Development of Quinoline Linked Hybrid: Synthesis Characterization and Antiparasitic Potential
1Department of Molecular Biology and Biotechnology, BAC Sabour, Bihar Agricultural University Sabour, Bhagalpur 813210, Bihar, India,
2Pharmacy Academy, Faculty of Pharmacy, IFTM University, Moradabad, Uttar Pradesh, India.
3Department of Pharmacy-IBMER, Mangalaytan University Aligarh, Uttar Pradesh, India.
4Department Pharmaceutical Chemistry, Bansal College of Pharmacy Bhopal, Madhya Pradesh, India.
5Department of Pharmacognosy, Teerthanker Mahaveer College of Pharmacy, Teerthanker Mahaveer University, Delhi Road Moradabad, Uttar Pradesh, India.
6Department of Biochemistry, Symbiosis Medical College For Women, Symbiosis International (Deemed University), Pune, India.
7Nagpur College of pharmacy, wanadongri, Hingna road, Nagpur, Maharashtra, India.
8Career Point School of Pharmacy, Career Point University, Kota, Rajasthan, India.
9Department of Biochemistry, School of Life Sciences, Vels Institute of Science, Technology and Advanced Studies, Chennai, Tamil Nadu, India
Corresponding Author E-mail:shobana.sls@vistas.ac.in
DOI : http://dx.doi.org/10.13005/ojc/420106
ABSTRACT:Parasitic diseases have been a significant issue to world health especially in the tropical and subtropical areas, where the existing chemotherapeutic agents have proved to have limitations such as drug resistance, drug toxicity, and poor efficacy thus rendering disease control ineffective. In this regard, quinoline-based compounds have remained of significant interest based on their established antiparasitic and long-standing clinical efficacy. The current review gives an overview of the quinoline-linked hybrid molecules as the future antiparasitic agents with a focus on their design, synthesis procedures, characterizations, and biological performance. Quinoline is a privileged scaffold that possesses desirable chemical and pharmacological properties and by hybridizing with various partner pharmacophores, is a rational approach to increase potency, expand target interactions, and decrease resistance liability. The major synthetic strategies, starting with the classical quinoline-forming reactions, all the way to the current coupling, click chemistry and green methodology are addressed in the framework of efficient hybrid construction. The review also discusses in vitro and in vivo antiparasitic evaluation plans, the use of standardized measurements of potency, the evaluation of selectivity, pharmacokinetics and safety profiling. Mechanistic data on the antiparasitic activity such as inhibition of heme detoxification, impairment of mitochondrial activity, stimulation of oxidative stress, synergetic multi-target effects by partner pharmacophores is critically examined. Lastly, the existing issues, future opportunities and translation concerns are discussed, to inform rational optimization and clinical improvement. Taken together, this review highlights the potential of quinoline-based hybrids as a flexible and potent platform towards development of next-generation antiparasitic therapeutics.
KEYWORDS:Antiparasitic agents; Drug resistance; Molecular hybridization; Quinoline hybrids; Synthetic methodologies; Structure–activity relationship
Introduction
Quinoline and its analogues have been at the center stage of drugs with diverse biological effects and extensive clinical interest because of their broad spectrum of tissue activity and long history of clinical use.1 The quinoline nucleus is a highly preferred heterocyclic structure that has been used to design some of the most iconic drugs, especially in the treatment of antiparasitic therapy segment. The principle of interaction of this framework with key parasitic targets and interference with key biochemical pathways is clearly demonstrated by classic antimalarial agents like quinine and chloroquine, and subsequent generations of synthetic quinoline analogues.2 Quinoline possesses structural versatility and good physicochemical characteristics including planarity, aromaticity, and ability to undergo a variety of substitutions, that is why quinoline is a core of rational design to create new therapeutic agents.3 The problem of parasitic disease is still a significant issue of global concern (preserving its relevance in the tropical and subtropical areas), with protozoan parasitism, which is associated with Plasmodium, Leishmania and Trypanosoma, being a significant cause of major morbidity and mortality.4 Although chemotherapy has improved greatly, the efficacy of the available antiparasitic medications is getting weak due to development of drug resistance, insufficient activity on other stages of the life-cycle, toxicity, and high cost of treatment. Malaria, leishmaniasis, and trypanosomiasis are still an extremely significant burden on health care systems, and there is an urgent necessity to develop new efficient and cost-effective treatment methods. In that respect, the discovery and design of novel chemical molecules with enhanced potency and safety profiles is among the priorities of the modern drug discovery research. A potential solution to these problems is the so-called molecular hybridization concept, which means the covalent connection of two or more active pharmacological moieties into one molecular structure.5,6 Quinoline-linked hybrids are created as a combination of the pharmacodynamic benefits of the quinoline scaffold and those of another biologic partner, possibly leading to increased efficacy, multitarget activity, and decreased chances of developing resistance. Hybrid molecules can also concurrently regulate multiple biological pathways/targets by bringing together different pharmacophores into one molecule, a feature especially beneficial in complicated parasitic infections where biochemical network redundancy and plasticity can compromise monotherapy. The justification of the development of quinoline-based hybrids is also justified by a considerable amount of evidence which proves the wide-ranged antiparasitic potential of quinoline derivatives.8,9 The quinoline compounds are also reported to disrupt several important processes in the parasites including heme detoxification, DNA replication, mitochondrial functionality and redox homeostasis.10,11 Combined with other pharmacophores, e.g. triazoles, chalcones, coumarins, benzothiazoles or other heterocyclic systems, the resultant hybrids can have synergistic or addictive effects which are stronger than those of the starting compounds. Further, pharmacokinetics properties can be optimized with the help of hybridization, selectivity in favor of the parasite cell against host cell can be increased, and adverse effects can be reduced. The development of synthetic organic chemistry has enabled the synthesis of hybrids containing quinoline under a large structural diversity to a great extent.11 Complex hybrid molecules can be made efficiently and reproducibly in modern methodologies, such as click chemistry, metal-catalyzed cross-coupling reactions, and one-pot multicomponent reactions. Besides being applicable in the high-speed production of compound libraries in biological screening, these synthetic innovations provide structure-activity relationship (SAR) studies that are critical in rational optimization.2
Coupled with synthesis, strong characterization via spectroscopic and analytical methods including nuclear magnetic resonance spectroscopy, mass spectrometry, infrared spectroscopy and X-ray crystallography is to provide sound structural confirmation and quality evaluation of newly synthesized hybrids.13 Simultaneously with synthetic developments, systematic biological testing has demonstrated proactive antiparasitic properties of a wide range of quinoline conjugated hybrids in both in vitro and in vivo models. A few studies describe nanomolar to low micromolar potency with drug-sensitive and drug-resistant strains of parasites with desirable selectivity indices. These results support the promising future of quinoline hybrids as a second generation antiparasitic drug. Nevertheless, variability in experimental models, assay conditions and reporting standards across studies may complicate direct comparison and this has highlighted the importance of extensive reviews that critically analyze and bring together existing data.14 Against this backdrop, the current review will seek to deliver a comprehensive and recent review in the evolution of the quinoline-linked hybrids as antiparasitic agents. The review is based upon design strategies, synthetic methods, and characterization methods and then their antiparasitic potential, mechanisms of action, and structure-activity relate are critically discussed.15 The article aims to provide important information to researchers working in the fields of medicinal chemistry, pharmacology, and drug discovery as well as to facilitate the logical further development of quinoline-based hybrid molecules into a clinically effective antiparasitic treatment, based on the consolidation of scattered literature and an emphasis on the existing challenges and future perspectives.16,17
Quinoline scaffold: chemical features and drug-like properties
Core quinoline chemistry and common substitution patterns
Quinoline scaffold is a bicyclic heteroaromatic structure formed by a benzene ring fused with a pyridine ring; it has unique electronic and structural properties, which are much desired in medicinal chemistry. The basicity and protonation and hydrogen bonding capabilities of the heteroatom combined with the presence of a ring nitrogen at the 1-position (pyridine nitrogen) are added with the ability to bind biological targets. This combined aromatic system is also flexible and planetary, which is usually helpful in p-p stacking interactions with aromatic amino acid residues and nucleic acid bases. The quinoline nucleus has a high tolerance to various chemical transformations because of the intrinsic stability of its nucleus, and thus has been significantly diversified.18,19
 |
Figure 1: Structure of Quinoline Click here to View Figure |
The replacement of the quinoline ring system is dominant in the regulation of biological activity and physicochemical characteristics.20,21 The 2 and 4 positions of the pyridine ring are usually functionalized to add either alkyl, aryl or heteroaryl groups, which may affect the electronic distribution and steric profile of the ring. In classical antimalarial agents, especially the 4-position has been also widely utilized, with amino or alkylamino side chains increasing interaction with parasitic targets. Modifications of the benzene ring are commonly employed to optimize lipophilicity, metabolic stability and binding affinity by substituting the 6, 7, and 8 positions.23 Electron-donating groups like methoxy or alkyl moieties can enhance membrane permeability and electron-withdrawing groups like halogen or nitro groups can enhance potency by enhancing target interactions or reducing metabolic degradation.24
The quinoline core is also versatile and can be subjected to the addition of functional handles that can be hybridized like hydroxyl, amino, carboxyl, or halogen groups.25 Such substituents may also be used as anchoring points to linkers that allow quinoline to be transported to other pharmacophores to create hybrid molecules that have better biological properties. In general, quinoline is an extremely versatile scaffold due to its clearly defined substitution patterns and synthetic accessibility. A major, historically significant group of bioactive compounds, especially the field of antiparasitic and antimicrobial drug discovery, are natural products that are based on a quinoline ring as a core structural motif. The most notable include quinoline alkaloids that are plant-based and which have been significant in the history of medicinal chemistry.25,26 The most renowned natural quinoline-bearing compound is quinine, the compound isolated out of the bark of Cinchona species, and it is one of the earliest effective start-up treatments of malaria. Not only has its discovery transformed antimalarial treatment, but it has also formed the quinoline nucleus as a privileged scaffold in the treatment of the parasitic diseases. Quinine Structurally, quinine has a quinoline ring attached to a complex bicyclic amine system, a mixture that forms the basis of its strong biological action and lead to the design of many synthetic quinoline analogues. Besides quinine, a number of similar natural quinoline alkaloids are also derived by the Cinchona bark and have a similar quinoline backbone, including quinidine, cinchonine, and cinchonidine.27,28 The pharmacological characteristics of these compounds are varied; they include antimalarial, antiarrhythmic, and antimicrobial activities, which defines the versatility of the quinoline framework with regard to interactions with a variety of biological targets. Outside of Cinchona, one can find quinoline and quinoline-like natural products found in other families of plants, in marine organisms and in microorganisms, but are less common than other heterocyclic alkaloids. Some Rutaceae plants synthesize simple quinoline alkaloids which can exhibit antimicrobial and cytotoxic effects, indicating that the scaffold has greater ecological and biological functions.
