Antioxidant Assay Guided Separation of the Methanol Stem Fraction of Binahong (Anredera Cordifolia) using Cyclic Voltammetry Method
Noviany, Ratu Dwi Gustia Rasyidi and Andi Setiawan
Department of Chemistry, Faculty of Matematics and Natural Sciences, University of Lampung, 35145, Bandar Lampung, Indonesia.
Corresponding Author E-mail: noviany@fmipa.unila.ac.id
DOI : http://dx.doi.org/10.13005/ojc/330124
The aim of this study was to identify and characterize groups of antioxidant active naturally from methanol fraction of the stem of Anredera cordifolia throughantioxidant assay-guided separation. The methanol fraction (MF) displayed the highest antioxidant activity, compared to ethyl acetate fraction (EAF) and hexane fraction(HF) using cyclic voltammetry method. Ascorbic acid was used as a positive control in this research.The repeated fractionation of active MF by MPLC and HPLC analysis yielded a constituent with strong antioxidant property. The active constituent(RNV-1) was predicted as a flavonoid type based on the UV spectrum data. The antioxidant activity results exhibited that RNV-1had a coefficient ofantioxidant activity (K) sligtly higher than K ofascorbat acid with the values of 0.055 and 0.052 respectively. It was found that RNV-1 demonstrated anexcellent antioxidant activity in comparison with the standard antioxidant activity through antioxidant assays-guided separation. The results obtained suggested that the stem of A. cordifolia can be used potentially asa source of natural antioxidants by contributing beneficial health effects.
KEYWORDS:Antioxidant assay-guided separation; Anredera cordifolia; binahong; flavonoid; cyclic voltammetry
Download this article as:| Copy the following to cite this article: Noviany N, Rasyidi R. D. G, Setiawan A. Antioxidant Assay Guided Separation of the Methanol Stem Fraction of Binahong (Anredera Cordifolia) using Cyclic Voltammetry Method. Orient J Chem 2017;33(1). |
| Copy the following to cite this URL: Noviany N, Rasyidi R. D. G, Setiawan A. Antioxidant Assay Guided Separation of the Methanol Stem Fraction of Binahong (Anredera Cordifolia) using Cyclic Voltammetry Method. Orient J Chem 2017;33(1). Available from: http://www.orientjchem.org/?p=28445 |
Introduction
Interest in plant natural products is certainly undergoing a renaissance at the present time. Many plants have been investigated in order to identify them depending on the amount of phytochemicals they have and on their potential use. Recently, the phenolic compounds particularly flavonoids have received increasing attention among other phytochemicals, due to their wide distribution in the plant kingdom and the various of their biologicalactivities, such anti-inflammatory,antithrombotic, antiviral, and hepatoprotective as well as their antioxidant activities1,2. Furthermore, clinical studies have suggested a positive effect offlavonoids in human health and nutrition, such as adecreased risk of heart disease, hormonally dependentcancers, menopausal symptoms, osteoporosis, and cardiovascular disease3. Diets high in flavonoids, fruits, and vegetables are protective against a variety ofdiseases, in particular cardiovascular disease, and some types of cancer. Antioxidants and dietary fiber are believed to be the principalnutrients responsible for these protective effects.The numerous human health related properties of flavonoidsthat widely described inepidemiological studies, are mainly based on their antioxidant activities.Antioxidants and dietary fiber are believed to be the principalnutrients responsible for these protective effects4. Currently,extensive research on natural antioxidants have been carried out by many researchers5-10. The potential antioxidant source can be found also inAnredera cordifolia plant. A. cordifoliais one of herbal medicine plant belonging of family Basellaceae. This plant is native to Southern America and is widespread in China, Korea, and Indonesia. The local names of this plant are binahong (Indonesia), Dheng shan chi (China), and madeira vine (England)11. All parts of A. cordifoliaincluding leaves, stems, roots and flowers are utilized in folk medicine, but leaves is the most often used. Wahyuni12 reported that the leaves extracts of A. cordifoliahave been shownto produce a variety of compounds such as saponin, alkaloid, phenolic, and flavonoid which exhibited some interesting effects such as antibaterial, anti-inflammation, and antioxidant properties. However, no previous research on the antioxidant activity of the chemical