 perovskites were created using the sol-gel method. This study’s results suggest that the sol-gel method could be used to make fine above perovskites with a 98% yield. The above perovskites having an orthorhombic configuration that crystallised inside the orthorhombic Pnma space group were successfully synthesised, according to the results11,12. La+3 has eight coordinates and is joined to eight O2 atoms. The current work aims to prepare iron-based perovskites and is an attempt in that direction. Higher saturation magnetisation (MS) and paramagnetic behaviour were demonstrated by these synthesised perovskites, which also absorbed more visible light during photodegradation. Therefore, in the process of photodegradation, these perovskites function as a photocatalyst.
The impact of stoichiometry for iron on the structural and magnetic characteristics of perovskites with LFO which are produced by an effective sol-gel process is clarified by this work.
Materials and Methods
Synthesis of pure iron excess and deficit in LaFeO3 perovskites by the sol-gel method
Sol-gel synthesis of iron excess and deficit in LFO perovskites
The perovskite LFO powders were created using calcination and sol-gel techniques. Five grammes of La(NO3)3. 6H2O and 4.8 g of Fe(NO3)3. 9H2O (in a ratio of 1:1.05), as well as 5 g of La(NO3)3. 6H2O and 4.4 g of Fe(NO3)3. 9H2O (in a ratio of 1:0.95), were the starting materials. They were dissolved in 200 millilitres of ethyl alcohol in a 500 millilitre beaker and left to stir at room temperature for two hours. 5.05g citric acid solution was introduced to the mixture. After 5.5 g of citric acid had been stirred for two hours. A soluion of ammonia was introduced to adjust the pH scale down to 7 at 70°C after an hour of stirring. 3.2 millilitres of the solution of ethylene glycol was added to 100 millilitres of a 50% solution at 160°C after 3.6 millilitres of ethylene glycol had been stirred for an hour. Before being grounds for calcinations in a muffle furnace at 800°C for eight hours, the mixture was agitated until it turned black. As a result, the powdered LFO perovskite materials had a 98% absolute excess and deficit of iron.
The following reaction occurs in the creation of excess iron in LaFe1.05O3 perovskite material:
The following reaction occurs in the creation of deficit iron in LaFe1.05O3 perovskite material:
Results and discussions
UV- visible spectroscopy
indicates that LFO produced using this technique is active as a photocatalyst.
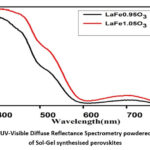 |
Figure 1: UV-Visible Diffuse Reflectance Spectrometry powdered samples of Sol-Gel synthesised perovskitesClick here to View Figure |
Band gap energy
Plots of Kubelka-Munk (KM) of synthesised perovskites15 are displayed in Fig. 2. The bandgaps of LFO nanoparticles with excess and deficient iron are 1.98 eV and 2.03 eV, accordingly. These values are helpful for more photocatalysis research because they are less than the orthorhombic LFO perovskites bandgap energies.
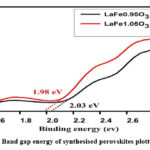 |
Figure 2: Band gap energy of synthesised perovskites plotted by KM.Click here to View Figure |
XRD
By contrasting the outcomes with the typical JCPDS card numbers 88-0641, respectively16, diffraction of X-ray examination verified the powder was made up of synthesised compounds that have an orthorhombic configuration that crystallized in the group of an orthorhombic Pnma space. Crucially, the perovskite structure was the only secondary phase detected. Using the formula where (d) is the interplanar distance, a,b,c and h,k,l are lattice variables and indices of crystallographic planes17, compound’s lattice parameters were found using X-Ray Diffraction Pattern. The sol-gel approach yielded powdered synthesised perovskites with a= 5.5647 Ao , b=7.8551 Ao, c= 5.556 Ao values. The unit cell volumes of the synthesised chemicals are 242.86 Ao3. Synthesised perovskites exhibit orthorhombic structure in their X-ray diffraction (XRD) patterns (Fig. 3).
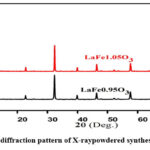 |
Figure 3: The diffraction pattern of X-raypowdered synthesisedperovskites.Click here to View Figure |
FESEM and EDS
Iron overload and deficiency in LFO perovskite18 were investigated using FESEM and EDS spectra. Large, irregularly shaped lumps that adhere to one another are shown in FESEM pictures of excess LFO perovskite and iron deficiency. The EDS analysis confirms the accurate amounts of La, Fe, and O chemicals in pure LaFeO₀ perovskites. Each element’s weight proportion is shown in the relevant EDS statistics. Figures 4 and 5 show that the FESEM and EDS results for iron-containing LFO perovskites excess and deficit don’t change much.