Biological relevance of natural quinoline-containing compounds is directly associated with the ability to participate in p-p interactions, hydrogen bonding, and metal coordination that allows them to disrupt the essential cellular processes of DNA replication, redox balance, and metabolism. The effective example of quinoline alkaloids in nature has had an immense impact on contemporary drug design that has encouraged wholesome synthetic alteration and hybridisation of technology in an effort to improve efficiency and address some of the challenges like toxicity and resistance. In general, natural products containing a quinoline core still remain essential chemical templates, connecting traditional medicine with modern day pharmaceutical innovation.29,30
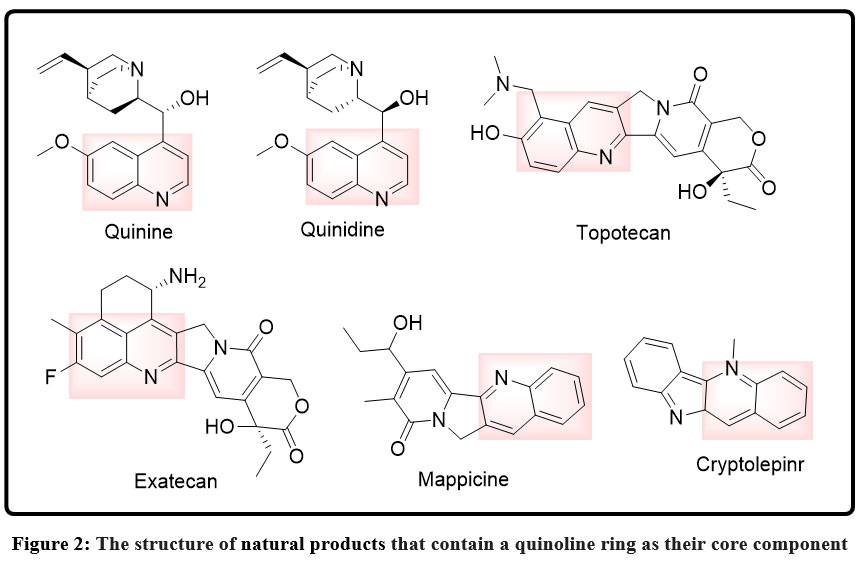 |
Figure 2: The structure of natural products that contain a quinoline ring as their core component Click here to View Figure |
A quinoline ring is a central structural feature of a great number of clinically important and marketed drugs, highlighting the long-term usefulness of this heterocycle in drug development. The quinoline nucleus is normally in the form of a rigid, planar, bicyclic system of a merged benzene and pyridine ring, which is a characteristic that enables a strong p-p stacking, hydrogen bonding, ionic interaction with the biological targets32,33 In a number of antimalarial drugs, the quinoline ring is replaced at key locations to increase accumulation in parasitic compartments, and to become more specific to the target. An example is classical 4-aminoquinoline drugs that have an aminoalkyl side chain at the 4-position making the drug more basic and facilitating localisation in acidic intracellular conditions, a characteristic directly correlated to the therapeutic action of the drug. Other drugs containing quinoline with replacements at the 7- or 8-positions, including halogen or methoxy groups, are marketed, and regulate lipophilicity, stability in the metabolism, and drug activity.34,35
In addition to the antimalarials, quinoline rings also occur in antibacterial, antifungal, and antiarrhythmic drugs. The quinoline core is frequently found to be a pharmacophoric core in these agents, to which other functional groups needed to interact with a particular target are attached. Heteroatoms, unsaturated bonds, or bulky substituents that are attached to the quinoline scaffold can be used to fine-tune the binding affinity and enzyme, nucleic acid, or membrane-associated target selectivity. In other instances, the quinoline ring has been incorporated into more complicated polycyclic or hybrid structures, as indicating its ability to tolerate a wide variety of chemical environments and mechanisms of action.
Structurally, the quinoline containing drugs that are marketed have shown how minor alterations of the central frame can produce highly divergent pharmacological properties. This versatility underlies the reason why the quinoline ring remains part of the contemporary drug discovery initiatives. The clinical experience of the drugs based on quinoline gives a solid rationale to the continued attempts to develop new quinoline analogs and hybrids with better efficacy, safety, and resistance profiles, especially in relation to infectious and parasitic diseases.
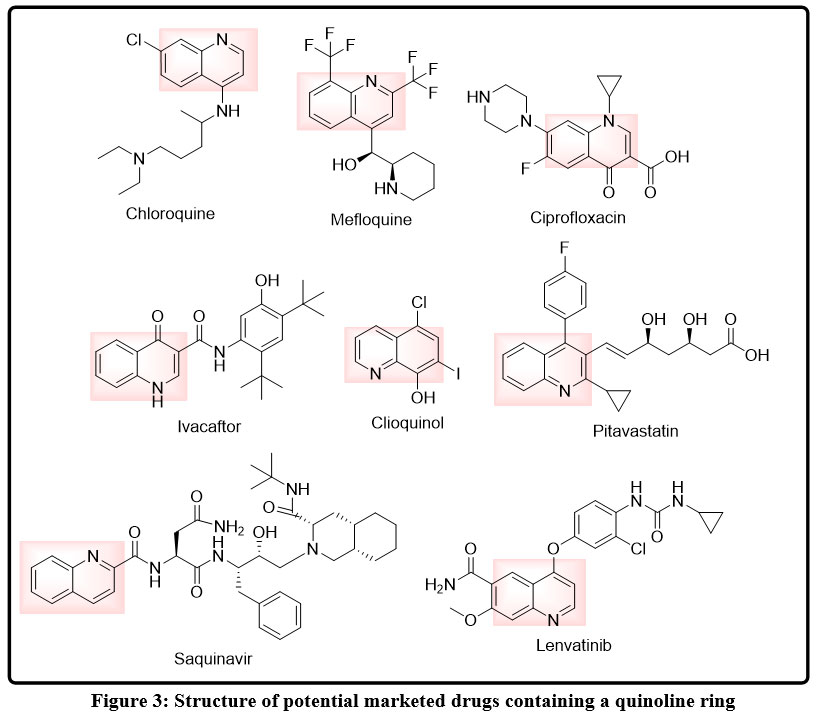 |
Figure 3: Structure of potential marketed drugs containing a quinoline ring Click here to View Figure |
Pharmacophore features relevant to antiparasitic activity
The antiparasitic properties of quinoline derivatives are strictly dependent on certain pharmacophore properties of the scaffold.36,37 The basic nitrogen atom is one of the most important properties and it can be protonated under physiological or acidic intracellular conditions. This property is used in parasitic infections like malaria where quinoline compounds are accumulated in acidic compartments of the parasite and therefore it increases the concentration of drugs at the site of action. Quinole ring is aromatic and heteroaromatic which further facilitates high interactions with the biological macromolecules via p-p stacking and hydrophobic contact. The quinoline based compounds have been known to disrupt various crucial parasitic processes. One of the eminent processes includes impairment of heme detoxification pathways,38,39 especially in the blood-stage parasites. The planar quinoline nucleus has the potential to bind heme moieties, which do not allow them to polymerize to non-toxic forms and cause oxidative stress and death of parasites. Also, parasitic DNA, topoisomerases, and mitochondrial enzymes were reported to be interacting with quinoline derivatives, which suggest their possible use as an agent with several targets. The presence of substituents on the quinoline core has a major impact on these pharmacophoric interactions. Side chains that contain secondary or tertiary amines have the ability of increasing the binding due to their ionic interactions and hydrophobic groups facilitate penetration through the membrane and intracellular target availability. The second pharmacophore may also confer complementary functionality upon the incorporation of quinoline into hybrid molecules, e.g. enzyme inhibition or redox modulation; thus, delivering synergistic antiparasitic activity. It is this activity of quinoline with the addition of other functionalities by hybrid partners that drives the increasing attention to quinoline-linked hybrids as effective antiparasitic agents.40,41
ADME/PK considerations for quinoline derivatives
Absorption, distribution, metabolism and excretion (ADME) properties are very important determinants of clinical success of quinoline-based compounds as far as drug development is concerned. The derivatives of quinolines are associated with a positive oral bioavailability because of their aromatic character and moderate lipophilicity, which facilitates passive diffusion through biological membranes. A fundamental nitrogen can also increase solubility during the protonated form and allow absorption in the gastrointestinal tract. The lipophilicity and ionization state of quinoline compounds determine their distribution properties. A number of quinoline analogs have been shown to exhibit a wide tissue distribution and this can be useful to target intracellular parasites that may be located in the liver and the spleen. Nonetheless, tissue accumulation can be also high, so one should be concerned about off-target effects and toxicity, and optimization must be conducted carefully. Another factor that is important is plasma protein binding, which in excess can decrease the amount of free drug that is available to do the pharmacological action.42,43 Quinoline derivatives are mainly metabolized by hepatic enzymes, such as cytochrome P450 isoforms. The most frequent metabolic pathways are oxidation, dealkylation, conjugation reactions; they may result in detoxification or, in certain instances, reactive reactions. Metabolic stability and half-life can be improved by strategic substitution of the quinoline ring, e.g. addition of sterically hindered groups, or metabolically stable substituents. In hybrid molecules, linker design and partner pharmacophore are other aspects that have a considerable influence on metabolic behavior, either protecting vulnerable sites or generating new metabolic liabilities. Quinoline compounds are excreted through renal or biliary pathways, which are usually based on molecular weight, polarity, and conjugation. Lipophilicity and polarity should therefore be balanced in order to attain efficient clearance without reducing efficacy. Notably, ADME and pharmacokinetic (PK) optimization has to be factored into the initial design of quinoline-linked hybrids to allow the optimization of increased antiparasitic activity to meet tolerable in vivo behavior.44
Design strategies for quinoline-linked hybrids
Types of partner pharmacophores