components from the stem part of A. cordifoliacould be found in the literature.Since this plant has been a valuable traditional remedy for treating infectious diseases therefore, more studies are still needed to further verify the potential of this plant as useful source of antioxidant chemicals. The antioxidant determination of extracts, fractions, and/or components is currently maintained mainly by DPPH assay. Using an electroanalytical approach for detection of their antioxidant activity is rare13. Electrochemicalprocedures representadvantageous possibility to measurea reduction and oxidation processesin organic compounds that can be helpful in how thesecompounds are metabolised by living organisms14.Recenlty, Keffous et al15 reported the antioxidant activity of Limoniastrum feei aqueousextract by using cyclic voltammetrymethod.The results showed that the cyclic voltammetry of the extract indicates one oxidation irreversible peak at approximately 300–320 mV/(Ag/AgCl). Based on those views, in the present study we sought to evaluate the antioxidant activityof fraction and the isolatedconstituent from the stem of A. cordifolia through antioxidant assay-guided separation by using cyclic voltammetry method. Materials and Methods Plant material The stems of A. cordifoliawere collected in August 2015 fromKompleks Perumnas Way Kandis in Bandar Lampung, Indonesia. The identity of the plant specimen was identified at the Herbarium Bogoriense, Research Centre for Biology, Indonesia Institute of Sciences Bogor, Indonesia and a voucher specimen was deposited at the herbarium. Extraction and isolation The dried and powdered stems (500 g) of A. cordifolia were extracted with n- hexane,ethyl acetate, and methanol sequentially at room temperature for one week with occasional maceration.Extraction was repeated three times and the fractions were filtered and evaporated using a rotary evaporator at 40oC. The weightof each fractions were 5.0 g, 4.5 g, and 1.3 g respectively. All the fractions obtained were stored at −20 °C for further analysis.The methanol fraction(MF) was proceeded to fractionation due to its active properties in antioxidant assay using cyclic voltammetry method. The MF was subjected to MPLC (Buchi/Sepacoterm) with photo diode array(PDA) detector. The analysis were performed with a Cosmosil 75C18-OPN C18 column where methanol-water were used as the mobile phase in a gradient mode (70%-30%) at a flow rate of 1,0 mL/min. The separation processes were repeated for five timesand each fraction was analyzed by HPLC (Shimadzu/LC-20A Prominence). The analysis were performed with a Cosmosil column (150 x 4,6 mm) as follows:methanol˗water (7:3) was used as the mobile phase;the flow-rate of the mobile phase was 1,0 mL/min;the effluent was monitored by a wavelength absorbance detector. Characterization of target compound was carried out using Spectroscopy UV-Vis analysis (Agilent UV-Vis Cary 100 Spectrofotometer). Antioxidant Assay All the fractions obtained were evaluated for their antioxidant activity by using cyclic voltammetry method as described by Masek et al14. The most active fractionwas selected to further fractionate with MPLC analysis. The cyclic voltammetry(CV) method was used,employing an potensiostat-galvanostat analytical unit (eDAQ Pty Ltd) with three-electrode system.Platinumwas used as anode and auxiliary electrode. The potential of thetested electrode was measured versus a silver/silver chloride referenceelectrode.Potassium chlorida from Sigma Aldrichwas used asupporting electrolyte and ascorbic acid(Merck)was used as positive control.Prior to measurements,all the solutions were deoxidised with nitrogen. Theeffect of scan rate on the electrooxidation of tested components in an anhydrousmedium was assessed.
Results and Discussion
Isolation and Characterization All the fractionsincluding n-hexane (HF), ethyl acetate (EF), and methanol (MF) were screened for theirantioxidant activity using CVtechnique. The results demonstrated that the MFhas the most active property in antioxidant assay among HF and EF. Therefore, the MF was subjected to fractionation further and purification by MPLC and HPLC analyses. The results of MPLC separation gave 19 sub-fractions. The major component peaks was produced at the retention time of 2.5 minutes with UV detection at 220 and 254 nm (Fig.1). Each sub-fractions obtained was tested for its antioxidant activity and the results showed that sub-fraction 4, 5, and 6 displayed the higher antioxidant activity compared with other fractions.