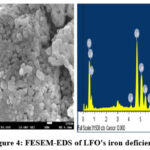 |
Figure 4: FESEM-EDS of LFO’s iron deficiency.Click here to View Figure |
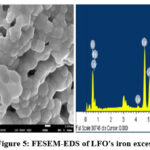 |
Figure 5: FESEM-EDS of LFO’s iron excessClick here to View Figure |
XPS
Data on the binding energies of synthetic materials are shown in panels a–d of Figures 6 and 7. The iron excess and deficit of the LFO-800 nanostructure XPS survey spectrum are shown in Figures 6a and 7a. The coexistence of lanthanum (La), oxygen (O), and iron (Fe) in the sample is approved, and distinct elemental peaks are not identified. The basic spectra of La3+ In the oxide shape were assigned to the two significant signals that La displayed simultaneously at 836.94 eV, 865.73 eV(Fig. 6b and 7b)19. Based on the high- firmness of XPS analysis in iron (Figs. 6c,7c) which displayed distinct peaks with two binding energies of Fe 2p3/2 and Fe 2p1/2 at 710.86 eV, 724.70 eV, and 743.65 eV (standard values: Fe 2p1/2 – 852.6 eV; Fe 2p3/2 – 869.3 eV), Fe is present in Fe3+ oxidation states in the LaFeO3-80020. The fitted binding energies also showed lower and higher binding energies after La-Fe interactions. Located at 530.30 eV, 532.10 eV two peaks in Figures 6d and 7d indicated the presence of oxygen (O2-) in the LaFeO3 perovskite lattice. Utilising the aforementioned helpful information from the XPS survey is recommended. That is very similar to the excess of LFO and the chemical structure of iron deficiency.
 |
Figure 6: LFO-800 XPS spectra photocatalyst’s iron deficiency: Survey spectra (a) La3d (b)Fe 2p (c), O 1s (d).Click here to View Figure |
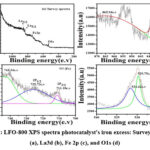 |
Figure 7: LFO-800 XPS spectra photocatalyst’s iron excess: Survey spectra (a), La3d (b), Fe 2p (c), and O1s (d).Click here to View Figure |
VSM analysis
The magnetisation Field (H) and (M) hysteresis loops seen in Figure 8 were recorded at 25 0C using a vibrating sample magnetometer (VSM). As stated by Gehring21,22 and The statistical distribution of Fe3+ in the octahedron structure may be the origin of bulk magnetisation and mild ferromagnetism, and the development of lattice defects may also lead to these phenomena. Figure 8 in this paper displays the room temperature magnetisation for synthesised perovskites at different magnetic fields of 20 kOe. LaFe₀.₉₅O₃ (with iron deficit) shows paramagnetic behaviour and higher saturation magnetisation (Mₛ) among the two samples under study, while LaFe₁.₀₅O₃ (with abundant iron) shows weak ferromagnetic behaviour, as reported. LaFe₀.₉₅O₃ perovskites function as efficient photocatalysts by, while is occurring, absorbing more visible light.
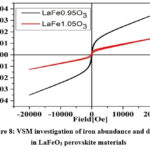 |
Figure 8: VSM investigation of iron abundance and deficit in LaFeO3 perovskite materials.Click here to View Figure |
Conclusion
This study shows how the amount of iron in an LFO perovskites mixture changes its structure and magnetic properties when it is made using a good sol-gel method. An FESEM and p-XRD analysis showed that the compounds in question had an irregular shape, an orthorhombic structure, and a pure phase. Using EDS and XPS analysis, Elemental composition tracked down surface structure, valence state of chemical the important components of the aforementioned perovskites. Iron-deficient LaFexO3 (LaFe0.95O3) showed paramagnetic behaviour with increased saturation magnetisation (MS), while LaFe₁.₀₅O₃ (with abundant iron) shows weak ferromagnetic behaviour according to VSM research.
These results should be expanded by more research on how iron stoichiometry affects other functional characteristics like electrical conductivity or catalytic activity.
Acknowledgment
For providing the required infrastructure, the authors would like to thank CFRD-OU and the Department of Chemistry at Osmania University.
Conflict of Interest
Any financial, personal, or other links with other individuals or organisations that may have an impact on their work are among the conflicts of interest that all writers are asked to declare.