The quinoline-linked hybrid molecule design is mostly guided by the wise choice of an appropriate partner pharmacophore that can potentially either complement or enhance the inherent antiparasitic effect of the quinoline backbone.45,46 The chemical diversity needed to counteract the complex parasitic targets has been covered through the discovery of a wide variety of pharmacophores. The simplest partners include aryl and substituted aryl groups, which tend to be introduced to control lipophilicity, enhance membrane permeability and enhance hydrophobic interactions with biological targets. These moieties may also result in p-p stacking interactions that are specifically important in DNA- or heme-mediated mechanisms of action. Imidazoles, benzimidazoles, pyridines, thiazoles and oxadiazoles are common heterocyclic pharmacophores that are conjugated with quinoline because they have proven bioactivity against parasitic enzymes and receptors. The heterocycles may tend to add more hydrogen bond acceptors or donors, which increases binding specificity, and potency. The other significant group of partner pharmacophores is chalcones, with its a,b-unsaturated carbonyl being prized due to the ability to react with nucleophilic amino acids of parasitic proteins and interfere with redox balance. The hybrids of quinoline and chalcone have demonstrated a significant antiparasitic effect caused by the synergistic action of the heme binding and the oxidative pressure47,48 The triazoles, especially the 1, 2, 3 triazole are also very popular in the hybrid design due to their stability, rigidity and high capacity of hydrogen-bonding. They usually serve as linkers and pharmacophores, increasing the binding affinity without affecting the pharmacokinetic desirability. Peptide and peptidomimetic counterparts are considered to enhance target selectivity and take advantage of certain transporter systems in parasites, but problems of stability and bioavailability persist. Metal complexes that incorporate quinoline ligands have also been studied more recently as a result of their distinct action modes, such as redox cycling and enzyme inhibition, which provide alternative approaches to drug resistance overcoming.49
Linker strategies
The type of connection between the quinoline core and its partner pharmacophore is a decisive factor of the biological activity of hybrid molecules. Formation of rigid molecules with defined conformations can be achieved by direct fusion, in which the nucleus of quinoline is covalently fused to another aromatic or heterocyclic system without a spacer in between. This rigidity can potentially increase target specificity and binding strength, but also decreases flexibility that are needed to interact with multiple targets. Spacers, usually alkyl and aryl, are frequently used to achieve the best spatial distance between the pharmacophores, in which each moiety is able to independently react with its binding site50,51 The importance of these spacers should be understood to be length, flexibility and polarity which should be optimized because very long or flexible spacers may lower the efficiency of binding and metabolic susceptibility. On the other hand, short and stiff spacers can limit conformational flexibility but increase stability. Improved metabolic stability and preservation or enhancement of biological activity is often achieved by bio-isosteric linkers, including amide, urea, carbamate, or triazole units. These linkers are capable of replicating functional groups and they become less vulnerable to enzyme degradation. A more sophisticated design strategy is cleavable linkers, especially when using prodrug delivery or targeted delivery.52-56 These types of linkers are variously configured to be enzymatically/chemically cleaved in the parasite or in particular tissues, to release the active quinoline moiety and its partner pharmacophore in the site of action. The method is capable of enhancing selectivity and minimizing systemic toxicity, but linker stability must be tightly regulated.
Rational design approaches
Quinoline-linked hybrids are often rationally designed by either a target-based or a phenotypic screening strategy with its own benefits. Design Target-based design is based on being able to know all about one particular molecular target, e.g. a parasitic enzyme or a receptor. Here, quinoline and its partner pharmacophore are identified and optimised to react with specific binding sites, typically based on structural biology information. The strategy allows the examination of structure-activity relationship (SAR) accurately and supports potency and selectivity optimization. It is however overly reliant on correct target validation and can fail to consider off-target or multitarget effects which can promote overall efficacy. Conversely, phenotypic screening does not require prior knowledge of the molecular target and places compounds into a defined set based on their capability to inhibit the growth or survival of the whole-cell assays.57 This method is especially useful in the discovery of antiparasitic drugs, where complicated and overlapping biological pathways may complicate the performance of single-target therapies. Phenotypic screening can be used to discover quinoline-linked hybrids which can potentially act on multiple targets at once minimizing chances of resistance development. Although phenotypic approaches may be more time-consuming regarding the elucidation of the mechanisms involved, the compounds obtained may be more translational.
Computational design and docking-guided hybridization
Calculation programs have found their way in the experimental design of quinoline-linked hybrids, which offer complementary information to the experimental methods. Due to molecular modeling and docking studies, the binding modes, the key interactions between the hybrid molecules and the parasitic targets can be predicted and the binding affinities of the hybrids can be estimated. Docking-directed hybridization is used to select pharmacophore combinations and linkers length rationally to maximize the number of complementary interactions in the target binding pocket.58 Besides docking, quantitative structure-activity relationship (QSAR) models, and molecular dynamics are also being used to assess the flexibility, stability and dynamics of interaction of quinoline hybrids. These methods are used to predict ADME and toxicity profile to prematurely remove poor candidates. Virtual screening of compound libraries can also be used to speed up the process of identifying leads by prioritizing hybrids that have an attractive predicted properties to be synthesized and tested biologically. Generally, strategic pharmacophore selection, optimized linker design, rational experimental strategy, and computational modeling have been integrated into a powerful framework to develop the quinoline-linked hybrids. This complex design approach increases the chances of discovering potent selective and drug-like antiparasitic drug agents that can help solve the current therapeutic needs.
Synthetic methodologies
Classical synthetic routes to quinoline cores
The formation of the quinoline nucleus has been the key to heterocyclic chemistry throughout the last hundred years, and some of the classical reactions are still of great importance in medicinal chemistry of the modern era.
The Skraup synthesis is one of the oldest and most popular ones. It entails condensing an aniline to glycerol in the presence of a strong acid and an oxidizer agent, which results in the production of the quinoline ring system. Although it has several advantages in the strength of reaction and the capacity to produce either unsubstituted nor merely substituted quinolines, the Skraup reaction has quite59,60a number of weaknesses in the form of exhausting reaction conditions, side reactions, and lack of tolerance of some functional groups, which may limit its use in the complex hybrid synthesis.
Another basic method is the Friedlander synthesis, especially appreciated by its versatility. The process entails condensing an o-aminobenzaldehyde or an o-aminoketone with a carbonyl molecule that has an active methylene group. The Friedlander reaction is particularly convenient with respect to synthesizing substituted quinolines with conditions which are relatively milder than Skraup synthesis, and more flexible on the substitution patterns at 2- and 4-positions. Its adaptability has seen it become a favorite pathway to quinoline intermediates that can subsequently be functionalized or conjugated to other pharmacophores.61,62
This reaction of Doebner-Miller is more similar to the Skraup synthesis except that it uses a,b-unsaturated carbonyl compounds in place of glycerol. This will allow the insertion of substituents at desired sites at the quinoline ring, especially at the 2-position. Despite its acidic and at times harsh reaction conditions, it has been modified to make its use more practical by the use of alternative catalysts and solvents. Other classical techniques, the ConradLimpach reaction and Pfitzinger reaction, are also part of the synthetic toolbox, allowing the access to 4-hydroxyquinolines or quinoline carboxylic acids, which can be further derivatized and hybridized.