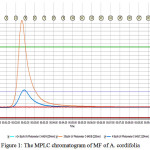 |
Figure 1: The MPLC chromatogram of MF of A. cordifolia |
Further purification of sub-fractions 4, 5, and 6 were conducted by crystalization for 1-2 days. Compound RNV-1 (10 mg) was afforded by recrystalization from sub-fraction 5. The HPLC analysis of isolated RNV-1 exposed the presence of major constituent peak with aretention time of 2.67 minutes which indicated that the constituent is quite pure for further analysis by spectroscopy method. The HPLC chromatogramof RNV-1 can be shown in Fig.2
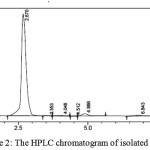 |
Figure 2: The HPLC chromatogram of isolated RNV-1 |
The UV spectrum of RNV-1 revealed over the scan range of 200–400 nm (Fig.3). RNV-1 exhibits a maximum absorbance at 216 nm with additional absorbances at 260 nm and 323 nm. The results indicated that RNV-1 has a conjugated system in its structure that could be suggested as phenolic type structure such as flavonoids. Accordingly, the presence of flavonoid compounds was identified in this research based on the shift in absorption maximum caused by addition of sodium acetate/sodium hidroxide. The absorption characteristics of a flavonoid compound may vary depending on the type of complex it forms with the shift reagents used15,16. A bathochromic shift of 8 nm in band II was observed after addition of sodium hidroxide, implying the presence of 7-OH in flavones or flavonols.Similarly, a red shift of 63 nm in band I can indicate the flavonoids to be 3-hydroxy flavones or 3,5-dihydroxy flavones16.Nevertheless, the completed structure analysis is still on going in this research, to confirm the exact structure of purified compound.
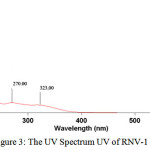 |
Figure 3: The UV Spectrum UV of RNV-1 (MeOH) |
Antioxidant Activity In this study, the electrode reactions characterizing electrochemical oxidationof tested components at the platinum electrode were evaluated by CV. Half-wave potential of a peak in a cyclic voltammogramcorrespondsto a potential of a peak and is characteristic for each of the subsequent step of theinvestigated electrode reaction14. Cyclic voltammograms recorded in MF solution (red-curve) (Fig. 5) show the small wave appearin the potential range from 0.25 to 0.8 V. Thiswave is relativelylow compare with the peak attributed to theflavonoids oxidation as reported by Masek et al14, it can be caused by oxidation of impuritiessuch as water and other organic substances in MF17.
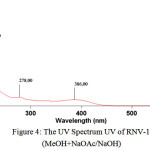 |
Figure 4: The UV Spectrum UV of RNV-1 (MeOH+NaOAc/NaOH) |
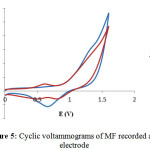 |
Figure 5: Cyclic voltammograms of MF recorded at Pt electrode |
Cyclic voltammograms recorded in some sub-fraction 4 (sF-4) (blue-curve), 5 (sF-5) (red-curve), and 6 (sF-6) (green-curve) of MF (Fig. 6) exhibitedthat sF-5 has the most active in antioxidant assay. A peak which characterize atleast one electrode step of sF-5 electrooxidation in thepotential range from 1.0 to 1.5 V in voltammogram.
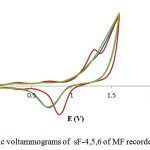 |
Figure 6: Cyclic voltammograms of sF-4,5,6 of MF recorded at Pt electrode Click here to View figure |
In the case of the isolated compound, RNV-1, the cyclic voltammogram recorded in different concentrations (4, 8, and 12 ppm) showed peaksindicating one electrode of RNV-1 electrooxidation in each concentrationswith similarly in thepotential range from 1.0 to 1.5 V (Fig.7)
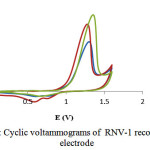 |
Figure 7: Cyclic voltammograms of RNV-1 recorded at Pt electrode |
Cyclic voltammogram recorded in positive control, ascorbat acid (Fig.8), exhibited that ascorbat acid has less active than with RNV-1 in antioxidant assay. This results also supported by determining a coefficient ofantioxidant activity (K) for both isolated compound and ascobat acid. As expected, RNV-1have demonstratedexcellent antioxidant activity in comparison with the standard antioxidant, ascobat acid.