References
- Shi, D.; Adinolfi, V.; Comin, R.; Yuan, M.; Alarousu, E.; Buin, A.; Chen, Y.; Hoogland, S.; Rothenberger, A.; Katsiev, K.; Losovyj, Y.; Zhang, X.; Dowben, P. A.; Mohammed, O. F.; Sargent, E. H.; Bakr and O. M., Science 2015, 347, 519-522
CrossRef - Yang, W. S.; Park, B. W.; Jung, E. H.; Jeon, N. J.; Kim, Y. C.; Lee, D. U.; Shin, S. S.; Seo, J.; Kim, E. K.; Noh, J. H.; Seok and S. I., Science 2017, 356, 1376-1379
CrossRef - Heo, S.; Seo, G.; Lee, Y.; Lee, D.; Seol, M.; Lee, J.; Park, J. B.; Kim, K.; Yun, D. J.; Kim, Y. S.; Shin, J. K.; Ahn, T. K.; Nazeeruddin and M. K., Energy Environ. Sci. 2017, 10, 1128-1133
CrossRef - Saliba, M.; Matsui, T.; Domanski, K.; Seo, J.-Y.; Ummadisingu, A.; Zakeeruddin, S. M.; Correa-Baena, J.-P.; Tress, W. R.; Abate, A.; Hagfeldt, A and Gratzel, M., Science 2016, 354, 206-209
CrossRef - Alharbi, E. A.; Alyamani, A. Y.; Kubicki, D. J.; Uhl, A. R.; Walder, B. J.; Alanazi, A. Q.; Luo, J.; Burgos-Caminal, A.; Albadri, A.; Albrithen, H.; Alotaibi, M. H.; Moser, J. E.; Zakeeruddin, S. M.; Giordano, F.; Emsley, L and Gratzel, M., Commun. 2019,10, 3008
CrossRef - Ahn, N.; Son, D. Y.; Jang, I. H.; Kang, S. M.; Choi, M.; Park and N. G., Am. Chem. Soc. 2015, 137, 8696-8699
CrossRef - Huang, P.; Chen, Q.; Zhang, K.; Yuan, L.; Zhou, Y.; Song, B. and Li. Y., Mater. Chem. A. 2019, 7, 6213-6219
CrossRef - Park, B. W.; Philippe, B.; Jain, S. M.; Zhang, X.; Edvinsson, T.; Rensmo, H.; Zietz, B.; Boschloo, G. , Mater. Chem. 2015, 3, 21760-21771
CrossRef - Liu, X.; Wang, Y.; Xie, F.; Yang, X.; Han, L., ACS Energy Lett. 2018, 3, 1116-1121
CrossRef - Guerrero, A.; You, J.; Aranda, C.; Kang, Y. S.; Garcia-Belmonte, G.; Zhou, H.; Bisquert, J.; Yang, Y., ACS Nano. 2016, 10, 218-224
CrossRef - Shi, D.; Adinolfi, V.; Comin, R.; Yuan, M.; Alarousu, E.; Buin, A.;Chen, Y.; Hoogland, S.; Rothenberger, A.; Katsiev, K.; Losovyj, Y.;Zhang, X.; Dowben, P. A.; Mohammed, O. F.; Sargent, E. H.; Bakr, O.M., 2015, 347, 519-522
CrossRef - Aristidou, N.; Eames, C.; Sanchez-Molina, I.; Bu, X.; Kosco, J.; Islam, M. S.; Haque, S. A., Commun. 2017, 8, 15218
CrossRef - Yang, W. S.; Park, B.-W.; Jung, E. H.; Jeon, N. J.; Kim, Y. C.; Lee, D. U.; Shin, S. S.; Seo, J.; Kim, E. K.; Noh, J. H., 2017 ,356, 1376-1379
CrossRef - Prasanna, R.; Gold-Parker, A.; Leijtens, T.; Conings, B.; Babayigit, A.; Boyen, H. G.; Toney, M. F.; McGehee, M. D., Am. Chem. Soc. 2017 , 139, 11117- 11124
CrossRef - Mekhemer, Gamal AH, Scientific Reports. 2023,1 ,7453
CrossRef - Hammouda, Samia Ben, Applied Catalysis B: Environmenta. 2018,233, 99-111
CrossRef - Kojčinović, Jelena, Materials Chemistry Frontiers. 2022, 9, 1116-1128
CrossRef - Wu, Mudi, Science of The Total Environment. 2021, 795 ,148904
CrossRef - Abdel-Khalek, E. K., Physica B: Condensed Matter. 2022, 624, 413415
CrossRef - Kim, Bae-Jung, Journal of the American Chemical Society. 2019, 13, 5231-5240
CrossRef - Chang, Hui, ACS catalysis, 2020, 6 , 3707-3719
CrossRef - Nenning, Andreas, The Journal of Physical Chemistry C, 2016, 3 , 1461-1471
CrossRef
Accepted on: 28 Mar 2025
Second Review by: Dr. Rafid S. Dawood
Final Approval by: Dr. Abdelwahab Omri

