The Povarov reaction is a potent multicomponent reaction, which allows tetrahydroquinoline scaffolds to be synthesized in high efficiency using readily accessible starting materials, which are usually an aniline, an aldehyde and an electron-rich alkene in the presence of acid. Mechanistically, the reaction is conducted by formation of the initial imine and then aza-Diels-Alder cycloaddition which enables the rapid formation of molecular complexity in a single step. Because of its high atom economy, structural diversity, and ability to react with a broad array of substituents, the Povarov reaction has widespread use in medicinal63-64 chemistry in the constitution of biologically active quinoline-based compounds, such as anticancer and antiparasitic agents.
The Combes quinoline synthesis is a classical procedure to make quinoline derivatives by condensing anilines with acid catalysts with b-diketones. This reaction normally occurs at a strong acidic condition causing cyclization and subsequent dehydration to produce quinoline ring. It is an effective procedure in preparing 4-substituted quinolines. The reaction can be diversified in terms of substituents on the aniline ring and is tolerant to diversity. However, functional group compatibility can be restrained by harsh reaction conditions although it is effective. However, the Combes synthesis is still a significant pathway in heterocyclic chemistry and medicinal chemistry studies.65
Methods for constructing the hybrid linkage
The next step after synthesis of quinoline core is the construction of the hybrid linkage which binds quinoline with its partner pharmacophore. One of the most effective and efficient ways of doing this has been found in click chemistry, and especially in the copper(I)-catalyzed azide-alkyne cycloaddition. The reaction is very yielding, highly regioselective and tolerant to a broad variety of functional groups, therefore, it is an ideal reaction in the assembly of quinoline-triazole hybrids. The resultant triazole ring tends to add other pharmacological properties, including increased metabolic stability and hydrogen-bonding properties.66
SuzukiMiyaura, Sonogashira, Heck and Buchwald-Hartwig couplings are widely used to cross-couple quinoline derivatives with aryl or heteroaryl partners. These catalytic techniques with palladium enable the specific regulation of the formation of carbon-carbon or carbon-heteroatom bonds and are especially useful in the formation of structurally diverse hybrid collections. Amidation is another popular approach, in particular, when quinoline carboxylic acids or amines are conjugated with peptide-like or heterocyclic partners. The amide bonds are chemically stable and predictable in geometry but could be enzymatically degraded in the body.67
Introduction of flexible linkers with secondary or tertiary amines that may enhance solubility, and biological activity are typically achieved by reductive amination. Michael addition reactions are also used to generate quinoline moieties in electrophilic partners, which are a,b-unsaturated carbonyls, by nucleophilic addition of quinoline. All these approaches have their own benefits, and the strategy of the linkage is usually determined by the targeted physicochemical and biological characteristics of the end hybrid molecule.
One-pot and tandem strategies for hybrid synthesis
One-pot and tandem reaction approaches have become more and more popular in synthesis of quinoline-linked hybrids to enhance synthetic efficiency, decrease time and resource use. In one-pot reactions, several steps that form bonds are done in a single reaction vessel without the intermediates being isolated. The method will reduce the number of purification cycles and minimize the amount of solvent used and can usually enhance the yield. An example of this is the formation of quinoline and then functionalization or conjugation with a partner pharmacophore achieved sometimes in a sequence of one68operation. Tandem or cascade reactions extend this principle by enabling the enforcement of many transformations in the same reaction conditions in a programmed series of chemical reactions. These strategies are especially appealing to building complicated hybrid molecules with a high structural density. Tandem reactions can be used in the context of quinoline hybrids in which the quinoline core is first produced by cyclization and the hybrid linkage is installed in situ by some form of functional group transformation. These methods are highly optimized to prevent side reactions, however, they have tremendous benefits as far as atom economy, scalability and sustainability are concerned.69
Green and sustainable approaches
Green and sustainable synthetic methodologies have become increasingly significant in the synthesis of quinoline-linked hybrids to address increasing environmental and economic issues. One of such techniques is the microwave-assisted synth which has shown outstanding advantages such as short reaction time, high yield and selectivity. Formation of rings This is a reaction type in which the formation of quinoline rings can be accelerated by microwave irradiation, usually in solvent-free or low-solvent conditions. This is a reaction type, in which the formation of quinoline rings can be accelerated by microwave irradiation, typically under solvent-free or low-solvent conditions. Another potentially viable green substitute is flow chemistry, which provides accurate adjustment of reaction conditions (temperature, pressure, residence time). Continuous-flow systems are safer, reproducible, and scalable, so these systems are appealing to both laboratory scale synthesis and industrial use. The reduction of solvents and the utilization of clean solvents, i.e. water, ethanol or ionic liquids, also leads to sustainable synthesis. Also, the application of reusable catalysts and the use of non-toxic reagents are consistent with the principles of green chemistry and are becoming increasingly popular in the contemporary quinoline chemistry.70
Practical tips and common challenges
Although a wide range of synthetic approaches is available, there are a number of practical difficulties with the preparation of quinoline-linked hybrids. Regioselectivity in quinoline ring formation and functionalization is a widespread issue especially when there are several active sites. Whether it is the pattern of substitution that is desired, careful selection of starting materials, catalysts, and reaction conditions is necessary. Protecting group strategies are commonly needed to help protect delicate functional groups when synthesizing in multiple steps but overprotecting may lead to complexity in synthesis and lower the overall efficiency.
Further increase of laboratory protocols to gram or kilogram scales also presents new challenges such as heat, reaction reproducibility, and efficiency of purification. Other reactions that are good in small-scale can either show lower yields or be unsafe at large-scale. Hence, scalability and robustness should be taken into consideration early, particularly in the case of hybrids that are to be used in sophisticated biological examination. Consideration of these issues during syntheses and optimization of these methods is the only solution to enable the efficient and reproducible production of quinoline-linked hybrids that will further facilitate their future use as promising antiparasitic agents.
In vitro antiparasitic evaluation
In vitro antiparasitic testing is an essential step in preclinical assessment of the quinoline-linked hybrids, which gives mechanistic understanding, comparative strength statistics, and initial indications of selectivity before in vivo research. Such tests are usually compared with a panel of clinically relevant protozoan parasites, which mostly include Plasmodium species causing malaria, Leishmania species causing visceral and cutaneous leishmaniasis, and Trypanosoma species causing African trypanosomiasis and Chagas disease, although some studies also include other parasitic organisms depending on the scope of treatment. The most commonly used in vitro malaria model, as both drug sensitive and drug resistant, is plasmodium falciparum, as it is seen to be clinically relevant worldwide with well known culture systems. In the case of leishmaniasis, promastigote and intracellular amastigote forms of Leishmania donovani or Leishmania major are usually used, since these stages represent different features of parasite biology and drug sensitivity. With trypanosomiasis, bloodstream forms of Trypanosoma brucei or intracellular forms of Trypanosoma cruzi are often used to measure antiparasitic activity. The organism and stage of life-cycle used is also a significant consideration, such that quinoline-linked hybrids can exhibit a stage-dependent activity associated with their mechanism of action and accumulation behavior within cells.
To assess the antiparasitic activity of quinoline hybrids, many different types of assays are used, the most common of which is cell-based viability assays because they are physiologically relevant. The assays are a measurement of parasite growth inhibition or viability loss after the test compounds have been added, often through a colorimetric, fluorometric or luminescent readout in terms of metabolic activity, DNA content or reporter gene expression. These assays are integrated to measure compound permeability, intracellular stability, and target engagement. Simultaneously, enzyme-based assays are frequently used to investigate certain molecular processes, especially when quinoline hybrids are created to disrupt certain pathways of parasites. To illustrate, assays measuring heme binding or inhibition of hemozoins formation have been extensively employed in malaria studies, which is indicative of the well-known action of quinoline derivatives in interfering with heme detoxification. Equally, topoisomerase inhibition assay, redox enzyme assay, and mitochondrial enzyme activity are used to clarify the possible targets in Leishmania and Trypanosoma. More translational-relevant assays, like liver-stage malaria models or intracellular amastigote assays in macrophages, provide additional stage-specificity to the assay, and further approximate the biological condition of infection.
Quantitative analysis of in vitro data is based on standardized measures that provide the ability to compare between compounds and experiment. The most reported parameter is the half-maximal inhibitory concentration or IC50, which is the concentration needed to reduce parasite growth by 50 per cent under specific conditions. In other instances, the half-maximal effective concentration (EC50) has been indicated especially when functional readouts other than growth inhibition are used. Such measures of potency are commonly supplemented with analyses of cytotoxicity against mammalian cell lines, and the selectivity index (SI), the ratio of host cell toxicity to antiparasitic potency can be determined. A high index of selectivity is usually regarded as evidence of a good therapeutic window and is one of the criteria of prioritizing quinoline-linked hybrids to develop. It is necessary that consistent reporting be done of assay conditions, incubation times, and parasite strains in order to be able to replicate and meaningfully compare IC50, EC50, and SI values across studies.