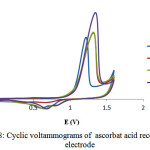 |
Figure 8: Cyclic voltammograms of ascorbat acid recorded at Pt electrode |
Conclusions The current study was projected to assess the antioxidant activity of fractions and purified compound from the stem of A. cordifolia by using cyclic voltammetry method. The MF exhibited the highest antioxidant property, among the fractions. The antioxidant effects of MF justified the folkloric use of this medicinal plant by the natives in clinical management. Antioxidant assay-guided fractionation and purification of MF resulted in the identification of isolated RNV-1 that was suggested as the flavonoid type compound. The antioxidant activity of RNV-1 was verified the potential of this plant as useful source of antioxidant chemicals in treatment of oxidative damages. The structure elucidation of RNV-1 by using various spectroscopy methods is still in progress. Acknowledgments The authors are grateful to Directorate of Research and Community Services, Directorate General of Higher Education, The Ministry of Research, Technology and Higher Education, Republic of Indonesia that provide fund for this project to be undertaken through Hibah Pascasarjana (Postgraduated Research Grant) Scheme 2016 with contract number of 963/UN26.21/8/PP/2016, 28 October 2016. References
- Marin, F.R.; Frutos, M.J.; Perez-Alvarez, J.A.; Martinez-Sanchez, F.; Del Rio, J.A.Studies in Natural Products Chemistry.2002, 26, 741-778. CrossRef
- Shahid, M. A; Shahzad, F.; Sobia, A.; Sahai, T.; Tripathi A.; Singh, H.M.; Khan and Umesh.Anti-Infective Agents in Med. Chem.2009, 8,211-225. CrossRef
- Du, H; Yubi, H., Yixiong, T. Appl Microbiol Biotechnol. 2010, 86, 1293-1312. CrossRef
- Grotewold, E. The Science of Flavonoids,Published by Springer Science Business Media, Inc.2006, 1-3. CrossRef
- Saito, K.; Kohno, M.; Yoshizaki, F.; Niwano, Y. Plant Foods Hum. Nutr.2008, 63 (2), 65-70. CrossRef
- Delgado A. J.; Gamero S. E.; Vald´es S. E.; Gonz´alez-G. D. Food Control.2012,24 (1-2), 136-141. CrossRef
- Sreelatha, S.; Padma, P. R. Plant Foods Hum. Nutr.2009, 64 (4), 303-311. CrossRef
- Onoja, S. O.; Anaga, A. O.Int. J. Pharm. Phytochem. Res.2015,7(2), 340-346.
- Arullappan, S.; Rajamanickam, P.; Thevar, N.; Narayanasamy, D.; Yee, Y. H.; Kaur, P.; Mahandan, M.Trop. J. Pharma. Res.2015,14 (7), 1199-1205. CrossRef
- Sudha, A.; Srinivasan, P. BioMed Res.Int. 2014, 1-10. CrossRef
- Lestari, D.; Sukandar, E. Y.; Fidrianny, I.Int. J. Pharm. Clin. Res.2015,7(6), 435-439.
- Wahjuni, S..Ind. J. Biomed. Sci.2014,8 (1), 4-7.
- Hanuštiaka, P.; Mikelova, R.; Potěšila, D.; Hodek, P.; Stiborova, M.; Kizek, R. Biomed. Papers.2005,149 (1).
- Masek, A.; Zaborski, M.; Chrzescijanska, E.Food Chem.2011, 127, 699-704. CrossRef
- Keffous, F.; Belboukhari, N.; Sekkoum, K.; Djeradi, H.; Cheriti, A.; Aboul,E.H.Y. Cogent Chemistry.2016, 2, 118-141. CrossRef
- Schütz, K.; Kammerer, D.R.; Carle, R.; Schieber, A. Rapid Commun. Mass Spectrom.2005, 19, 179-186. CrossRef
- Mabry, T.J.; Markham, K.R.; Thomas, M.B. The Systematic Identification of Flavonoids, Published bySpringer-Verlag New York, USA.1970,354
- Zieja, J.; Gadomska, T. J.; Stojek, Z. Electroanalysis.2001, 13(8/9), 621-625. CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.