The validity of antiparasitic case studies in vitro is dependent on appropriate experimental controls and reference drugs. Common positive controls in Plasmodium tests are standard antimalarial drugs like chloroquine, quinine or artemisinin derivatives and common references in leishmanial and trypanosomal systems are amphotericin B, miltefosine or pentamidine. Negative controls which are usually used to determine baseline growth and viability entail the use of vehicle-treated parasites, but the untreated controls are used to confirm assay integrity. When these controls are included, not only is the performance of the assays validated, but they also can be used to benchmark quinoline-linked hybrids against an established therapy in the clinic. These comparisons prove especially useful to determine activity with drug-resistant strains where hybrids might show retained or increased potency in comparison with standard drugs.
In order to enable a systematic analysis, numerous studies have summarized representative quinoline hybrids and their in vitro antiparasitic performances to comparative tables by parasite species. Such tables generally give an overview of such essential structural characteristics, assay models, values of potency and selectivity indices, and typical reference compounds in a concise manner. Collected by type of parasites, these compilations can allow one to quickly find pharmacophore combinations of linker strategies of greater activity essence. Together, in vitro antiparasitic testing of quinoline-conjugated hybrids can be a strong and instructive platform to inform medicinal chemistry optimization, understand the action of the hybrids, and to select promising hybrids to be used in further in vivo and translational testing.
In vivo studies and preclinical evaluation
In vivo studies form a final step in the preclinical development of quinoline-linked hybrids: they give a unified evaluation of the efficacy, pharmacokinetics and safety in a living biological environment that cannot be completely modeled in in vitro models. The animal models used in the evaluation of antiparasitics are usually chosen to be as similar as possible to the pathology of a disease and biology of a parasite in the human body. The rodent model is also commonly employed in malaria research, in which Plasmodium berghei, Plasmodium yoelii or Plasmodium chabaudi infection of mice are used to evaluate the antimalarial activity of a compound in its blood-stage, suppressive features and survival. The models are used to rapidly screen a set of candidate compounds, and dose-response can be evaluated in controlled conditions. In the case of leishmaniasis, both visceral and cutaneous models are also in wide use, which usually consists of infecting mice or hamsters with Leishmania donovani, Leishmania infantum, or Leishmania major. Visceral models determine the parasite burden of target organs (liver and spleen) and cutaneous models determine the extent of lesions and parasite clearance at the site of infection. Murine models of trypanosomiasis typically involve the ideas of Trypanosoma brucei or Trypanosoma cruzi in which the parasitemic parasite tissues and lifespan of the parasites can be used as the main measure of the therapeutic effects of treatments. These animal models can give significant understanding of the in vivo performance of quinoline hybrids, such as to reach their target tissues, survive at therapeutic levels and have delayed antiparasitic impacts.
In vivo evaluation always involves pharmacokinetic and bioavailability tests since they will provide information on whether the promising activity of quinoline-linked hybrids in vitro can be transferred into effective systemic exposure. Pharmacokinetic experiments are usually associated with a time-dependent study of plasma and tissue concentrations after various routes of drug administration as oral, intraperitoneal, or intravenous. Such parameters as maximum concentration, time to reach maximum concentration, area under the concentration time curve, half lipid, clearance and volume of distribution are used to have a quantitative framework of drug disposition. Quinoline derivatives tend to have a good oral bioavailability as a result of their aromatic and moderately lipophilic character, but hybridization can have a far-reaching impact on the absorption and distribution pattern. The distribution of the tissue is of special interest in the development of antiparasitic drugs since the required effective concentrations should be reached in the areas of parasite reservation, including liver, spleen, bone marrow, or central nervous system. Pharmacokinetic data can often inform selection of linker length, partner pharmacophore polarity, and partner drug clearance to achieve exposures adequate and clearance rates acceptable.
The toxicological assessment and safety signals should be identified as a measure to guarantee that the increased antiparasitic efficacy is not linked to the toxicity of the host. Rodent studies of acute and subacute toxicity are usually carried out in order to determine the highest tolerated dose and monitor possible negative effects that could manifest on behavior, body mass, and organ, especially liver and kidney, performance. As an indicator of organ-specific toxicity, hepatic and renal biochemical markers and histopathological assessment of the key organs are regularly evaluated. Quinoline-based compounds have traditionally been linked with some safety issues, including cardiotoxicity or neurotoxicity at high doses and are therefore of special safety-profiling interest to quinoline-linked hybrids. The hybrides could alleviate or add to these risks, depending on the type of the partner pharmacophore and linker, and this offers a strong case to carry out the thorough in vivo safety testing at a very early stage of development.
The choice of efficacy endpoints in in vivo antiparasitic trials is based on the therapeutic benefit and translatability. The typical outcomes are a decrease in parasitemia, a decrease in the parasite burden in the tissue in a model of cutaneous leishmaniasis, regression of the lesion, extension of the survival, and inhibition of the relapse of the disease. Dose-response and duration of study of the treatment give a clue on the best dosing schedule and therapeutic index. Other aspects of translational considerations are; efficacy comparison with known reference drugs in the same conditions, assessing activity in relation to drug-resistant isolates and potential combination with other available treatments. These in vivo and preclinical analyses, in combination, are a crucial linkage between in vitro discovery and clinical development to make informed decisions about the development of quinoline-linked hybrids as functional antiparasitic drug development.
Mechanisms of antiparasitic action
The antiparasitic effect of quinoline-conjugated hybrids is due to an interaction between long-established quinoline-mediated effects and synergies between the partner pharmacophores, which results in multifaceted and frequently synergistic mechanisms of action. The quinoline scaffold is linked to one of the best-characterized pathways: heme detoxification inhibition, in especially case of parasites in the blood-stage of malaria. In the process of hemoglobin digestion, toxic free heme is expelled in the digestive vacuole of the parasite and has to be detoxified by polymerization into untoxic hemozoin. The quinoline derivatives, due to their structural features, planar aromatic backbone, and simple nitrogen, are able to accumulate in this acidic compartment, and bind to the free heme, thus preventing its polymerization. This leads to accumulation of toxic heme-drug complexes leading to oxidative damage, disruption of the membrane and eventual death of the parasites. In addition to the effects involving the heme, quinoline-based compounds have also been demonstrated to interact with nucleic acids, and these may either intercalate DNA or disrupt nucleic acid-processing enzymes, including topoisomerases. Such interactions are capable of disabling DNA replication and transcription resulting in cell cycle arrest and apoptosis-like death in parasites. Also, mitochondrial dysfunction is another significant quinoline-related process, especially in protozoa, where the energy metabolism and redox depends on mitochondrial integrity. Quinoline derivatives can disrupt mitochondrial membrane potential, prevent electron transport or disrupt enzymes of vital metabolic pathways, which affect the viability of parasites. Oxidative stress is also an addition towards antiparasitic effect, quinoline-induced redox imbalance may overwhelm the antioxidant defence of the parasite, resulting in lipid peroxidation, protein oxidation and cell damage.
In case of incorporation of quinoline with other molecules, the partner-pharmacophore usually incorporate other mechanistic layers that complement or increase antiparasitic action. They can be synergistic, with both pharmacophores simultaneously influencing related pathways to achieve greater overall effect or they can be complementary in that different targets are simultaneously altered. An example is to include quinoline with heterocyclic groups which are reported to inhibit particular parasitic enzymes, thus heme detoxification blockade can be used with precise enzyme inhibition, and chances of resistance emergence are minimized. Partners with chalcone group have the ability to provide electrophilic centers that are able to modify thiol functional groups, disrupting redox homeostasis and increasing oxidative stress generated by the quinoline group. Hybrids that contain triazole are able to disrupt sterol biosynthesis or protein-ligand interactions, as well as to provide metabolically stable linkers that reposition optimally the spatial orientation of the pharmacophores. In hybrids containing metal complexes, the partner moiety can provide redox cycling or metal-mediated inhibition of enzyme, and provide an additional cytotoxic action that acts in a parallel manner to classical quinoline effects. More complicated, but additional partners, such as peptide or peptidomimetic, can provide increased selectivity, based on developmental program transporters or intracellular compartments, and efficiently targeting the quinoline core to where it is required to act.
The arguments in favor of these mechanistic suggestions are based on a synthesis of biochemical tests, omics-ready research, and state-of-the-art imaging. Biochemical assays are still essential mechanisms of mechanistic validation, such as heme binding assays, parasitic enzyme inhibition assays, mitochondrial membrane potential measurements, and reactive oxygen species production assays. These assays offer direct functional evidence of chemical structure/biological effect. Transcriptomic and proteomic investigations have been used more and more to understand the global cellular responses to quinoline-linked hybrids with changes in gene and protein expression related to stress response mechanisms, metabolic perturbation, and programmed cell death. These types of omics are useful especially in determining off-target effects and in the discovery of hitherto unknown pathways that lead to antiparasitic activities. Biochemical data is further supported by imaging methods, such as fluorescence microscopy and electron microscopy, which can be used to visualize cellular localization of hybrid compounds, cellular morphology of parasites, and organelle damage, such as digestive vacuole or mitochondrial damage. All these multidisciplinary pieces of evidence reinforce the idea that quinoline-linked hybrids do not use a single mechanism of action since they are antiparasitic in a well-coordinated, multi-target interaction. This is one of the major advantages of the hybrid molecules because this increase in potency along with minimizing resistance and further development is practical with regard to the current generation of hybrid molecules as antiparasitic drug discovery candidates.
Future perspectives and recommendations
The strategic combination of new design ideas, new biology tests, and translational prospectus will give rise to future innovation of quinoline hybrids as antiparasitic agents in solving current and future problems in treating parasitic diseases. The rational hybridization strategy, continuing to be explored, is likely to give compounds with higher potency, greater selectivity and less resistance liability, especially by purposeful assembly of quinoline scaffolds with pharmacophores to complementary or orthogonal biological pathways. More attention should be paid to the development of hybrids with balanced physicochemical characteristics, since not only should an increase in the antiparasitic activity but also a positive pharmacocytokinetic and safety profile be evaluated. The importance of early integration of ADME and toxicity screening with predictive in silico models can greatly decrease late-stage attrition and also facilitate optimization of lead. Biologically, further research should feature use of clinically relevant parasite strains more frequently, such as multidrug resistant isolates and physiologically relevant systems such as intracellular and stage specific assays to improve translational relevance. The lack of standardization of in vitro and in vivo assessment regimens, such as provision of selectivity indices, dosing schedules and comparative effectiveness with reference drugs will provide a cross-study comparison and hasten overall advancement in the field. The computational chemistry, artificial intelligence, and machine learning provide strong opportunities to inform the selection of the pharmacophore, optimization of linkers, and target prioritization to navigate the chemical space better and find high-quality lead inferences. In addition, systems-level technologies, including transcriptomics, proteomics, and metabolomics, deserve increased adoption in order to understand the pathobiology of action, identify resistance mechanisms, and guide the combination strategy. Synthetically, there will be the need to embrace green and scalable approaches in order to facilitate sustainable drug development and subsequent translation to industry. Lastly, there is a high degree of communication between medicinal chemists, parasitologists, pharmacologists and clinicians, which is highly encouraged to correlate the early-stage discovery activities with clinical therapeutic imperatives. Through the adoption of interdisciplinary approaches and emphasis on scientific rigor as well as practical usefulness, quinoline-linked hybrids can help create a meaningful contribution to the future generation of safe, effective, and accessible antiparasitic therapy.
Conclusion
Quinoline-linked hybrids are a very promising and strategically important category of compounds in the continued quest to find useful antiparasitic therapy using quinoline-linker, which integrates the pharmacological significance of the quinoline scaffold with the versatility and synergic potential of molecular hybridization. Quinoline has exceptionally wide chemical flexibility, which permits accurate structural manipulation and innate incorporation with various partner pharmacophores to permit the rational design of multifunctional molecules capable of meeting the complex and adaptive biology of parasitic organisms. As indicated in the review, the development of synthetic approaches has hugely increased the availability of structurally diverse quinoline hybrids, and the techniques of characterization used today has provided the means of ensuring the structural validity and reliability. In vitro and in vivo analyses have consistently shown that quinoline conjugated hybrids can be used to attain enhanced antiparasitic activity, selectivity, and in most instances still maintain activity against drug-resistant strains of parasites. Notably, the complex actions of these hybrids (inhibition of heme detoxification and interference with mitochondrial activity as well as redox balance and enzyme inhibition) are indicative of their advantage compared to single-acting agents and they are in favor of their ability to counteract the emergence of resistance. Complementary pharmacophore incorporation also enhances therapeutic effect by acting synergistically or by acting on multiple targets and has provided a logical direction towards longer-lasting and more effective therapies. Meanwhile, there are still challenges concerning pharmacokinetics, toxicity, scalability and predictability in translational studies, and integration of ADME optimization, safety evaluation, and clinically relevant models further up the drug development pipeline is needed. All the evidence provided above makes quinoline-linked hybrids a resilient and versatile discovery platform in antiparasitic drugs. Through ongoing interdisciplinary cooperation, systematic assessment strategies, and careful combination of computational and experimental methods, these hybrid molecules can be highly promising to support unmet clinical requirements in parasitic infections and even move towards the development of safe, effective and readily available therapeutic options.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
References
- Abdelbaset, M. S., Abdel-Aziz, M., Abuo-Rahma, G. E. D. A., Abdelrahman, M. H., Ramadan, M., & Youssif, B. G. M. (2018). Novel quinoline derivatives carrying nitrones/oximes nitric oxide donors: Design, synthesis, antiproliferative and caspase-3 activation activities. Archiv der Pharmazie, *352*(1), 1800270.
CrossRef - Ajani, O. O., Iyaye, K. T., Audu, O. Y., Olorunshola, S. J., Kuye, A. O., & Olanrewaju, I. O. (2018). Microwave assisted synthesis and antimicrobial potential of quinoline-based 4-hydrazide-hydrazone derivatives. Journal of Heterocyclic Chemistry, *55*(1), 302–312.
CrossRef - Ajani, O. O., Iyaye, K. T., & Ademosun, O. T. (2022). Recent advances in chemistry and therapeutic potential of functionalized quinoline motifs–a review. RSC Advances, *12*(30), 18594–18614.
CrossRef - Aldhahrani, A. (2025). In Vitro biological evaluation of some hybrid molecules bearing 2-quinoline as anti-inflammatory and anti-cancer agents. Journal of Molecular Structure, *1310*, 138500.
CrossRef - Alizadeh, A., Rezvanian, A., & Zhu, L. G. (2010). Novel heterocyclic β-nitroenamines-based on a one-pot three-component reaction: A facile synthesis of fully substituted 1H-pyrrolo[1,2-a]-fused-1,3-diazaheterocycles. Tetrahedron, *66*(51), 6631–6636.
CrossRef - Almandil, N. B., Taha, M., Rahim, F., Wadood, A., Imran, S., Alqahtani, M. A., Bamarouf, Y. A., Ibrahim, M., & Mosaddik, A. (2019). Synthesis of novel quinoline-based thiadiazole, evaluation of their antileishmanial potential and molecular docking studies. Bioorganic Chemistry, *85*, 109–116.
CrossRef - Angajala, G., Aruna, V., & Subashini, R. (2020). An efficient nano-copper catalyzed base-free Knoevenagel condensation: A facile synthesis, molecular modelling simulations, SAR and hypoglycemic studies of new quinoline tethered acridine analogues as PPARγ agonists. Journal of Molecular Structure, *1220*, 128601.
CrossRef - Costa, C. A., Lopes, R. M., Ferraz, L. S., Esteves, G. N., Di Iorio, J. F., Souza, A. A., Silva, M. M., de Sousa, A. P., Pinheiro, D. P., de Oliveira, R. B., Krettli, A. U., & da Silva, L. E. (2020). Cytotoxicity of 4-substituted quinoline derivatives: Anticancer and antileishmanial potential. Bioorganic & Medicinal Chemistry, *28*(11), 115511.
CrossRef - Desai, N. C., Kotadiya, G. M., Jadeja, K. A., Shah, K. N., Malani, A. H., Manga, V., & Bhuva, V. D. (2021). Synthesis, antitubercular, antimicrobial activities and molecular docking study of quinoline bearing dihydropyrimidines. Bioorganic Chemistry, *115*, 105173.
CrossRef - Desai, N. R., Patel, N. B., & Patel, V. P. (2019). Synthesis, crystal structure, and molecular docking studies of novel 2-(4-benzoylpiperazin-1-yl) quinoline-3-carbaldehyde. Chemical Data Collections, *22*, 100251.
CrossRef - Diaconu, D., Amariucai-Mantu, D., Mangalagiu, V., Antoci, V., Zbancioc, G., & Mangalagiu, I. I. (2021). Ultrasound assisted synthesis of hybrid quinoline-imidazole derivatives: A green synthetic approach. RSC Advances, *11*(60), 38297–38301.
CrossRef - Douadi, K., Chafaa, S., Douadi, T., Al-Noaimi, M., & Kaabi, I. (2020). Azoimine quinoline derivatives: Synthesis, classical and electrochemical evaluation of antioxidant, anti-inflammatory, antimicrobial activities and the DNA/BSA binding. Journal of Molecular Structure, *1217*, 128305.
CrossRef - Eissa, S. I., Farrag, A. M., Abbas, S. Y., El Shehry, M. F., Ragab, A., Fayed, E. A., & Ammar, Y. A. (2021). Novel structural hybrids of quinoline and thiazole moieties: Synthesis and evaluation of antibacterial and antifungal activities with molecular modeling studies. Bioorganic Chemistry, *110*, 104803.
CrossRef - El-Gamal, K. M., El-Morsy, A. M., Saad, A. M., Eissa, I. H., & Alswah, M. (2018). Synthesis, docking, QSAR, ADMET and antimicrobial evaluation of new quinoline-3-carbonitrile derivatives as potential DNA-gyrase inhibitors. Journal of Molecular Structure, *1166*, 15–33.
CrossRef - Fan, Y. L., Cheng, X. W., Wu, J. B., Liu, M., Zhang, F. Z., Xu, Z., & Feng, L. S. (2018). Anti-plasmodial and antimalarial activities of quinolone derivatives: An overview. European Journal of Medicinal Chemistry, *146*, 1–14.
CrossRef - Ferreira, L. M., de Sá, E. M. F., Pinto, M. E. F., & Kümmerle, A. E. (2025). A review on quinolines: New green synthetic methods and bioactive potential. European Journal of Pharmaceutical Sciences, *191*, 106589.
CrossRef - Ganesan, M. S., Raja, K. K., Murugesan, S., Kumar, B. K., Rajagopal, G., & Thirunavukkarasu, S. (2020). Synthesis, biological evaluation, molecular docking, molecular dynamics and DFT studies of quinoline-fluoroproline amide hybrids. Journal of Molecular Structure, *1217*, 128360.
CrossRef - Garudachari, B., Isloor, A. M., Satyanarayana, M. N., Fun, H. K., & Hegde, G. (2012). Synthesis, characterization and anti-microbial studies of some new quinoline incorporated benzimidazole derivatives. European Journal of Medicinal Chemistry, *54*, 900–906.
CrossRef - George, R. F., Samir, E. M., Abdelhamed, M. N., Abdel-Aziz, H. A., & Abbas, S. E. S. (2019). Synthesis and anti-proliferative activity of some new quinoline based 4,5-dihydropyrazoles and their thiazole hybrids as EGFR inhibitors. Bioorganic Chemistry, *82*, 212–223.
CrossRef - Ghanim, A. M., Girgis, A. S., Kariuki, B. M., Samir, N., Said, M. F., Abdelnaser, A., Nasr, S., & Bekheit, M. S. (2022). Design and synthesis of ibuprofen-quinoline conjugates as potential anti-inflammatory and analgesic drug candidates. Bioorganic Chemistry, *119*, 105557.
CrossRef - Ghasemi, M., Mohammadi-Khanaposhtani, M., Rezaei, S., Asadi, M., Faramarzi, M. A., Bandarian, F., Hosseini, S., Biglar, M., Larijani, B., & Mahdavi, M. (2025). Quinoline-piperazine derivatives as potential α-glucosidase inhibitors: Synthesis, biological evaluation, and in silico studies. Journal of Molecular Structure, *1315*, 138842.
CrossRef - Ghorab, W. M., El-Gaby, M. S. A., Alsaid, M. S., El-Senduny, F. F., Badria, F. A., & Sherif, M. H. (2023). Design, synthesis, and molecular modeling study of certain quinazolinone derivatives targeting poly (ADP-ribose) polymerase 1 (PARP-1) enzyme as anti-breast cancer and radio-sensitizers. Journal of Molecular Structure, *1281*, 135117.
CrossRef - Govindarao, K., Sreenivasulu, C., Subbaiah, D. R. C. V., Raju, C. N., & Naga Raju, C. (2022). Quinoline conjugated 2-azetidinone derivatives as prospective anti-breast cancer agents: In vitro anti-proliferative and anti-EGFR activities, molecular docking and in-silico drug likeliness studies. Journal of Saudi Chemical Society, *26*(2), 101428.
CrossRef - Hayat, K., Afzal, S., Saeed, A., Murtaza, A., Ur Rahman, S., Khan, K. M., Larik, F. A., Hassan, M., & Raza, H. (2019). Investigation of new quinoline derivatives as promising inhibitors of NTPDases: Synthesis, SAR analysis and molecular docking studies. Bioorganic Chemistry, *87*, 218–226.
CrossRef - Hegazy, M. E., El-Hagrassi, A. M., El-Ansari, M. A., El-Behairy, M. F., & Abdel-Aziz, M. M. (2024). Tailored quinoline hybrids as promising COX-2/15-LOX dual inhibitors endowed with diverse safety profile: Design, synthesis, SAR, and histopathological study. Bioorganic Chemistry, *144*, 107138.
CrossRef - Hofny, H. A., Mohamed, M. F., Gomaa, H. A. M., El-Sayed, A. A., & Youssef, M. M. (2021). Design, synthesis, and anti-bacterial evaluation of new quinoline-1,3,4-oxadiazole and quinoline-1,2,4-triazole hybrids as potential inhibitors of DNA gyrase and topoisomerase IV. Bioorganic Chemistry, *114*, 105137.
CrossRef - Hossain, M. (2018). A review on heterocyclic: Synthesis and their application in medicinal chemistry of imidazole moiety. Science Journal of Chemistry, *6*(5), 83–94.
CrossRef - Hu, Y. Q., Zhang, S., Xu, Z., Lv, Z. S., Liu, M. L., & Feng, L. S. (2017). Quinoline hybrids and their anti-plasmodial and anti-malarial activities. European Journal of Medicinal Chemistry, *139*, 22–47.
CrossRef - Kalita, J., Chetia, D., & Rudrapal, M. (2019). Design, synthesis, antimalarial activity and docking study of 7-chloro-4-(2-(substituted benzylidene)hydrazineyl)quinolines. Journal of Medicinal Chemistry and Drug Design, *2*(1), 928–937.
CrossRef - Kardile, R. A., Khedkar, V. M., Chaturbhuj, G. U., Sarkate, A. P., Lokwani, D. K., & Shinde, D. B. (2021). Design and synthesis of novel conformationally constrained 7,12-dihydrodibenzo[b,h][1,6]naphthyridine and 7H-Chromeno[3,2-c] quinoline derivatives as topoisomerase I inhibitors: In vitro screening, molecular docking and ADME predictions. Bioorganic Chemistry, *115*, 105198.
CrossRef - Karpun, Y., Panchuk, R., Chopyak, V., Hrytsak, M., & Gzella, A. (2021). The investigation of antimicrobial activity of some S-substituted bis1,2,4-triazole-3-thiones. Pharmacia, *68*(3), 673–683.
CrossRef - Kaushik, C. P., Sangwan, J., Luxmi, R., Kumar, D., & Pahwa, A. (2019). One-pot facile synthesis, crystal structure and anti-fungal activity of 1,2,3-triazoles bridged with amine-amide functionalities. Synthetic Communications, *49*(3), 391–405.
CrossRef - Kumar, A., Srivastava, K., Kumar, S. R., Puri, S. K., & Chauhan, P. M. S. (2023). Novel hybrids of quinoline with pyrazolylchalcones as potential anti-malarial agents: Synthesis, biological evaluation, molecular docking, and ADME prediction. Chemico-Biological Interactions, *369*, 110296.
CrossRef - Li, B., Yao, J., He, F., Liu, J., Lin, Z., Liu, S., Huang, M., Zhou, C., Liu, Y., & Luo, H. (2021). Synthesis, SAR study, and bioactivity evaluation of a series of Quinoline-Indole-Schiff base derivatives: Compound 10E as a new Nur77 exporter and autophagic death inducer. Bioorganic Chemistry, *113*, 105008.
CrossRef - Li, S., Hu, L., Li, J., Zhu, J., Zeng, F., Huang, Q., Sun, H., & Liu, Z. (2019). Design, synthesis, Structure-Activity Relationships and mechanism of action of new quinoline derivatives as potential antitumor agents. European Journal of Medicinal Chemistry, *162*, 666–678.
CrossRef - Li, X.-Y., Liu, Y., Chen, X. L., Lu, X. Y., Liang, X. X., Zhu, S. S., Mo, D. L., & Su, G. F. (2020). 6π-Electrocyclization in water: Microwave-assisted synthesis of polyheterocyclic-fused quinoline-2-thiones. Green Chemistry, *22*(14), 4445–4449.
CrossRef - Mahgoub, S. (2018). Synthesis, characterization and antimicrobial activity of some novel quinoline derivatives bearing pyrazole and pyridine moieties. Egyptian Journal of Chemistry, *61*(6), 1035–1049.
CrossRef - Medeiros, G. A., Correa, J. R., de Andrade, L. P., Lopes, T. O., de Oliveira, H. C., Diniz, A. B., de Melo, I. S., Magalhães, K. G., de Oliveira, H. C. B., & da Silva Júnior, E. N. (2021). A benzothiadiazole-quinoline hybrid sensor for specific bioimaging and surgery procedures in mice. Sensors and Actuators B: Chemical, *328*, 128998.
CrossRef - Mohamed, N., & El-Sayed, R. (2024). Synthetic approaches for quinoline heterocycles fused at face b: A review. Synthetic Communications, *54*(1), 1–25.
CrossRef - Mohassab, A. M., Hassan, H. A., Abdel-Aziz, M., Dalby, K. N., & Kaoud, T. S. (2021). Design and synthesis of novel quinoline/chalcone/1,2,4-triazole hybrids as potent anti-proliferative agents targeting EGFR and BRAFV600E kinases. Bioorganic Chemistry, *107*, 104510.
CrossRef - Muálem de Moraes Alves, M., Arcanjo, D. D. R., Carvalho, R. C. V. d., Amorim, L. V., Santos, I. L. d., Santos, R. R. L., da Silva, A. L. M., de Oliveira, A. P., de Moura, R. O., & Barbosa-Filho, J. M. (2021). Methods of macrophages activation and their modulation for the prospection of new antileishmania drugs: A review. Bioscience Journal, *37*, e37077.
CrossRef - Nainwal, L. M., Tasneem, S., Akhtar, W., Verma, G., Khan, M. F., Parvez, S., & Shaquiquzzaman, M. (2019). Green recipes to quinoline: A review. European Journal of Medicinal Chemistry, *164*, 121–170.
CrossRef - Nikookar, H., Mohammadi-Khanaposhtani, M., Imanparast, S., Faramarzi, M. A., Ranjbar, P. R., Mahdavi, M., & Larijani, B. (2018). Design, synthesis and in vitro α-glucosidase inhibition of novel dihydropyrano[3,2-c]quinoline derivatives as potential anti-diabetic agents. Bioorganic Chemistry, *77*, 280–286.
CrossRef - Panchal, N. B., Patel, M. M., & Patel, H. D. (2023). From Molecules to Medicine: The Remarkable Pharmacological Odyssey of Quinoline and It’s Derivatives. Oriental Journal of Chemistry, *39*(4), 808–827.
CrossRef - Porter, A. G., & Jänicke, R. U. (1999). Emerging roles of caspase-3 in apoptosis. Cell Death & Differentiation, *6*(2), 99–104.
CrossRef - Ramprasad, J., Kumar Sthalam, V., Linga Murthy Thampunuri, R., Bhukya, S., Ummanni, R., Balasubramanian, S., & Rao, V. J. (2019). Synthesis and evaluation of a novel quinoline-triazole analogs for antitubercular properties via molecular hybridization approach. Bioorganic & Medicinal Chemistry Letters, *29*(20), 126671.
CrossRef - Rani, A., Sharma, A., Legac, J., Rosenthal, P. J., Singh, P., & Kumar, V. (2021). A trio of quinoline-isoniazid-phthalimide with promising antiplasmodial potential: Synthesis, in-vitro evaluation and heme-polymerization inhibition studies. Bioorganic & Medicinal Chemistry, *39*, 116159.
CrossRef - Rehman, M. U., & Ahmad, B. (2025). Anti-bacterial and anti-fungal pyrazoles based on different construction strategies. European Journal of Medicinal Chemistry, *271*, 116400.
CrossRef - Rezvanian, A. (2016). An expedient synthesis strategy to the 1,4-dihydropyridines and pyrido[1,2-a]quinoxalines: Iodine-catalyzed one-pot four-component domino reactions. Tetrahedron, *72*(46), 7381–7388.
CrossRef - Rezvanian, A. (2021). Synthesis of novel 1,3-cyclohexadiene derivatives bearing 2-oxo-quinoline moiety via a 4-CR strategy. ChemistrySelect, *6*(41), 11376–11384.
CrossRef - Rezvanian, A. (2022). Use of dialkyl acetylenedicarboxylates in the multicomponent synthesis of heterocyclic structures. ChemistrySelect, *7*(15), e202200719.
CrossRef - Rezvanian, A., Alizadeh, A., & Zhu, L. G. (2017). Five-component synthesis of dihydropyridines based on diketene. Tetrahedron, *73*(30), 4474–4480.
CrossRef - Rezvanian, A., & Heravi, M. M. (2018). Metal-free assemblage of four C−N and two C−C bonds via a cascade five component diastereoselective synthesis of pyrido[1,2-a]pyrimidines. ChemistrySelect, *3*(45), 12735–12741.
CrossRef - Rezvanian, A., & Heravi, M. M. (2020). Quinoline conjugated imidazopyridine and pyridopyrimidine synthesis in water as highly selective fluoride sensors via a catalyst-free four-component reaction. Monatshefte für Chemie – Chemical Monthly, *151*(7), 1087–1095.
CrossRef - Rezvanian, A., & Heravi, M. M. (2020). Cascade process for direct synthesis of indeno[1,2-b]furans and indeno[1,2-b]pyrroles from diketene and ninhydrin. Molecular Diversity, *24*(3), 625–634.
CrossRef - Rezvanian, A., & Heravi, M. M. (2021). Sequential four-component protocol for the synthesis of pyrido[1,2-a]pyrimidin-6-one derivatives in water. Chemical Papers, *75*(8), 3921–3931.
CrossRef - Rezvanian, A., & Heravi, M. M. (2024). A versatile approach for one-pot synthesis of hybridized quinolines linked to fused N-containing heterocycles in water. Molecular Diversity.
CrossRef - Roy, D., Anas, M., Manhas, A., Saha, S., Kumar, N., & Panda, G. (2022). Synthesis, biological evaluation, Structure-Activity Relationship studies of quinoline-imidazole derivatives as potent antimalarial agents. Bioorganic Chemistry, *121*, 105671.
CrossRef - Selvakumaran, M., & Prasath, M. (2023). Novel Benzimidazole linked piperidine derivatives screened for anti-bacterial and anti-oxidant properties with Density Functional and Molecular Mechanic tools. Results in Chemistry, *5*, 100827.
CrossRef - Shruthi, T. G., Subramanian, S., & Eswaran, S. (2020). Design, synthesis and study of antibacterial and antitubercular activity of quinoline hydrazone hybrids. Heterocyclic Communications, *26*(1), 137–147.
CrossRef - Sri, C. D., & Rao, B. S. (2025). Updates on Intrinsic Medicinal Chemistry of 1,4-dihydropyridines, Perspectives on Synthesis and Pharmacokinetics of Novel 1,4-dihydropyrimidines as Calcium Channel Blockers: Clinical Pharmacology. Current Topics in Medicinal Chemistry, *25*(1), 1–20.
CrossRef - Sun, A., Li, Y., Wang, L., Zhang, M., Zhao, L., & Li, D. (2023). Optimization and antifungal activity of quinoline derivatives linked to chalcone moiety combined with FLC against Candida albicans. European Journal of Medicinal Chemistry, *249*, 115117.
CrossRef - Taha, M., Sultan, S., Imran, S., Rahim, F., Zaman, K., Wadood, A., Khan, F., & Khan, K. M. (2019). Synthesis of quinoline derivatives as diabetic II inhibitors and molecular docking studies. Bioorganic & Medicinal Chemistry, *27*(18), 4081–4088.
CrossRef - Taheri, S., Nazifi, M., Mansourian, M., Hosseinzadeh, L., & Shokoohinia, Y. (2019). Ugi efficient synthesis, biological evaluation and molecular docking of coumarin-quinoline hybrids as apoptotic agents through mitochondria-related pathways. Bioorganic Chemistry, *91*, 103147.
CrossRef - Teja, C., & Khan, F. R. N. (2020). Radical transformations towards the synthesis of quinoline: A review. Chemistry – An Asian Journal, *15*(24), 4153–4167.
CrossRef - Tian, G., & Zhang, S. (2023). Recent advances in 1,2,3- and 1,2,4-triazole hybrids as anti-microbials and their SAR, a critical review. European Journal of Medicinal Chemistry, *247*, 115004.
CrossRef - Tilekar, K., Kale, M. B., Upadhaya, D., & Kumar, V. (2022). Current status and future prospects of molecular hybrids with thiazolidinedione (TZD) scaffold in anti-cancer drug discovery. Journal of Molecular Structure, *1265*, 133330.
CrossRef - Trivedi, H. D., Patel, K. D., & Patel, H. D. (2022). Synthesis, molecular modeling, ADMET and, fastness studies of some quinoline encompassing pyrimidine azo dye derivatives as potent anti-microbial agents. Chemical Data Collections, *39*, 100870.
CrossRef - Upadhyay, A., Kushwaha, P., Gupta, S., Dodda, R. P., Ramalingam, K., Kant, R., & Srivastava, S. K. (2018). Synthesis and evaluation of novel triazolyl quinoline derivatives as potential antileishmanial agents. European Journal of Medicinal Chemistry, *154*, 172–181.
CrossRef - Van de Walle, T., Boone, M., Van Puyvelde, J., Combrinck, J., Smith, P. J., Chibale, K., & D’hooghe, M. (2020). Synthesis and biological evaluation of novel quinoline-piperidine scaffolds as antiplasmodium agents. European Journal of Medicinal Chemistry, *198*, 112330.
CrossRef - Venkatarao, V., & Subramanian, S. (2019). Synthesis and biological evaluation of chalcone fused quinoline derivatives as anti-cancer agents. Chemical Data Collections, *22*, 100245.
CrossRef - Walayat, K., Imran, M., Iqbal, S., & Sultana, S. (2023). Recent advances in the piperazine-based anti-viral agents: A remarkable heterocycle for anti-viral research. Arabian Journal of Chemistry, *16*(4), 104559.
CrossRef - Yadav, P., & Shah, K. (2021). Quinolines, a perpetual, multipurpose scaffold in medicinal chemistry. Bioorganic Chemistry, *109*, 104639.
CrossRef - Zeleke, D., Eswaramoorthy, R., Belay, Z., & Melaku, Y. (2020). Synthesis and antibacterial, antioxidant, and molecular docking analysis of some novel quinoline derivatives. Journal of Chemistry, *2020*, 1324096.
CrossRef - Zhang, G. F., Liu, X., Zhang, S., Pan, B., & Liu, M. L. (2018a). Ciprofloxacin derivatives and their anti-bacterial activities. European Journal of Medicinal Chemistry, *146*, 599–612.
CrossRef
Accepted on: 23 Nov 2025
Second Review by: Dr. Devendra Pathak
Final Approval by: Dr. Ioana Stanciu
















